Abstract
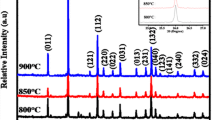
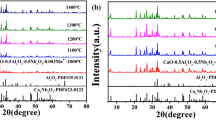
In this book chapter, the divalent europium-activated alkaline-earth-metal pyrophosphate M2P2O7 (M = Ca, Sr, Ba) phosphors were effectively prepared via a conventional high temperature sintering reaction technique in a reducing atmosphere at 900 ℃. The synthesized materials have been systematically investigated using powder X-ray diffraction. The XRD pattern exhibit that Ca2P2O7 has a β-phase, Sr2P2O7 has an α-phase, Ba2P2O7 has a β-phase and the crystal structure of the M2P2O7 (M = Ca, Sr, Ba) hosts was unaffected with the introduction of Eu2+ ions. The nuclear magnetic resonance (NMR), infrared (IR) and Raman spectroscopy of β-Ca2P2O7, α-Sr2P2O7 and γ-Ba2P2O7 were reported and discussed in accordance with the structural peculiarities of Alkaline earth phosphates. The luminescence properties of Eu2+ in these phosphate materials were explored in detail in both vacuum ultraviolet (VUV) and ultraviolet (UV) regions. The emission spectra of β-Ca2P2O7: 1%Eu2+, α-Sr2P2O7: 1%Eu2+ and γ-Ba2P2O7:1%Eu2+ exhibit a large asymmetric band blue-emitting under the excitation of 320 nm, caused by the 4f65d1 → 4f7[8S7/2] transition of Eu2+ at room temperature. The excitation spectra display a broad and intense absorption in the 250–400 nm wavelengths region, which is matched with the blue emission band using for emitting n-UV (NUV) LED chips (360–400 nm). The above results indicate that diphosphate phosphors could be promising for generating blue-materials for white LED applications.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Blasse, G.J.: Energy transfer between inequivalent Eu2+ ions. Solid State Chem. 62, 207–211 (1986). https://doi.org/10.1016/0022-4596(86)90233-1
Briche, S., Zambon, D., Chadeyron, G., Boyer, D., Dubois, M., Mahiou, R.: Comparison of yttrium polyphosphate Y(PO3)3 preparedby sol–gel process and solid-state synthesis. J. Sol-Gel Sci. Technol. 55, 41–51 (2010). https://doi.org/10.1007/s10971-010-2211-z
Chen, K.J., et al.: An investigation of the optical analysis in white light-emitting diodes with conformal and remote phosphor structure. IEEE J. Disp. Technol. 9, 915–920 (2013). https://doi.org/10.1109/JDT.2013.2272644
Derbel, M., Mbarek, A., Fourati, M.: Photoluminescence properties of CdSrP2O7: Eu2+ blue phosphor for White LED applications. Optik 127, 5870–5875 (2016). https://doi.org/10.1016/j.ijelo.2016.04.020
ElBelghitti, A.A., Elmarzouki, A., Boukhari, A., Holt, E.M.: σ-dibarium pyrophosphate. Acta Cryst. C 51, 1478–2148 (1995). https://doi.org/10.1107/S0108270195001739
Hagman, L.O., Jansson, I., Magneli, C.: The crystal structure of α-Sr2P2O7. Acta Chem. Scand. 22, 1419–1429 (1968)
Hami, W., Daoud, M., Zambon, D., Elaatmani, M., Zedzouti, A.: Synthesis and characterization of AgYP2O7 pyrophosphate activated with Tb3+, Sm3+ and Dy3+ ions. Inorg. Chem. Commun. 102, 192–198 (2019). https://doi.org/10.1016/j.inoche.2019.02.013
Hami, W., et al.: Synthesis, structural characterization and luminescent properties of Tb3+-doped AgLaP2O7 phosphors. Ceram. Int. 44, 19184–19190 (2018). https://doi.org/10.1016/j.ceramint.2018.06.271
Hao, Z., et al.: Phase dependent photoluminescence and energy transfer in Ca2P2O7: Eu2+, Mn2+ phosphors for white LEDs. J. Lumin. 128, 941–944 (2008). https://doi.org/10.1016/j.jlumin.2007.11.035
Hee-Suk, R., et al.: luminescence properties of Ca5(PO4)SiO4:Eu2+ green phosphor for near UV-based White LED. Mater. Lett. 70, 37–39 (2012). https://doi.org/10.1016/j.matlet.2011.11-088
Ma, X., et al.: Synthesis and luminescence phosphor Ba3LaK(PO4)3F for n-UV White-LEDS. Polyhedron 119, 223–226 (2016). https://doi.org/10.1016/j.poly.2016.09.001
Mahajan, R., Kumar, S., Praksh, R., Kumar, V., Choudhary, R.J., Phase, D.M.: X-ray photoemission and Spectral investigations of Dy3+ activated magnesium pyrophosphate phosphors. J. Alloys Compd. 777, 562–571 (2019). https://doi.org/10.1016/j.jallcom.2018.10.355
Mbarek, A.: Synthesis and visible-near-infrared photoluminescence emission of Er3+-doped LnP5O14 (Ln=La, Gd) ultraphosphates. Inorg. Chem. Commun. 98, 1–6 (2014). https://doi.org/10.1016/j.inoche.2018.09.031
Moutataouia, M., Zambon, D., Boyer, D., Lamire, M., El Ammari, L., Mahiou, R.: Crystallographic and optical properties of Eu3+ doped K2Na3P3O10 triphosphate. J. Alloys Compd. 748, 206–215 (2018). https://doi.org/10.1016/j.jallcom.2018.02.284
Sun, J., Zhang, X., Li, T.: New Eu2+-activated borophosphate phosphors Ba3(ZnB5O10)PO4 for near-ultraviolet-pumped white light-emitting diodes. Mater. Lett. 212, 343–345 (2018). https://doi.org/10.1016/j.matlet.2017.10.125
Webb, N.C.: Acta Crystallogr. 21, 942–948 (1966). https://doi.org/10.1107/S0365110X66004225
Zhang, Z., Wang, Y.: Photoluminescence of Eu2+-doped CaMgSi2xO6+2x (1.00⩽x⩽1.20) phosphors in UV–VUV region. J. Lumin. 128, 383–386 (2008). https://doi.org/10.1016/j.jlumin.2007.09.006
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this paper
Cite this paper
Derbel, M., Mbarek, A. (2022). Blue-Emitting Phosphors M2P2O7:1%Eu2+ (M = Ca, Sr, Ba) Pyrophosphates for LED Lighting. In: Bouraoui, T., et al. Advances in Mechanical Engineering and Mechanics II. CoTuMe 2021. Lecture Notes in Mechanical Engineering. Springer, Cham. https://doi.org/10.1007/978-3-030-86446-0_26
Download citation
DOI: https://doi.org/10.1007/978-3-030-86446-0_26
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-86445-3
Online ISBN: 978-3-030-86446-0
eBook Packages: EngineeringEngineering (R0)