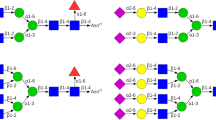
Abstract
Human interferon-gamma (hIFN\(\gamma \)) is a key immunomodulating secretory glycoprotein. Although glycosylation is not necessary for its activity, it does affect the physico-chemical properties of the cytokine. Recently, a technology was developed for the secretory expression of glycosylated hIFN\(\gamma \) and its highly prone to aggregation mutant K88Q in insect cells. In addition, the proteins were labeled with specific tag peptides added to their N-termini. It was experimentally observed, that the obtained fusion proteins have significantly reduced biological activity and when glycosylated, they were resistent to enterokinase action and the tag could not be removed. Here we report the development of in silico models of glycosylated His\(_6\)-FLAG-hIFN\(\gamma \) fusion proteins and employ long-scale molecular dynamics (MD) simulations to explain these unexpected experimental results and to study in detail the effect of the tags and glycosylation on the structure and dynamics of the fusion glycoproteins.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Abraham, M.J., Murtola, T., Schulz, R., Páll, S., Smith, J.C., Hess, B., Lindahl, E.: Gromacs: high performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 1–2, 19–25 (2015)
Ailor, E., Takahashi, N., Tsukamoto, Y., Masuda, K., Rahman, B.A., Jarvis, D.L., Lee, Y.C., Betenbaugh, M.J.: N-glycan patterns of human transferrin produced in trichoplusia ni insect cells: effects of mammalian galactosyltransferase. Glycobiology 10(8), 837–847 (200)
Arakawa, T., Hsu, Y., Chang, D., Stebbing, N., Altrock, B.: Structure and activity of glycosylated human interferon-gamma. J. Interferon Res. 6(6), 687–695 (1986)
Arakawa, T., Hsu, Y., Yphantis, D.: Acid unfolding and self-association of recombinant escherhia coli derived human interferon \(\gamma \). Biochemistry 26, 5428–5432 (1987)
Berendsen, H.J.C., Postma, J.P.M., van Gunsteren, W.F., DiNola, A., Haak, J.R.: Molecular dynamics with coupling to an external bath. J. Chem. Phys. 81, 3684 (1984)
Bohne-Lang, A., von der Lieth, C.W.: Glyprot: in silico glycosylation of proteins. Nucleic Acids Res. 33(suppl\(\_\)2), W214–W219 (2005)
Bussi, G., Donadio, D., Parrinello, M.: Canonical sampling through velocity rescaling. J. Chem. Phys. 126(1), 014101 (2007)
Ealick, S.E., Cook, W.J., Vijay-Kumar, S., Carson, M., Nagabhushan, T.L., Trotta, P.P., Bugg, C.E.: Three-dimensional structure of recombinant human interferon-\(\gamma \). Science 252, 698–702 (1991)
Essmann, U., Perera, L., Berkowitz, M.L., Darden, T., Lee, H., Pedersen, L.G.: A smooth particle mesh Ewald method. J. Chem. Phys. 103(19), 8577–8593 (1995)
Farrar, M.A., Schreiber, R.D.: The molecular cell biology of interferon-gamma and its receptor. Annu. Rev. Immunol. 11(1), 571–611 (1993)
Grzesiek, S., Dobeli, H., Gentz, R., Garotta, G., Labhardt, A., Bax, A.: H-1, c-13, and n-15 NMR backbone assignments and secondary structure of human interferon-gamma. Biochemistry 31(35), 8180–8190 (1992)
Guvench, O., Mallajosyula, S.S., Raman, E.P., Hatcher, E., Vanommeslaeghe, K., Foster, T.J., Jamison, F.W., MacKerell, A.D.: Charmm additive all-atom force field for carbohydrate derivatives and its utility in polysaccharide and carbohydrate-protein modeling. J. Chem. Theory Comput. 10, 3162–3180 (2011)
Hess, B.: P-lincs: a parallel linear constraint solver for molecular simulation. J. Chem. Theor. Comput. 4(1), 116–122 (2008)
Hockney, R., Goel, S., Eastwood, J.: Quiet high-resolution computer models of a plasma. J. Comput. Phys. 14(2), 148–158 (1974)
Huang, J., Rauscher, S., Nawrocki, G., Ran, T., Feig, M., de Groot, B.L., Grubmüller, H., MacKerell Jr., A.D.: Charmm36m: an improved force field for folded and intrinsically disordered proteins. Nat. Methods 14, 71–73 (2016)
James, D.C., Freedman, R.B., Hoare, M., Ogonah, O.W., Rooney, B.C., Larionov, O.A., Dobrovolsky, V.N., Lagutin, O.V., Jenkins, N.: N-glycosylation of recombinant human interferon-\(\gamma \) produced in different animal expression systems. Bio/Technology 13, 592–596 (1995)
Jo, S., Song, K.C., Desaire, H., MacKerell, A.D., Im, W.: Glycan reader: automated sugar identification and simulation preparation for carbohydrates and glycoproteins. J. Comput. Chem. 32(14), 3135–3141 (2011)
Krachmarova, E., Tileva, M., Lilkova, E., Petkov, P., Maskos, K., Ilieva, N., Ivanov, I., Litov, L., Nacheva, G.: His-flag tag as a fusion partner of glycosylated human interferon-gamma and its mutant: Gain or loss? BioMed Res. Int. 2017, 3018608 (2017)
Loss, A., Lütteke, T.: Using NMR data on glycosciences.de. Methods Mol. Biol. 1273, 87–95 (2015)
Parrinello, M., Rahman, A.: Crystal structure and pair potentials: a molecular-dynamics study. Phys. Rev. Lett. 45, 1196 (1980)
Parrinello, M., Rahman, A.: Polymorphic transitions in single crystals: a new molecular dynamics method. J. Appl. Phys. 52, 7182 (1981)
Petkov, P., Lilkova, E., Ilieva, N., Nacheva, G., Ivanov, I., Litov, L.: Computational modelling of the full length hifn-\(\gamma \) homodimer. In: Lirkov, I., Margenov, S. (eds.) Large-Scale Scientific Computing, LSSC 2017, Lecture Notes in Computer Science, vol 10665, pp. 544–551. Springer, Cham (2018)
Qiao, L., Lee, K.J., Ko, K.: Characterization of the glycan structures of glycoprotein ga733-fc expressed in a baculovirus-insect cell system. Bull. Korean Chem. Soc. 36(1), 139–149 (2015)
Rendić, D., Wilson, I.B.H., Paschinger, K.: The glycosylation capacity of insect cells. Croat. Chem. Acta 81(1), 7–21 (2008)
Sareneva, T., Cantell, K., Pyhälä, L., Pirhonen, J., Julkunen, I.: Effect of carbohydrates on the pharmacokinetics of human interferon-gamma. J. Interferon Res. 13(4), 267–9 (1993)
Sareneva, T., Pirhonen, J., Cantell, K., Julkunen, L.: N-glycosylation of human interferon-\(\gamma \): glycans at asn-25 are critical for protease resistance. Biochem. J. 308(1), 9–14 (1995)
Sareneva, T., Pirhonen, J., Cantell, K., Kalkkinent, N., Julkunen, L.: Role of n-glycosylation in the synthesis, dimerization and secretion of human interferon-\(\gamma \). Biochem. J. 303, 831–840 (1994)
Thiel, D., le Du, M.H., Walter, R., D’Arcy, A., Chène, C., Fountoulakis, M., Garotta, G., Winkler, F., Ealick, S.: Observation of an unexpected third receptor molecule in the crystal structure of human interferon-\(\gamma \) receptor complex. Structure 8, 927–936 (2000)
Acknowledgements
This work was supported in part under the Programme for young scientists’ career development at the Bulgarian Academy of Sciences (DFNP-17-146/2017) and under Grants DN-11/20/2017 and DNTS-Austia-01-2/2013 of the Bulgarian Science Fund. Computational resources were provided by the supercomputer Avitohol@BAS and the HPC Cluster at the Faculty of Physics of Sofia University “St. Kl. Ohridski”.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this paper
Cite this paper
Lilkova, E., Ilieva, N., Petkov, P., Krachmarova, E., Nacheva, G., Litov, L. (2021). Molecular Dynamics Simulations of His\(_6\)-FLAG-hIFN\(\gamma \) Fusion Glycoproteins. In: Georgiev, I., Kostadinov, H., Lilkova, E. (eds) Advanced Computing in Industrial Mathematics. BGSIAM 2018. Studies in Computational Intelligence, vol 961. Springer, Cham. https://doi.org/10.1007/978-3-030-71616-5_23
Download citation
DOI: https://doi.org/10.1007/978-3-030-71616-5_23
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-71615-8
Online ISBN: 978-3-030-71616-5
eBook Packages: Intelligent Technologies and RoboticsIntelligent Technologies and Robotics (R0)