Abstract
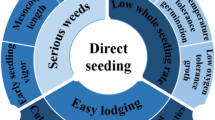
The changing climate and water availability strongly affect the current state of agricultural production. While the global temperature rises, the occurrence of extreme climatic conditions becomes erratic. This current scenario has driven the development of rice varieties and cultivation practices that require less water and favor mechanization. Although puddled transplanted rice has been more widely used in the past, direct seeding has been gaining popularity in recent years, especially due to its water- and labor-saving features. This technique allows full crop establishment from seeds that were directly sown in the field, thus avoiding puddling, transplanting, and maintaining standing water. Consequently, it offers promising positive environmental effects including decreasing the release of greenhouse gases and increasing water-use efficiency. Historically, rice varieties bred for transplanting are also used in direct seeding, which limits the maximum yield potential of field trials. The success of direct seeding relies strongly on the development of rice varieties with robust crop establishment. Anaerobic germination, seed longevity, and early seedling vigor are the key traits required to achieve this. This chapter expounds on the physiology, molecular mechanisms, genetics, and relevance of the enumerated traits for direct seeding. A brief discussion of breeding for rice varieties with improved germination under direct seeding is also provided.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Rice is grown on close to 160 million ha of land across the world, producing 700 million t of paddy. Notably, more than 90% of the worldwide rice supply is cultivated and consumed in Asia (Fig. 1). A significant volume of this production is contributed by small and marginal farm holdings that principally grow rice with limited inputs under an unpredictable water supply. Rice can be categorized into different types based on the topographical location where it is grown and the prevailing ecosystem. Topographically, rice is grouped into five main classes: upland, shallow lowland, mid-lowland, semi-deep, and deepwater ecosystems. Further, these ecosystems are classified as irrigated or rainfed based on the availability of irrigation water. In cases where water is maintained throughout most of the crop growth period (80%), the area is classified as irrigated. However, rainfed areas are those where the sole water source is rainfall and ponded water availability is uncertain.
2017 worldwide (a) milled rice production (tons) and (b) total rice consumption. (Data retrieved from http://ricestat.irri.org:8080/wrs2/, June 2018)
The start of the Green Revolution led to a marked change in plant architecture and cultivation practices that primarily suited highly productive irrigated environments. Several varieties of rice with promising yield potential and input responsiveness were developed and adapted across irrigated and rainfed rice-growing areas. Although this was advantageous in increasing grain yield, the consequences of the Green Revolution came with an environmental cost. As noted by Pingali (2012), the policy underlying the Green Revolution prompted misguided overuse of fertilizer and crop intensification in unbefitting environments. On a sizable scale, the environment is subjected to unintended consequences such as soil degradation, water-table diminution, and chemical runoff, which ultimately contribute to the escalating climate change.
This review aims to summarize crop establishment practices in rice in relation to climate change and water scarcity . We begin by describing the current state of climate change and the need for new crop management approaches to mitigate the worsening conditions. The review then summarizes major crop establishment methods in rice and demonstrates their suitability to varying growth scenarios. Further, the review discusses crop establishment in direct-seeded rice in detail covering relevant traits with their underlying physiology and genetics. Finally, we briefly describe the breeding approach to systematically include new traits in programs on direct-seeded rice breeding.
2 Climate Change and Water Scarcity
The state of the environment strongly affects the world’s agricultural capacity (FAO 2016). Climate change and water scarcity are two developing stories that confound the downside in agricultural production. Annually, a total of 3853 km3 of groundwater is withdrawn, 69% of which (i.e., 2769 km3) is used for agricultural irrigation while 19% and 12% are allocated for industry and municipalities, respectively (FAO 2016). The repercussions of climate change are believed to drastically affect the water and agricultural status of the world. Predictions suggest that climate change can elicit an increase in temperature, a shift in the patterns of precipitation, the occurrence of more extreme climatic events, and further water deficit (Barker 2009).
Climate is described as the environmental condition over a location in a span of 30 years. Temperature and precipitation are two of the most pressing factors that regulate Earth’s climate. Remarkably, over the past 100 years, an average increase of 0.74 °C has been recorded in the global surface temperature (IPCC 2007) as a result of the increase in atmospheric methane, nitrous oxide, and other greenhouse gases (GHGs). Since rainfall has a direct relationship with temperature, the amount of annual rainfall scales up with temperature. In the past 100 years, there has been an increase of 2% in overall precipitation ; however, because this has large regional discrepancies, the benefits have been limited (IPCC 2007). Moreover, the occurrence of extreme climatic events is increasing and this leads to a decline in rice production. Long-term experiments on rice yields coupled with crop simulation models have reported that the yield decline can partly be attributed to the fluctuating climatic conditions (Aggarwal 2008). Figure 2 illustrates the average yearly precipitation across different countries in 2011. Notably, some of the major rice-producing countries such as Vietnam, Myanmar, Thailand, the Philippines, Bangladesh, Nepal, India, Burundi, and Nigeria also have the highest precipitation rates (Kreft et al. 2016). In India, for example, it was estimated that the variability in temperature and rainfall would result in a 10–40% loss in agricultural production by 2080–2100 (Aggarwal et al. 2010). Projection models suggest that direct climate impacts on maize, soybean, wheat, and rice production can account for losses of 400–1400 Pcal (8–24% of the current total) (Elliott et al. 2014). As mentioned by Barker et al. (1998), the world must develop and advocate for policies, investments, and infrastructure that can adapt to the ever-changing climatic parameters.
Average yearly precipitation (cm) across different countries. (Image retrieved from https://nelson.wisc.edu, July 2018)
Water scarcity is a state when water is insufficient to meet the demand of all sectors of a particular place or demography. It has diverse origins, including decreasing water tables, declining water quality, faulty irrigation systems, and growing water competition from industrial and urban sectors (Bouman et al. 2007). It can be categorized into two major types: physical and economic water scarcity.
Physical water scarcity, in its simplest form, occurs when water resources are depleted; hence, the water supply is not able to meet human and environmental demand (Molden 2007; OCHA 2010). As further discussed by Molden (2007), physical scarcity is commonly associated with, but not limited to, arid areas. It also occurs in areas with an abundant water supply yet the resources (i.e., irrigation) are allocated only to certain sectors.
Economic water scarcity is due to the lack of capability to withdraw water given that there is a sufficient amount of reserve (Molden 2007; OCHA 2010). This relates to the lack of infrastructure to enable the withdrawal of water. However, this is also linked to the inequitable allocation of infrastructure; hence, the water supply is limited to certain groups. Signs indicative of economic water scarcity are inadequate infrastructure development, high cost of irrigation, fluctuating water availability, flooding, and long-term drought. Therefore, it is important to address the proper management of water resources to maximize water utility and prevent water contamination.
Physical and economic water scarcities are widespread across different countries. As of 2016, it was revealed that about four billion people suffer from water scarcity for at least 60 days in a year (Mekonnen and Hoekstra 2016). In Asia alone, it is expected that per capita water availability will decline across different countries by 2050 (Fig. 3). This decline could range from 1911 to 63,135 m3 for different countries (Kumar and Ladha 2011). Several reasons are reported to be causing this decline, such as booming population, decreasing water table, deteriorating water quality, inept irrigation, and rising competition with nonagricultural sectors (Kaur and Singh 2017).
Per capita water availability (in cubic meters) based on population growth rate among different rice-producing countries across 100 years (1950–2050). (Source: Kumar and Ladha 2011)
It is estimated that approximately four billion people worldwide are constrained by extreme water scarcity for blue water (fresh surface water and/or groundwater that is extracted from the earth and not returned due to evaporation into the atmosphere or incorporation into a product), of which more than half are from China and India (Mekonnen and Hoekstra 2016), two of the highest rice-producing Asian countries. Therefore, it can be expected that physical and economic water scarcities are going to induce a sharp deterioration in agriculture’s portion of freshwater allocation in Asia.
Water is also lost in the field throughout the cropping season because of factors such as surface runoff, seepage, percolation, evaporation, and transpiration (Bouman et al. 2007). However, only water loss through transpiration is considered to be productive since this is useful for plant growth and development. Although 1432 L of water are needed to produce 1 kg of rough rice (Rice Knowledge Bank 2017), only about 10% of the total absorbed water is used by the plant for its development (Chavarria and dos Santos 2012). To this end, a large portion of water input becomes unused, leading to a loss in farmers’ investments. In the present-day scenario of water scarcity, the puddled transplanted system of rice is becoming increasingly unsustainable. With more than 90% of global rice being cultivated and expended in Asia and the large water requirements of conventional rice cultivation systems, it is evident that water scarcity is going to severely affect rice production on the continent. The high water requirement of puddled transplanted rice makes it more vulnerable to water scarcity and demands more drastic changes in cultivation practices than in other crops.
3 Rice Establishment Methods
Historically, in Asia, direct seeding was a preferred cultivation practice over transplanting (Grigg 1974). Although transplanting was more labor- and input-intensive, it provided a yield advantage and was later adopted due to the increased availability of labor because of drastic population expansion. In the sections below, we describe the two main cultivation practices popular in Asia.
3.1 Puddled Transplanted Rice
Transplanting 20–30-day-old seedlings in puddled soil is currently the most preferred practice in Asia. Puddling decreases water seepage by compacting the soil during land preparation, which improves weed suppression, eases seedling establishment, and creates an anaerobic condition that increases soil nutrient availability (e.g., zinc, iron, and phosphorus) (Fanish 2016). Puddling ensures high crop growth and yield but it is not the most water- and labor-efficient option (Dixit et al. 2016). Rice requires two to three times more water than other cereals (Barker et al. 1998), especially when it is grown under puddled conditions lasting to about 80% of the crop’s growth duration. Moreover, repeated puddling damages soil properties such that the soil aggregates are dismantled, the permeability of the soil subsurface layers decreases, and, at shallow depth, hard-pans are formed. Also, puddled transplanted rice (PTR) requires longer crop duration due to the delay in plant maturity caused by transplanting shock and nursery time. Although transplanting is favored by low wages and adequate water supply, the decreasing labor and water availability in most rice-growing regions in Asia requires a shift in cultivation practices to make rice farming a sustainable and profitable enterprise.
3.2 Direct-Seeded Rice
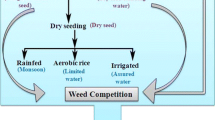
The current climate and social scenarios in Asia have led to increased efforts in developing rice varieties and cultivation practices requiring less water and favoring mechanization. These include operations such as direct seeding and alternate wetting and drying. Among all the water-saving technologies, dry direct seeding is the most water-efficient and it favors mechanization, which reduces labor requirements.
Direct seeding is the crop establishment practice from seeds that were directly sown in the field instead of transplanting seedlings grown from the nursery. It comes mainly in three forms: dry direct-seeded rice (dry-DSR), wet direct-seeded rice (wet-DSR), and water seeding. Kumar and Ladha (2011) described and differentiated these methods based on field conditions. Dry-DSR involves broadcasting, dibbling, or drilling of dry seeds on unpuddled soil, which was either conventionally tilled or not tilled at all. Meanwhile, wet-DSR is done by sowing pregerminated rice seeds onto or into puddled soil and is known as aerobic and anaerobic wet-DSR, respectively. Lastly, water seeding uses pregerminated seeds that are broadcast onto the field with standing water. In this case, the field may be either puddled (wet-water seeding) or unpuddled (dry-water seeding).
DSR is considered as an opportunity to advance rice production practices in water-scarce areas into a high-water-use-efficient cultivation practice (Fanish 2016). Through dry direct seeding, three basic field operations are avoided: puddling, transplanting, and maintaining 4–5 cm of standing water throughout the season. Further, PTR fields are one of the biggest sources of greenhouse gases, particularly methane. As reported by Pathak et al. (2013), in the districts of Punjab, DSR decreased total global warming potential by about 33%, from 2.0 to 4.6 t CO2 eq./ha to 1.3–2.9 t CO2 eq./ha. In DSR, the production of both methane and carbon dioxide was less than that in PTR. This shows the promising positive effects of DSR on the environment.
DSR is also known for its water- and labor-saving attributes. It is reported to save around 30% of water (Fanish 2016) and 11.2% of labor costs (Akhgari and Kaviani 2011). Apart from this, the labor requirement for DSR is spread out through the season, promoting mechanization and the use of family labor instead of depending on hired labor. This makes rice farming more profitable for farmers and also allows continued operations throughout the season despite labor unavailability. Furthermore, DSR saves the plants from transplanting injury; hence, the plants reach physiological maturity in fewer days and this leads to early crop maturity.
In general, PTR varieties are also used in direct seeding. The unavailability of proper varieties developed for direct seeding is a major constraint to exploiting its maximum yield potential. Historically, previous approaches to the improvement of crop establishment in DSR involved refining cultural practices rather than rice varietal improvement. Ultimately, the success of DSR relies heavily on breeding for varieties with anaerobic germination (AG) tolerance, seed longevity, early seedling vigor, and the ability to germinate from deep soil.
4 Traits, Physiology, Genetics, and Breeding
4.1 Anaerobic Germination
Poor crop establishment remains a concern in areas that experience flooding after sowing and where fields are not properly leveled (Ismail et al. 2009). Flooding negatively affects germination and survival in most rice genotypes (Ismail et al. 2012). Conversely, it suppresses the growth of weeds and shrinks the cost of physical weeding and/or herbicide application. It is projected that approximately 30% of the total cultivation cost is saved when weed emergence is suppressed by flood.
The ability of seeds to sprout, grow, and survive under low oxygen (hypoxia) or very little to no oxygen (anoxia) is known as AG. This trait is a must-have for all direct-seeded rice systems. The trait ensures risk mitigation at the early stages of the crop and can be used as a weed control mechanism. The majority of the modern high-yielding rice varieties do not show any germination underwater. However, landraces that can maintain germination under flooded conditions have evolved in various parts of rice-growing areas. Some examples of such tolerant landraces are Khao Hlan On, Ma Zhan (red), Kalarata, Nanhi, and Khaiyan. Results of donor identification studies have shown tolerant landraces spread across all major groups of rice. However, indica and aus landraces have been used in genetic studies and breeding programs more than the other groups so far. Khao Hlan On and Ma Zhan (red) are currently used extensively as donors of the trait in breeding and marker-assisted selection (MAS) programs at the International Rice Research Institute (IRRI).
4.1.1 Physiology and Molecular Mechanisms of AG
Rice is the only cereal that can withstand water submergence; hence, it grows even under hypoxic conditions. In extreme conditions, submerged plants can experience anoxia when subjected to prolonged flooding. Under anoxia, the plant shifts to an alcoholic fermentation (AF) pathway rather than respiration for energy, in the form of adenosine triphosphate (ATP). During AF, only two ATPs are being produced vis-à-vis 38 ATPs are produced in aerobic respiration (Magneschi and Perata 2009). Therefore, the plant is 19 times less efficient in ATP production when exposed to anaerobic conditions. Notably, three common physiological responses allow rice seedlings to survive under anaerobic conditions: (1) longer coleoptile, (2) greater water imbibition, and (3) higher starch reserves.
According to Pradet and Bomsel, in 1978 (as cited by Kennedy et al. 1980), the only plant organ that can grow under anoxia is the rice coleoptile. Through coleoptile elongation, the plant can gain access to aeration, which enables the germination of the developing embryo (Ismail et al. 2012). Because of this, AG tolerance can be indirectly measured using coleoptile length. Hsu and Tung (2015) referred to this trait as the “anaerobic response index.” Alpi and Beevers (1983) demonstrated that seedlings develop coleoptiles at a higher speed when subjected to low environmental O2 concentration. However, they further observed that, despite its greater length, the coleoptile is thin and fragile with less fresh weight. This phenomenon can be referred to as the snorkel effect, whereby anoxia induces the development of a hollow coleoptile to access a better-aerated environment (Kordan 1974). This provides oxygen supply for the plant’s root and endosperm, which supports complete and vigorous seedling establishment. Adachi et al. (2015) evaluated the germination of tolerant (IR06F459) and intolerant (IR42) rice lines and revealed that the coleoptiles of IR06F459 had significantly longer coleoptiles than those of IR42. IR06F459 is an AG-tolerant line developed from backcrossing IR64 to Khao Hlan On. In addition, Ismail et al. (2009) revealed that water stress during germination decreased shoot and root length of intolerant genotypes by 81% and 68%, respectively (Fig. 4). This was incomparable to the decrease of 61% in shoot length and 7% in root length of the tolerant genotypes.
Mean shoot and root lengths of five flooding-tolerant and five flooding-sensitive genotypes of rice measured 21 days after sowing under control and flooded (10 cm) conditions. Values in parentheses are percentage decreases in length relative to the length in the control. Vertical bars indicate l.s.d. at p = 0.05. (Source: Ismail et al. 2009)
During flooding, seeds experience oxygen-limiting conditions. Under these conditions, the plant requires above-normal tolerance of anoxia to exhibit strong seed germination and growth (Ismail et al. 2009). In 1996, Yamauchi revealed that the use of genotypes that can withstand anaerobic conditions could help improve seedling establishment, weed control, and lodging resistance in DSR. Hence, the utility of genotypes with high germination rate (i.e., coleoptile and mesocotyl elongation) and seedling vigor under anaerobic conditions can curtail problems in crop establishment and even weed competition (Azhiri-Sigari et al. 2005). As revealed by Ismail et al. (2009) in their evaluation of tolerant (Cody, Khaiyan, Nanhi, and Khan Hlan On) and intolerant (FR13A, IR22, IR28, IR42, and IR64) genotypes, the survival of both groups decreased but there was a markedly larger decrease in the survival of the intolerant genotypes (Fig. 5). Among these tolerant genotypes, Khaiyan and Khao Hlan On displayed the highest survival rates, whereas the intolerant genotypes recorded a survival percentage of only about 5%.
The rapid germination of AG lines was credited by El-Hendawy et al. (2011) to rapid water imbibition under submerged conditions. They evaluated 58 contrasting genotypes, which showed a strong negative correlation (r = −0.46, p < 0.001) between water uptake and germination time. Further, the authors performed cluster analysis of the lines based on water content over different times of seed imbibition and revealed that tolerant genotypes clustered together within the group, which was characterized by rapid water uptake. Together, this suggests that high water uptake can induce rapid seed germination under anoxia. A proposed mechanism of action is that water uptake promotes sugar mobilization from the endosperm to the developing embryo (El-Hendawy et al. 2011), which provides energy for plant growth.
Apart from efficient water imbibition, carbohydrate content has the potential to enable plant growth under water stress. Ella and Setter (1999) suggested that the adverse effect of decreased ATP supply during AF is relieved when more carbohydrate is available for breakdown. As such, seeds with high starch content are deemed more tolerant of AG than seeds with high fat content (Magneschi and Perata 2009). Under anoxia, plant survival was found to be strongly correlated to starch content (r = 0.73–0.88) (Ella and Setter 1999). However, starch cannot be used as it is; hence, some also hypothesize the role of α-amylases, which are responsible for the breakdown of starch into soluble sugar (El-Hendawy et al. 2011). Rice, among the cereal crops under anoxia, is the only crop that expresses α-amylase mRNA (Perata et al. 1992, 1993). Also, a rice seed grown under anoxia expresses the whole set of enzymes needed for starch breakdown into its soluble forms (Ismail et al. 2009). These amylases regulate starch degradation to glucose, which is needed in the plants’ fermentative metabolism that is activated for ATP production under anoxia (Septiningsih et al. 2013). Adachi et al. (2015) reported in their study that the α-amylase activity in their tolerant line (IR06F459) was significantly higher than in the intolerant line (IR42). This was also in agreement with the findings of Illangakoon et al. (2016), for which the survival rates of their genotypes were positively correlated with α-amylase activity (r = 0.79) and soluble sugar content (r = 0.74). Ismail et al. (2009) reported that the soluble sugar concentration remained low for both tolerant and intolerant genotypes until the tolerant genotypes had higher soluble sugar at 4–8 days after sowing. These findings suggest that tolerant genotypes can break down carbohydrate reserves in the seed and mobilize the resulting monosaccharide, which can enable germination and growth under hypoxia. In 2015, Kretzschmar et al. found that OsTPP7 was expressed in the germinating tissues of a NIL-AG1 (tolerant) line while its expression was absent in IR64 (susceptible). Given that OsTPP genes (i.e., OsTPP1 and OsTPP2) in rice convert trehalose-6-phosphate (T6P) to trehalose, this mechanism can also be true for OsTPP7. Further analysis revealed that trehalose and sucrose were 2.3-fold and 2.0-fold higher in NIL-AG1 than in IR64. This suggests that OsTPP7 affects the conversion of T6P to trehalose, which signals low sugar availability; consequently, the plant enables starch mobilization from the endosperm reserve (source) to the coleoptile (sink).
Expression analysis, through RNA sequence (RNA seq), revealed that RAmy3D was highly expressed in tolerant genotypes under anoxia (Ismail et al. 2009). RAmy3D is a member of the Amy3 gene subfamily, which was found to be upregulated in rice embryos under anoxia (Hwang et al. 1999). RAmy3D, unlike RAmy1A from the Amy1 gene subfamily, lacks the gibberellic acid (GA)-responsive element in its promoter region (Lu et al. 1998) and is important in oligosaccharide degradation (Terashima et al. 1997). Under starvation, a glucose and sucrose receptor, RAmy3D, is activated by protein kinase SnRK1A (Lu et al. 2007). Meanwhile, SnRK1A is stimulated by calcineurin B-like protein kinase (CIPK15) under oxygen-limiting conditions (Kudahettige et al. 2010). Additionally, the interaction of calcineurin B-like proteins, such as CBL4 and CBL5, with CIPK15 under anoxia is viewed as a regulating mechanism in plant response during anoxia (Ho et al. 2017; Sadiq et al. 2011).
Although many other rice enzymes are downregulated during anoxia to inactivate energy-conserving steps, pyruvate decarboxylase (PDC), alcohol dehydrogenase (ADH), and aldehyde dehydrogenase (ALDH) are active in genotypes tolerant of anoxia (i.e., Khaiyan), as reported by Ismail et al. (2009). The upregulation of these enzymes proposes enhanced alcohol fermentation during anoxia, which uses nicotinamide adenine dinucleotide (NAD) for glycolysis and substrate-level phosphorylation (Saika et al. 2006; Shingaki-Wells et al. 2011). As a result, enough energy and carbon are supplied to the developing coleoptile under anoxia. Moreover, rice coleoptiles under anoxic conditions have been reported to exhibit a decrease in pH, which also suggests alcoholic fermentation pathway activation. In 2009, Ismail et al. showed that the concentration of ethylene increased significantly after only 3 days of imbibition. They also reported that the ethylene produced affects starch hydrolysis through the reduction of abscisic acid (ABA) synthesis while upregulating the synthesis of and sensitivity to gibberellic acid (GA) of the internode tissue. This enhances starch catabolite enzyme activity under stress, which enables seedling growth and survival under AG. Further, ethylene promotes coleoptile growth through cell expansion by regulating enzymes such as peroxidases, which are responsible for cell wall rigidity. Peroxidase activity decreases the plant cell wall’s lignin content and protein assembly (Waffenschmidt et al. 1993), which in turn decreases its rigidity and permits cellular elongation. Figure 6 shows the proposed mechanism of coleoptile elongation under anoxia as interpreted by Ismail et al. (2009).
Proposed mechanisms involved in enhancing germination and seedling growth under low-oxygen stress in rice. (Source: Ismail et al. 2009)
Other factors that are deemed responsible for cell expansion are a group of nonenzymatic proteins called expansins, which can affect cell wall loosening. These proteins attach at the cell wall interface between cellulose microfibrils and matrix polysaccharides, which results in a disturbance of non-covalent bonds, enabling cell wall loosening (McQueen-Mason and Cosgrove 1995). Thus far, EXPA2, EXPA4, EXPA7, EXPB12, EXPA1, EXPB11, and EXPB17 were reported to be induced during submergence stress (Juang et al. 2000; Lasanthi-Kudahettige et al. 2007; Takahashi et al. 2011).
4.1.2 Genetic Factors Underlying the AG Trait
Before evaluating the genetics behind AG, it was necessary to identify lines that can germinate under very limited oxygen supply. The largest AG screening conducted has been that of Angaji et al. in 2010. They screened 8000 rice accessions from the IRRI Genetic Resources Center, which contains collections from gene bank accessions, elite breeding lines, IR64 mutants, and introgression lines. However, only 0.23% (19 lines) showed greater than or equal to 70% survival, a subset of which is present in Table 1. Nevertheless, Hsu and Tung (2017) evaluated gene expression in six genotypes and discovered 3597 genes affected by water stress irrespective of the plant’s genetic background, 5100 genes differentially affected across genotypes, and 471 genes affected in a genotype-dependent manner. Hence, the different genotypes are expected to display different degrees of tolerant response to anoxia.
Both quantitative trait loci (QTL) mapping and genome-wide association studies (GWAS) are useful in identifying genomic regions underlying different phenotypic traits. QTL mapping uses markers for a trait of interest, which can categorize the sample population into genotypic groups (Sehgal et al. 2016), while GWAS link common single nucleotide polymorphisms (SNPs) to phenotypic traits, which can provide information on the association between particular genetic variants and phenotype (Pal et al. 2015). Table 2 summarizes the results of some of the previous QTL mapping studies for AG tolerance using different rice landraces as sources.
In 2004, Jiang et al. evaluated 81 recombinant inbred lines (RILs) from a japonica/indica cross, Kinmaze/DV85. Kinmaze germinates well under anoxia; hence, it was used as the donor parent. These RILs were screened using 137 restriction fragment length polymorphism (RFLP) markers and five QTLs were detected on chromosomes 1, 2, 5, and 7. These QTLs explained phenotypic variation ranging from 10.5% to 19.6% of the shoot lengths. According to the calculated additive effects, the positive effect of three QTLs (qAG-1, qAG-2, and qAG-7) was contributed by Kinmaze. Meanwhile, AG tolerance conferred by qAG-5a and qAG-5b was improved due to the recurrent parent, DV85.
Another set of rice lines was evaluated by Jiang et al. in 2006 using USSR5 and N22 as parents. USSR5 is a japonica subspecies from the former Soviet Union while N22 is an indica type. These were crossed and the generated F2 plants were evaluated for survival under AG and then genotyped using 121 simple sequence repeat (SSR) markers. Mapping revealed two QTLs on chromosomes 5 (qAG-5) and 11 (qAG-11) contributing to AG tolerance. Both QTLs were contributed by the USSR5 allele, which accounted for 11.0–15.5% of the coleoptile length variation.
Four QTLs were identified by Angaji in 2008 from a cross using Khaiyan, an aus-type tolerant variety from Bangladesh, as a donor. Khaiyan was crossed with semi-dwarf nonaromatic indica lowland modern rice variety IR64, and the F1s were backcrossed to IR64 to produce 150 BC2F2 lines. The identified QTLs are qAG-1, qAG-2-2, qAG-11, and qAG-12, which explained 12.0–29.2% of the variation in survival of the lines, while together these QTLs accounted for a total of 51.4% of the phenotypic variation. However, based on the additive values, only the QTL qAG-1 had the allele from Khaiyan, which increased AG tolerance.
Another promising donor for AG tolerance is Khao Hlan On, which is a tall aromatic japonica upland landrace from Myanmar used by Angaji et al. in their study in 2010. They crossed Khao Hlan On with IR64 and the F1 progenies were backcrossed and selfed to produce 423 BC2F2 lines. QTL mapping was done using 135 SSRs and one InDel marker. Five QTLs accounting for 17.9–33.5% of the variation in percentage survival were detected from chromosomes 1 (qAG-1-2), 3 (qAG-3-1), 7 (qAG-7-2), and 9 (qAG-9-1 and qAG-9-2). Further, from the values of the additive effects of the QTLs, it was inferred that AG tolerance was contributed by alleles from Khao Hlan On.
More recently, in 2013, Septiningsih et al. used 118 SSR markers to assess AG tolerance alleles from Ma-Zhan (red), a highly AG-tolerant landrace from China with 90% survival (Angaji et al. 2010). Ma Zhan (red) was crossed with IR42 and then backcrossed to produce BC2F3 families. The survival rates and genotype data of 118 SSR markers in 175 families revealed six QTLs related to AG on chromosomes 2, 5, 6, and 7, accounting for 5.6–30.3% of the phenotypic variation. These QTLs are qAG-2, qAG-5, qAG-6, qAG-7-1, qAG-7-2, and qAG-7-3. It must also be noted that all five of these QTLs were donated by Ma Zhan (red). QTL qAG-7-2 explained the highest amount of phenotypic variation, amounting to 30.3% in interval mapping (IM) and 31.7% in composite interval mapping (CIM), with LOD scores of 13.7 and 14.5, respectively. This QTL was further evaluated to confirm its presence and effect. The authors developed introgression lines bearing the homozygous alleles of qAG-7-2 from Ma Zhan (red) and revealed that these introgression lines have significantly higher survival (47.58 ± 4.72%) than the introgression lines bearing homozygous IR42 alleles (16.67 ± 2.99%). Moreover, the results confirmed the effect of the QTL, which rendered an additional 30% survival in line with the homozygous allele of Ma Zhan (red). Given that Ma Zhan (red) still had the highest survival rate (73.7 ± 14.19%) among all the lines analyzed, this suggests the effect of other QTLs distributed widely in the rice genome to improve AG tolerance.
Hsu and Tung (2015) used both GWAS and QTL mapping to detect genomic regions related to AG tolerance. A total of 144 RILs from a Nipponbare and IR64 cross were used in QTL mapping with 35,501 SNPs. The authors identified a peak on chromosome 1 using both single marker analysis (SMA) and interval mapping based on the linkage map of 355 SNPs filtered after Bonferonni correction with α = 0.05. For GWAS, 153 accessions and 36,901 SNPs were used. This revealed three genomic regions on chromosome 7 (25.12, 26.92, and 27.72 Mb); however, the major QTL on chromosome 1, which was detected in QTL mapping, was not identified using GWAS. Since this QTL on chromosome 1 cannot be tagged as a rare variant based on haplotype analysis, it was proposed that the haplotype and varietal relationships can account for the inconsistency in findings. As mentioned by Kang et al. (2008), genetic association studies are confounded by population structure and relatedness, which can produce several false positives. However, Brachi et al. (2011) noted that overcorrection for these factors can lead to false-negative results.
Remarkably, some of the identified QTLs overlap across different studies. Some of these are (1) qAG-2 detected by Jiang et al. (2004) and Septiningsih et al. (2013), (2) qAG-7-2 by Angaji et al. (2010) and qAG-7-3 from Septiningsih et al. (2013), and (3) qAG-1-2 from Angaji et al. (2010) and chromosome 1 QTL from Hsu and Tung (2015). Further assessment should be done to detect whether these QTLs are controlled by the same gene(s).
At present, two QTLs have been considered of prime importance for AG tolerance, AG1 (qAG-9-2) and AG2 (qAG-7-1) identified from Khao Hlan On and Ma Zhan (red), respectively (Miro and Ismail 2013). In 2015, Kretzschmar et al. fine-mapped QTL qAG-9-2 to a 50-kb region on chromosome 9. Based on Nipponbare as the reference genome, this region encompasses four genes and one transposable element. Contrasting this region to a de novo assembled IR64 sequence revealed a 20.9-kb deletion of AG1 spanning across LOC_Os09g20390/Os09g0369400 (OsTPP7) and a deletion of neighboring loci. Meanwhile, the QTL did not show any structural variation against Nipponbare apart from a 4-kb deletion that is also present in IR64.
4.2 Seed Longevity
The life cycle of seeds involves fertilization, dormancy, and germination. Bewley (1997) defined seed dormancy as “the failure of an intact viable seed to completely germinate under favorable conditions.” Associated with dormancy is seed longevity, which is the ability of the seed to remain viable after harvesting, and this is usually evaluated through seed germination ratio or percentage.
Seed longevity varies among different rice varieties as it is governed by both genetic and nongenetic factors. Some notable nongenetic factors are ambient environmental conditions during seed development, maturity at seed harvest, mechanical damage, presence of pathogens, moisture content, and storage conditions after harvest (Copeland and McDonald 1995). Seed longevity and viability usually decrease when exposed to high temperature and moisture (Roberts 1961; McDonald 1999). Yamauchi and Winn (1996) reported that low seed longevity or seed aging is a major issue in the establishment of seeds in anaerobic environments where direct seeding is practiced. If seed longevity fails, crop establishment decreases and weed pressure proliferates. Both scenarios can dramatically decrease crop yield; hence, the importance of varieties tolerant of seed aging.
4.2.1 Physiology and Molecular Mechanisms Affecting Seed Longevity
Seeds battle aging through protection and repair. The seed protection mechanism involves the development of a glassy cytoplasm, which limits cellular metabolic activities and antioxidant production during seed storage to inhibit the buildup of oxidized macromolecules that can diminish seed viability. Conversely, the seed repair system exploits DNA glycosylases and methionine sulfoxide reductases, which prevent damage accumulation in the DNA, RNA, and/or proteins during seed imbibition (Sano et al. 2016).
Seed aging is oftentimes linked with the oxidation of nucleic acids, proteins, and lipids (Bailly 2004), causing oxidative stress. This can be combated through passive mechanisms, such as nonenzymatic reactive oxygen species (ROS)-scavenging systems, and active mechanisms, such as enzymatic ROS detoxification (Sano et al. 2016). In enzymatic responses, seeds rely on antioxidant enzymes such as superoxide dismutase, catalase, glutathione, ascorbate peroxidase, monodehydroascorbate, dehydroascorbate, and glutathione reductase (Bailly 2004; Kumar et al. 2015). Petla et al. (2016) also reported the role of the repair enzyme l-isoaspartyl O-methyltransferase, overexpressed in transgenic rice, by restricting ROS accumulation and by repairing homeostasis-disruptive isopartlyl residues in proteins.
Among the nonenzymatic responses, the most important antioxidants are tocopherols or vitamin E, which prevents nonenzymatic lipid peroxidation. Vitamin E-mediated free radical scavenging in rice decreases lipid membrane peroxidation, which results in greater seed longevity (Hameed et al. 2014).
Goufo and Trindade (2013) enumerated other biochemical factors affecting the longevity of rice seeds that include phenolic acid, flavonoids, coumarins, tannins, and anthocyanins. For example, antioxidant polyphenols such as flavonoids in the seed coat, embryo, and endosperm of Arabidopsis have been shown to increase seed longevity (Debeaujon et al. 2000). It is worth noting that AG donor lines such as Khao Hlan On have red pericarps, which may have putative roles in both seed longevity and AG tolerance.
Interest has increased in the epigenetic basis of the seed life cycle from fertilization to dormancy until germination. van Zanten et al. (2013) reported that seeds require epigenetic factors such as chromatin condensation to enable the transition from seed dormancy to germination in Arabidopsis thaliana . These epigenetic modifications include condensation and methylation (Exner and Hennig 2008; Xiao et al. 2006), which affect chromatin accessibility under stress. Xiao et al. (2006) stated that mutated genes for DNA methylation result in nonviable seedlings and improperly developed embryo in A. thaliana. Interestingly, using a drought-tolerant barley variety, it was found that HvDME gene was highly expressed after 10 days of drought stress (Kapazoglou et al. 2013). This gene codes for DNA glycolases, which are responsible for DNA methylation affecting chromatin accessibility.
Recent work on the environmental effect on seed longevity likewise showed the influence of the pretreatments of seed drying at 45–60 °C (Whitehouse et al. 2017). It was revealed that this improves seed longevity in rice germplasm prior to storage. Furthermore, environmental transgenerational changes such as warmer parental growth environment affect the subsequent generation’s seed longevity in alpine trees (Silene vulgaris ) by providing transcripts of SvHSP17.4 and SvNRPD12 mRNAs to withstand heat stress (Mondoni et al. 2014). Moreover, the maternal environment of rice might have a putative role in seed longevity as was shown in barley (Nagel et al. 2015).
4.2.2 Genetic Factors Affecting Seed Longevity
Genome-wide association mapping in barley (Hordeum vulgare) showed that seed longevity is inherently affected by genetics, maternal environment, and seed deterioration while negating the effects of lipid-soluble tocochromanols, oil, and proteins (Nagel et al. 2015). Studies of different rice genotypes have shown that indica-type varieties have higher seed longevity than japonica-type (Chang 1991; Ellis et al. 1992). Table 3 summarizes some of the QTLs identified from different rice populations related to seed longevity.
In rice, three QTLs for seed longevity (qLG-2, qLG-4, and qLG-9) were detected on chromosomes 2, 4, and 9, respectively, using backcross inbred lines from a Nipponbare/Kasalath cross (Miura et al. 2002). QTL qLG-9 accounted for 59.5% of the phenotypic variation, which has a larger effect than the phenotypic variation effect of qLG-2 with 13.4% and qLG-4 with 11.6%. Further investigation by Sasaki et al. (2005) detected 12 germination QTLs as indices of seed longevity. In the latter study, qLG-9 was also detected and identified as RC9-2, which affected phenotypic variation by 12.5–12.8%.
Sasaki et al. (2015) fine-mapped the putative gene located in qLG-9 using advanced backcross progenies from the cross between japonica-type Nipponbare and indica-type Kasalath. They found that the candidate genes in the region encode for trehalose-6-phosphatase (Os09g039369400), TPP, and an unknown protein (Os09g039369500). Notably, Kretzschmar et al. (2015) mapped a candidate gene in the same region, which enhances anaerobic germination. This further affirms that the gene for seed longevity can be used for marker-assisted selection (MAS) for the breeding of aging tolerant rice for direct-seeded conditions to enable seedling germination and establishment even in anaerobic conditions.
4.3 Early Seedling Vigor
Seedling vigor reflects the plant’s potential to quickly emerge from the soil or water, which is a relevant trait for crop establishment, especially for direct-seeded rice. It is associated with germination and seedling growth, which enable good crop establishment and weed competition (Diwan et al. 2013). It is a complex trait that is governed by several genetic factors that affect the physiological, morphological, and biochemical processes of the plant.
4.3.1 Physiology and Molecular Mechanisms of Early Seedling Vigor
Some of the physiological traits that affect early seedling vigor are shoot length, shoot fresh weight, number of tillers per plant, mesocotyl length, root fresh and dry weight, germination rate, germination index, amylase activity, reducing sugar content, root activity, and chlorophyll content. Teng et al. (1992) evaluated the seedling growth of 38 rice varieties with differences in grain length, width, and weight. They revealed that the most vigorous seedlings were those with larger embryos and heavier endosperms.
Within the endosperm, the stored starch granules are composed of both amylose and amylopectin. These starch granules were distinguished based on their glucose residues (Huang et al. 2017). Amylose is made up of α-1,4-linked glucose residues; hence, it can be broken down into maltose by amylases. Amylopectin, however, cannot be split due to the presence of both α-1,4 and α-1,6 linkages. Starch hydrolysis is an important step for ATP production and for the generation of carbon skeletons for the development of new cellular components (Mitsui et al. 1996). Prior to photoautotrophy, seedlings gain the energy required for plant development from the breakdown of starch granules (i.e., amylose) in the endosperm (Cui et al. 2002b). This is regulated by starch-hydrolyzing enzymes such as α-amylases, which facilitate the cleavage of α-1,4-glucan bonds of starch (Hakata et al. 2012). Notably, this is considered as the first committed step in starch degradation.
α-Amylases are known to be initially expressed in the scutellar epithelial tissue and in the aleurone layer; they are then subsequently secreted into the starchy endosperm (Mitsui et al. 1996) to initiate starch breakdown. Therefore, at the seedling stage, plant vigor can be accounted for by higher amylose content and amylase activity, which provide the growing embryo with enough energy for development. Huang et al. (2017) used two cultivars, Yuxiangyouzhan and Huanghuazhan, to assess the morphophysiological traits governing seedling vigor. Yuxiangyouzhan (96%) showed a higher germination percentage than Huanghuazhan (87%). It was further revealed that Yuxiangyouzhan had higher seed amylose content and amylase activity. Additionally, Huang et al. (2017) cited the utility of using cultivars with thinner hulls, hence requiring less mechanical strength for coleoptile emergence.
4.3.2 Genetic Factors Affecting Early Seedling Vigor
Diwan et al. (2013) highlighted the importance of producing genotypes with early seedling vigor due to economic and environmental concerns. To address this, it is important to determine the genetics underlying seedling vigor. Several studies have been conducted to identify QTLs related to early seedling vigor. Table 4 summarizes some of the major QTLs related to early vigor that have been identified from different studies. Anumalla et al. (2015) surmised that RM259 and RM84 of chromosome 1; RM282, RM148, and RM85 of chromosome 3; RM26 of chromosome 5; and RM11 of chromosome 7 were the most promising markers for several seedling vigor traits. Nagavarapu et al. (2017) validated eight of the previously identified QTLs from different studies: qGR-1, qGP, and qGI-11 (Wang et al. 2010); RZ448 and RZ395 (Redoña and Mackill 1996); qFV-3-2, qFV-5-2, and qFV-10 (Zhou et al. 2007). The authors used 47 genotypes suitable for diverse ecosystems and estimated seedling vigor traits, followed by QTL analysis. Among the eight QTLs used for validation, six (qGR-1, qFV-10, qFV-3-2, qFV-5-2, qGP-6, and RZ395) were found to be associated with some of the selected genotypes. QTL qGR-1 for germination rate was detected in 12 genotypes; qFV-10 for seedling height under flooding in eight genotypes; qFV-3-2 for coleoptile emergence under flooding, qFV-5-2 for seedling height upon water draining, and RZ395 for mesocotyl elongation in three genotypes; and qGP-6 for germination percentage in two genotypes. Notably, some of the genotypes also showed multiple QTLs such as Dinesh with four and Pooja Sabita and Vivekadhan 62 each with two. Furthermore, Abe et al. (2012) identified OsGA20ox1 as a candidate gene for seedling vigor in rice. Using RILs from a cross between Kekashi and Dinghan Shali, the authors identified a putative QTL on chromosome 3, qPHS3-2, which affects 26.2% of the phenotypic variation in seedling height. This QTL region was then introduced to NILs with a background of an elite cultivar, Iwatekko. The region was fine-mapped into an 81-kb interval bearing the OsGA20ox1 gene that is related to gibberellin biosynthesis. Their further analysis revealed that both Dunghan Shali and the NILs had higher expression of OsGA20ox1 relative to Iwatekko, which suggests the utility of this gene for seedling vigor.
4.4 Breeding Rice with Improved Germination
Genetic studies on traits such as AG, seed longevity, and early seedling vigor have shown high genetic variation between modern rice varieties, breeding lines, and traditionally evolved cultivars. Systematic extraction of genes that confer stronger germination from traditional varieties is required to develop high-yielding varieties with good crop stand and improved weed competitiveness. With DSR covering a larger proportion of rice area and quickly becoming an ask of the future, it is clear that rice varieties have to evolve from their current state and reach much higher tolerance of adverse conditions during seed germination. Although a large number of DSR breeding programs target selection for early vigor, a thorough understanding of this trait is most important to make genetic gains. For example, it is believed that increased seed size allows better germination capacity. However, in the case of rice, grain shape is an important seed quality and has to be kept in line with the product requirements of specific environments. This limits the amount of improvement for the trait as the best lines have to be selected within grain shape classes. This may also lead to higher popularity of varieties with larger grains under DSR. Similarly, grain pericarp color has been associated with higher AG tolerance and seed longevity and this needs to be addressed through systematic genetic studies to be able to develop white rice with better germination and seed longevity. Trait development and breeding methods must be combined systematically to be able to leverage the advantage of alleles with major and minor effects.
Figure 7 shows a set of pipelines that allow a systematic flow of required traits into a breeding program. Since most modern rice varieties are deficient in these traits, the work begins in the trait development pipeline with the development of a phenotyping protocol and donor identification. Once this is complete, adequate mapping populations are to be developed that can support the identification of robust QTLs. It is also to be kept in mind during this process that the recipient be chosen correctly. Ideally, these recipients should have elite genetic backgrounds that can be used in a breeding program in the future or those DSR-adapted breeding lines or varieties that are deficient in these traits. Upon the identification of the QTLs, screening a part of the mapping population in a few key locations can test their effect in the target environments. This allows developing confidence in the effect of the QTL and investment in further fine-mapping can be carried out. Fine-mapped QTL regions and associated markers are delivered to the trait deployment pipeline, which rapidly develops high-yielding NILs of a wide range of recipient parents. Ideally, these should be the latest elite breeding lines from the variety development pipeline. These NILs are entered into the pipeline and checked for yield potential and trait performance across key testing sites. High-performing NILs may be advanced for release as well as recycled into the crossing process to develop the next generation of breeding lines. This cyclic process allows the rapid recycling of high-performing lines as parents and allows building on the achieved genetic gain to advance it further. Once released, the varieties developed through this pipeline are managed through a seed systems team for adequate placement and deployment in collaboration with national seed systems. This team also collects feedback and provides it to the above pipeline so that any possible problems can be addressed in the next wave of varieties. The improvement of breeding lines for seed germination-related traits is important for both inbred programs and hybrid breeding programs. Hybrids provide great potential for achieving high yields in direct-seeded environments. However, for hybrids to be successful in DSR, germination and seedling survival must be high, and good crop stand be achieved with low seed rates to keep seed costs down.
All three major groups of traits discussed previously should be targeted to be able to improve germination under direct-seeded conditions. Although anaerobic germination is one of the most important traits, it cannot provide the needed robustness without other traits. Early seedling vigor helps to boost the effect of anaerobic germination and leads to better tolerance. Vigorous and rapidly growing coleoptiles can emerge from deeper soil layers efficiently and allow more uniform germination. This is specifically helpful in cases where seeding is mechanized and field preparations are not adequate. Lastly, higher seed longevity helps in maintaining germination for relatively older seeds. In cases where farmers use their own seeds, the age of seeds normally varies from 6 to 8 months. It is observed that although this duration does not affect germination to a large extent, it may have an effect on early vigor and anaerobic germination. In some cases where seeds are not adequately stored, germination may also become affected. It is observed, however, that genetic variation exists for the trait and it can be harnessed to improve modern varieties similar to the previous group of traits. In general, multiple donor lines for the three groups of traits can be crossed to two to three elite recipient lines, each belonging to a different grain shape group. This will allow the development of nested association mapping populations with a common recipient parent within the grain shape group. Trait development activities can then be undertaken to identify and fine-map robust QTLs for each of the grain shape groups. Finally, the identified QTLs can be introgressed singly or pyramided into the recipient lines and can be further deployed in the breeding program as donor lines.
5 Conclusions
Globally, DSR systems are of high importance to sustain profitable rice production under the current agricultural scenario. Declining water and labor resources are driving rice away from the traditional transplanted system. DSR systems are profitable and more amenable to mechanization and are able to decrease water and labor requirements considerably vis-à-vis transplanted systems. However, rice varieties require a specific set of traits at various growth stages to be able to accumulate biomass and produce yield. In the absence of such varieties, the full potential of direct-seeded rice systems cannot be exploited. Further, the use of unsuitable rice varieties makes these systems risky and vulnerable to crop failure. These result in a low adoption of the technology despite its obvious benefits. Rice varieties containing DSR adaptation traits are thus critical to success in these systems.
An important phase in DSR systems is crop establishment. Although the use of high-yielding varieties is important, this will be futile if crop establishment fails. This becomes even more important in the case of hybrid rice, for which adoption depends on the minimization of seed rate and good crop establishment. Anaerobic germination, seed longevity, and early seedling vigor are some traits relevant to crop establishment under direct seeding. Anaerobic germination enables the embryo to sprout and the seedling to survive during anoxia, which can occur upon seeding when the field is intentionally or accidentally irrigated with water. Seed longevity renders aging tolerance to seeds, such that the seeds remain viable under temperature or moisture changes for a longer period. Lastly, early seedling vigor enables the growing seedlings to quickly emerge from the soil and/or water and gives them strong early crop establishment and weed competition.
The physiology and genetics of the aforementioned traits have been studied in the past; however, a lot remains to be unraveled to fully exploit these traits. Further, there remains a need to use these traits in breeding programs to develop DSR-specific varieties. This requires well-designed trait development, pre-breeding, and breeding pipelines with set criteria to advance only the most robust of products. The success of rice in DSR systems largely depends on the robustness of rice varieties at early stages to avoid the risk of crop failure and weed competition. Developing rice varieties with stronger germination capacity under varying conditions is one of the most important factors that will determine success in this area.
Change history
11 July 2021
“Owing to an error on the part of the editor and corresponding chapter author, the author sequence in the chapter opening page of chapter Crop Establishment in Direct-Seeded Rice: Traits, Physiology, and Genetics was presented wrongly. The author sequence has now been updated to be Fergie Ann Quilloy, Benedick Labaco, Carlos Casal Jr., Shalabh Dixit in the chapter opening page, table of contents, and wherever applicable throughout the book.”
References
Abe A, Takagi H, Fujibe T, Aya K, Kojima M, Sakakibara H et al (2012) OsGA20ox1, a candidate gene for a major QTL controlling seedling vigor in rice. Theor Appl Genet 125:647–657
Adachi Y, Sugiyama M, Sakagami JI, Fukuda A, Ohe M, Watanabe H (2015) Seed germination and coleoptile growth of new rice lines adapted to hypoxic conditions. Plant Prod Sci 18:471–475
Aggarwal PK (2008) Global climate change and Indian agriculture: impacts, adaptation and mitigation. Indian J Agric Sci 78(11):911
Aggarwal PK, Kumar SN, Pathak H (2010) Impacts of climate change on growth and yield of rice and wheat in the Upper Ganga Basin. WWF report. WWF, New Delhi, pp 1–44
Akhgari H, Kaviani B (2011) Assessment of direct seeded and transplanting methods of rice cultivars in the northern part of Iran. Afr J Agric Res 6(31):6492–6498
Alpi A, Beevers H (1983) Effects of O2 concentration on rice seedlings. Plant Physiol 71(1):30–34
Angaji S (2008) Mapping QTLs for submergence tolerance during germination in rice. Afr J Biotechnol 7(15):2551–2558
Angaji S, Septiningsih EM, Mackill D, Ismail AM (2010) QTLs associated with tolerance of flooding during germination in rice (Oryza sativa L.). Euphytica 172:159–168
Anumalla M, Anandan A, Pradhan S (2015) Early seedling vigour, an imperative trait for direct-seeded rice: an overview on physio-morphological parameters and molecular markers. Planta 241(5):1027–1050
Azhiri-Sigari T, Gines H, Sebastian LS, Wade L (2005) Seedling vigor of rice cultivars in response to seedling depth and soil moisture. Philipp J Crop Sci 30:53–58
Bailly C (2004) Active oxygen species and antioxidants in seed biology. Seed Sci Res 14:93–107
Baltazar M, Ignacio J, Thompson M, Ismail A, Mendioro M, Septiningsih E (2014) QTL mapping for tolerance to anaerobic germination from IR64 and the aus landrace Nanhi using SNP genotyping. Euphytica 197(2):251–260
Barker T (2009) Climate change and EU labor markets. Employment in Europe 2009. EU, Brussels
Barker R, Dawe D, Tuong T, Bhuiyan S, Guerra L (1998) The outlook for water resources in the year 2020: challenges for research on water management in rice production. In: Assessment and Orientation Towards the 21st Century. Proceedings of 19th Session of the International Rice Commission, Cairo, Egypt, 7–9 September 1998. FAO, Rome, pp 96–109
Bewley JD (1997) Seed dormancy and germination. Plant Cell 9:1055–1066
Bouman B, Lampayan R, Tuong T (2007) Water management in irrigated rice: coping with water scarcity. International Rice Research Institute, Los Baños
Brachi B, Morris GP, Borevitz JO (2011) Genome-wide association studies in plants: the missing heritability is in the field. Genome Biol 12:232
Chang TT (1991) Findings from a 28-year seed viability experiment. Int Rice Res Newsl 16:5–6
Chavarria G, dos Santos HP (2012) Plant water relations: absorption, transport and control mechanisms. In: Monanaro G (ed) Advances in selected plant physiology aspects. InTech, Rijeka. ISBN: 978-953-51-0557-2, https://www.intechopen.com/books/advances-in-selected-plantphysiology-aspects/plant-water-relations-absorption-transport-and-control-mechanisms
Cheng X, Cheng J, Huang X, Lai Y, Wang L, Du W (2013) Dynamic quantitative trait loci analysis of seed reserve utilization during three germination stages in rice. PLoS One 8:e80002
Copeland LO, McDonald MB (1995) Seed science and technology, 3rd edn. Chapman and Hall, London, pp 181–220
Cordero-Lara KI, Kim H, Tai TH (2016) Identification of seedling vigor-associated quantitative trait loci in temperate japonica rice. Plant Breed Biotechnol 4:426–440
Cui K, Peng S, Xing Y, Xu C, Yu S, Zhang Q (2002a) Molecular dissection of seedling-vigor and associated physiological traits in rice. Theor Appl Genet 105:745–753
Cui K, Peng S, Xing Y, Yu S, Xu C (2002b) Molecular dissection of relationship between seedling characteristics and seed size in rice. Acta Bot Sin 44(6):702–707
Debeaujon I, Léon-Kloosterziel KM, Koornneef M (2000) Influence of the testa on seed dormancy, germination, and longevity in Arabidopsis. Plant Physiol 122(2):403–414
Diwan J, Channbyregowda M, Shenoy V, Salimath P, Bhat R (2013) Molecular mapping of early vigour related QTLs in rice. Res J Biol 1:24–30
Dixit S, Grondin A, Lee CR, Henry A, Olds TM, Kumar A (2015) Understanding rice adaptation to varying agro-ecosystems: trait interactions and quantitative trait loci. BMC Genet 16:86
Dixit S, Kumar A, Woldring H (2016) Water scarcity in rice cultivation: current scenario, possible solutions, and likely impact. In: Regional: development and dissemination of climate-resilient rice varieties for water-short areas of South Asia and Southeast Asia. International Rice Research Institute, Los Baños, pp 3–26
Dong XY, Fan SX, Liu J, Wang Q, Li MR, Jiang X, Liu ZY, Yin YC, Wang JY (2017) Identification of QTLs for seed storability in rice under natural aging conditions using two RILs with the same parent Shennong 265. J Integr Agric 16(5):1084–1092
El-Hendawy S, Sone C, Ito O, Sakagami J (2011) Evaluation of germination ability in rice seeds under anaerobic conditions by cluster analysis. Res J Seed Sci 4(2):82–93
Ella ES, Setter TL (1999) Importance of seed carbohydrates in rice seedling establishment under anoxia. Acta Hortic 504:209–216
Elliott J, Deryng D, Müller C, Frieler K, Konzmann M, Gerten D et al (2014) Constraints and potentials of future irrigation water availability on agricultural production under climate change. Proc Natl Acad Sci U S A 111(9):3239–3244
Ellis RH, Hong TD, Roberts EH (1992) The low-moisture-content limit to the negative logarithmic relation between seed longevity and moisture content in three subspecies of rice. Ann Bot 69(1):53–58
Exner V, Hennig L (2008) Chromatin rearrangements in development. Curr Opin Plant Biol 11(1):64–69
Fanish SA (2016) Enhancing resource use efficiency (RUE) under direct seeded rice (DSR) system: a review. Am Eurasian J Agric Environ Sci 16(9):1534–1544
FAO (2016) AQUASTAT main database. Food and Agriculture Organization of the United Nations (FAO), Rome. http://www.fao.org/nr/water/aquastat/data/query/index.html?lang=eng
Fukuda A, Kataoka K, Shiratsuchi H, Fukushima A, Yamaguchi H, Mochida H et al (2014) QTLs for seedling growth of direct seeded rice under submerged and low temperature conditions. Plant Prod Sci 17(1):41–46
Goufo P, Trindade H (2013) Rice antioxidants: phenolic acids, flavonoids, anthocyanins, proanthocyanidins, tocopherols, tocotrienols, c-oryzanol, and phytic acid. Food Sci Nutr 2:75–104
Grigg DE (1974) The agricultural systems of the world: an evolutionary approach. Cambridge University Press, Cambridge
Guo L, Zhu L, Xu Y, Zeng D, Wu P, Qian Q (2004) QTL analysis of seed dormancy in rice (Oryza sativa L.). Euphytica 140:155–162
Hakata M, Kuroda M, Miyashita T, Yamaguchi T, Kojima M, Sakakibara H et al (2012) Suppression of alpha-amylase genes improves quality of rice grain ripened under high temperature. Plant Biotechnol J 10(9):1110–1117
Hameed A, Rasheed A, Gul B, Khan MA (2014) Salinity inhibits seed germination of perennial halophytes Limonium stocksii and Suaeda fruticosa by reducing water uptake and ascorbate dependent antioxidant system. Environ Exp Bot 107:32–38
Han L, Qiao Y, Zhang S, Zhang Y, Cao G, Kim J, Lee K, Koh H (2007) Identification of quantitative trail loci for cold response of seedling vigor traits in rice. J Genet Genom 34(3):239–246
Ho V, Tran A, Cardarelli F, Perata P, Pucciariello C (2017) A calcineurin B-like protein participates in low oxygen signalling in rice. Funct Plant Biol 44(9):917–928
Hsu SK, Tung CW (2015) Genetic mapping of anaerobic germination-associated QTLs controlling coleoptile elongation in rice. Rice (N Y) 8(1):1–12
Hsu SK, Tung CW (2017) RNA-Seq analysis of diverse rice genotypes to identify the genes controlling coleoptile growth during submerged germination. Front Plant Sci 8:1–15
Huang M, Zhang R, Chen J, Cao F, Jiang L, Zou Y (2017) Morphological and physiological traits of seeds and seedlings in two rice cultivars with contrasting early vigor. Plant Prod Sci 20(1):95–101
Hwang YS, Thomas B, Rodriguez R (1999) Differential expression of rice α-amylase genes during seedling development under anoxia. Plant Mol Biol 40:911–920
Illangakoon T, Ella E, Ismail A, Marambe B, Keerthisena R, Bentota A et al (2016) Impact of variety and seed priming on anaerobic germination-tolerance of rice (Oryza sativa L.) varieties in Sri Lanka. Trop Agric Res 28(1):26–37
IPCC (2007) Climate change 2007: mitigation. In: Metz B, Davidson OR, Bosch PR, Dave R, Meyer LA (eds) Working Group III contribution to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change (IPCC). Technical summary. IPCC, Geneva. Chapters 3 (Issues related to mitigation in the long term context) and 11 (Mitigation from a cross sectoral perspective)
Ismail AM, Ella ES, Vergara GV, Mackill DJ (2009) Mechanisms associated with tolerance to flooding during germination and early seedling growth in rice (Oryza sativa). Ann Bot 103(2):197–209
Ismail AM, Johnson DE, Ella ES, Vergara GV, Baltazar AM (2012) Adaptation to flooding during emergence and seedling growth in rice and weeds, and implications for crop establishment. AoB Plants 2012:1–18
Jiang L, Hou MY, Wang CM, Wan JM (2004) Quantitative trail loci and epistatic analysis of seed anoxia germinability in rice (Oryza sativa). Rice Sci 11(5–6):238–244
Jiang L, Liu S, Hou M, Tang J, Chen L, Zhai H et al (2006) Analysis of QTLs for seed low temperature germinability and anoxia germinability in rice (Oryza sativa L.). Field Crop Res 98(1):68–75
Jiang W, Lee J, Jin YM, Qiao Y, Piao R, Jang SM, Woo MO, Kwon SW, Liu X, Pan HY, Du X, Koh HJ (2011) Identification of QTLs for seed germination capability after various storage periods using two RIL populations in rice. Mol Cell 31:385–392
Juang J, Takano T, Akita S (2000) Expression of α-expansin genes in young seedlings of rice (Oryza sativa L.). Planta 211(4):467–473
Kang HM, Zaitlen NA, Wade CM, Kirby A, Heckerman D, Daly MJ, Eskin E (2008) Efficient control of population structure in model organism association mapping. Genetics 178:1709–1723
Kapazoglou A, Drosou V, Argiriou A, Tsaftaris AS (2013) The study of a barley epigenetic regulator, HvDME, in seed development and under drought. BMC Plant Biol 13:172
Kaur J, Singh A (2017) Direct seeded rice: prospects, problems/constraints and researchable issues in India. Curr Agric Res J 5(1):13–32
Kennedy RA, Barrett SC, Vander Zee D, Rumpho ME (1980) Germination and seedling growth under anaerobic conditions in Echinochloa crus-galli (barnyard grass). Plant Cell Environ 3:243–248
Kordan HA (1974) Patterns of shoot and root growth in rice seedlings germinating under water. J Appl Ecol 1(2):685–690
Kreft S, Eckstein D, Melchior I (2016) Global climate risk index 2017: who suffers most from extreme weather events? Weather-related loss events in 2015 and 1996 to 2015. Germanwatch Nord-Süd Initiative eV, Bonn
Kretzschmar T, Pelayo MF, Trijatmiko KR, Gabunada LM, Alam R, Jimenez R et al (2015) A trehalose-6-phosphate phosphatase enhances anaerobic germination tolerance in rice. Nat Plants 1(9):1–5
Kudahettige N, Pucciariello C, Parlanti S, Alpi A, Perata P (2010) Regulatory interplay of the Sub1A and CIPK15 pathways in the regulation of α-amylase production in flooded rice plants. Plant Biol 13(4):611–619
Kumar V, Ladha J (2011) Direct seeding of rice: recent developments and future research needs. Adv Agron 111:297–413
Kumar SJ, Prasad SR, Banerjee R, Thammineni C (2015) Seed birth to death: dual functions of reactive oxygen species in seed physiology. Ann Bot 116:663–668
Lasanthi-Kudahettige R, Magneschi L, Loreti E, Gonzali S, Licausi F, Novi G et al (2007) Transcript profiling of the anoxic rice coleoptile. Plant Physiol 144(1):218–231
Lee HS, Sasaki K, Atsushi H, Ahn SN, Sato T (2012) Mapping and characterization of quantitative trait loci for mesocotyl elongation in rice (Oryza sativa L.). Rice 5(1):13
Li L, Lin Q, Liu S, Liu X, Wang W, Hang NT, Liu F, Zhao Z, Jiang L, Wan J (2012) Identification of quantitative trait loci for seed storability in rice (Oryza sativa L.). Plant Breed 131:739–743
Lin Q, Wang W, Ren Y, Jiang Y, Sun A, Qian Y, Zhang Y, He N, Hang NT, Liu Z, Li L, Liu L, Jiang L, Wan J (2015) Genetic dissection of seed storability using two different populations with a same parent rice cultivar N22. Breed Sci 65:411–419
Lu CA, Lim EK, Yu SM (1998) Sugar response sequence in the promoter of a rice α-amylase gene serves as a transcriptional enhancer. J Biol Chem 273:10120–10131
Lu CA, Lin CC, Lee KW, Chen JL, Huang LF, Ho SL et al (2007) The SnRK1A protein kinase plays a key role in sugar signaling during germination and seedling growth of rice. Plant Cell 19(8):2484–2499
Magneschi L, Perata P (2009) Rice germination and seedling growth in the absence of oxygen. Ann Bot 103(2):181–196
McDonald MB (1999) Seed deterioration: physiology, repair and assessment. Seed Sci Technol 27:177–237
McQueen-Mason SJ, Cosgrove DJ (1995) Expansin mode of action on cell walls. Plant Physiol 107:87–100
Mekonnen MM, Hoekstra AY (2016) Four billion people facing severe water scarcity. Sci Adv 2(2):e1500323
Miro B, Ismail AM (2013) Tolerance of anaerobic conditions caused by flooding during germination and early growth in rice (Oryza sativa L.). Front Plant Sci 4:1–18
Mitsui T, Yamaguchi J, Akazawa T (1996) Physicochemical and serological characterization of alpha-amylase isoforms and identification of their corresponding genes. Plant Physiol 110(4):1395–1404
Miura K, Lin SY, Yano M, Nagamine T (2002) Mapping quantitative trait loci controlling seed longevity in rice (Oryza sativa L.). Theor Appl Genet 104:981–986
Molden D (2007) Water for food, water for life: a comprehensive assessment of water management in agriculture. International Water Management Institute, London
Mondoni A, Orsenigo S, Donà M, Balestrazzi A, Probert RJ, Hay FR, Abeli T (2014) Environmentally induced transgenerational changes in seed longevity: maternal and genetic influence. Ann Bot 113(7):1257–1263
Nagavarapu T, Keshavulu K, Subba Rao LV, Sengguttuvel P, Maganti SM (2017) Validation of seedling vigour QTLs in rice (Oryza sativa L.). Curr Trends Biotechnol Pharm 11(1):24–33
Nagel M, Kranner I, Neumann K, Rolletschek H, Seal CE, Colville L, Fernández-Marín B, Börner A (2015) Genome-wide association mapping and biochemical markers reveal that seed ageing and longevity are intricately affected by genetic background and developmental and environmental conditions in barley. Plant Cell Environ 38(6):1011–1022
OCHA (2010) Water scarcity and humanitarian action: key emerging trends and challenges. In: Policy Development and Studies Branch. Occasional policy briefing series No. 4. OCHA, New York, NY. 15 p
Pal LR, Yu CH, Mount S, Moult J (2015) Insights from GWAS: emerging landscape of mechanisms underlying complex trait disease. BMC Genomics 16(8):1–15
Pathak H, Sankhyan S, Dubey D, Bhatia A, Jain N (2013) Dry direct-seeding of rice for mitigating greenhouse gas emission: field experimentation and simulation. Paddy Water Environ 11:593–601
Perata P, Pozueta-Romero J, Akazawa T, Yamaguchi J (1992) Effects of anoxia on starch breakdown in rice and wheat seeds. Planta 188:611–618
Perata P, Geshi N, Yamaguchi J, Akazawa T (1993) Effect of anoxia on the induction of α-amylase in cereal seeds. Planta 191:402–408
Petla BP, Kamble NU, Kumar M, Verma P, Ghosh S, Singh A, Rao V, Salvi P, Kaur H, Saxena SC, Majee M (2016) Rice protein L-isoaspartyl O-methyltransferase isoforms differentially accumulate during seed maturation to restrict deleterious isoAsp and reactive oxygen species accumulation and are implicated in seed vigor and longevity. New Phytol 211(2):627–645
Pingali PL (2012) Green revolution: impacts, limits, and the path ahead. Proc Natl Acad Sci U S A 109(31):12302–12308
Redoña E, Mackill D (1996) Mapping quantitative trait loci for seedling vigor in rice using RFLPs. Theor Appl Genet 92:395–402
Rice Knowledge Bank (2017) How to manage water. http://www.knowledgebank.irri.org/step-by-step-production/growth/water-management
Roberts EH (1961) The viability of rice seed in relation to temperature, moisture content, and gaseous environment. Ann Bot 25(3):381–390
Sadiq I, Fanucchi F, Paparelli E, Alpi E, Bachi A, Alpi A et al (2011) Proteomic indentification of differentially expressed proteins in the anoxic rice coleoptile. J Plant Physiol 168:2234–2243
Saika H, Matsumura H, Takano T, Tsutsumi N, Nakazono M (2006) A point mutation of Adh1 gene is involved in the repression of coleoptile elongation under submergence in rice. Breed Sci 56:69–74
Sandhu N, Torres RO, Sta Cruz MT, Maturan PC, Jain R, Kumar A, Henry A (2014) Traits and QTLs for development of dry direct-seeded rainfed rice varieties. Exp Bot 66(1):225–244
Sano N, Rajjou L, North HM, Debeaujon I, Marion-Poll A, Seo M (2016) Staying alive: molecular aspects of seed longevity. Plant Cell Physiol 57(4):660–674
Sasaki K, Fukuta Y, Sato T (2005) Mapping of quantitative trait loci controlling seed longevity of rice (Oryza sativa L.) after various periods of seed storage. Plant Breed 124:361–366
Sasaki K, Takeuchi Y, Miura K, Yamaguchi T, Ando T, Ebitani A, Higashitani T, Yamaya A, Yano M, Sato T (2015) Fine mapping of a major quantitative trait locus, qLG-9, that controls seed longevity in rice (Oryza sativa L.). Theor Appl Genet 128(4):769–778
Sehgal D, Singh R, Rajpal V (2016) Quantitative trait loci mapping in plants: concepts and approaches. In: Rajpal VR, Rao SR, Raina SN (eds) Molecular breeding for sustainable crop improvement. Springer International Publishing, Cham, pp 31–59
Septiningsih EM, Ignacio JI, Sendon PM, Sanchez DL, Ismail AM, Mackill DJ (2013) QTL mapping and confirmation for tolerance of anaerobic conditions during germination derived from the rice landrace Ma-Zhan Red. Theor Appl Genet 126:1357–1366
Shingaki-Wells RN, Huang S, Taylor NL, Carroll AJ, Zhou W, Millar A (2011) Differential molecular responses of rice and wheat coleoptiles to anoxia reveal novel metabolic adaptations in amino acid metabolism for tissue tolerance. Plant Physiol 156:1706–1724
Singh UM, Yadav S, Dixit S, Ramayya P, Devi M, Raman K et al (2017) QTL hotspots for early vigor and related traits under dry direct-seeded system in rice (Oryza sativa L.). Front Plant Sci 8:286
Takahashi H, Saika H, Matsumura H, Nagamura Y, Tsutsumi N, Nishizawa NK et al (2011) Cell division and cell elongation in the coleoptile of rice alcohol dehydrogenase 1-deficient mutant are reduced under complete submergence. Ann Bot 108(2):253–261
Teng S, Vergara BS, Alejar AA (1992) Relationship of grain length, width, and weight to seedling vigor in rice (Oryza sativa L.). Philipp J Crop Sci 17(1):17–20
Terashima M, Hosono M, Katoh S (1997) Functional roles of protein domains on rice α-amylase activity. Appl Microbiol Biotechnol 47(4):364–367
Waffenschmidt S, Woessner JP, Beer K, Goodenough UW (1993) Isodityrosine cross-linking mediates insolubilization of cell walls in Chlamydomonas. Plant Cell 5(7):809–820
Wang Z, Wang J, Bao Y, Wang F, Zhang H (2010) Quantitative trait loci analysis for rice seed vigor during the germination stage. J Zhejiang Univ Sci B (Biomed and Biotechnol) 11:958–964
Whitehouse KJ, Hay FR, Ellis RH (2017) High-temperature stress during drying improves subsequent rice (Oryza sativa L.) seed longevity. Seed Sci Res 27(4):281–291
Xiao W, Custard KD, Brown RC, Lemmon BE, Harada JJ, Goldberg RB, Fischer RL (2006) DNA methylation is critical for Arabidopsis embryogenesis and seed viability. Plant Cell 18(4):805–814
Xie L, Tan Z, Zhou Y, Xu R, Feng L, Xing Y et al (2014) Identification and fine mapping of quantitative trait loci for seed vigor in germination and seedling establishment in rice. J Integr Plant Biol 56(8):749–759
Xue Y, Zhang SQ, Yao QH, Peng RH, Xiong AS, Li X, Zhu WM, Zhu YY, Zha DS (2008) Identification of quantitative trait loci for seed storability in rice (Oryza sativa L.). Euphytica 164:739–744
Yamauchi M (1996) Development of anaerobic direct-seeding technology for rice in the tropics. Crop research in Asia: achievements and perspectives. In: Proceedings of the 2nd Asian Crop Science Conference, Fukui, Japan, pp 198–203
Yamauchi M, Winn T (1996) Rice seed vigor and seedling establishment in anaerobic soil. Crop Sci 36(3):680–686
van Zanten M, Liu Y, Soppe WJ (2013) Epigenetic signalling during the life of seeds. In: Epigenetic memory and control in plants. Springer, Berlin, pp 127–153
Zhang Z, Su L, Li W, Chen W, Zhu Y (2005) A major QTL conferring cold tolerance at the early seedling stage using recombinant inbred lines of rice (Oryza sativa L.). Plant Sci 168:527–534
Zhou L, Wang J-K, Yi Q, Wang Y-Z, Zhu Y-G, Zhang Z-H (2007) Quantitative trait loci for seedling vigor in rice under field conditions. Field Crop Res 100(2–3):294–301
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2021 The Author(s)
About this chapter
Cite this chapter
Quilloy, F.A., Labaco, B., Casal, C., Dixit, S. (2021). Crop Establishment in Direct-Seeded Rice: Traits, Physiology, and Genetics. In: Ali, J., Wani, S.H. (eds) Rice Improvement. Springer, Cham. https://doi.org/10.1007/978-3-030-66530-2_6
Download citation
DOI: https://doi.org/10.1007/978-3-030-66530-2_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-66529-6
Online ISBN: 978-3-030-66530-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)