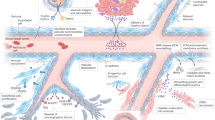
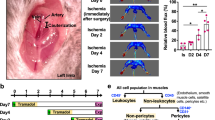
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- Ang-1:
-
Angiopoietin-1
- Ang-2:
-
Angiopoietin-2
- AP:
-
Alkaline phosphatase
- bFGF-2:
-
Basic fibroblasts growth factor – 2
- BM:
-
Basement membrane
- CLI:
-
Critical limb ischemia
- ECs:
-
Endothelial Cells
- EV:
-
Extracellular vesicle
- hiPSCs:
-
Human induced pluripotent stem cells
- IGF-1:
-
Insulin-like growth factor – 1
- ITGBL1:
-
Integrin subunit βlike – 1
- MBs:
-
Mesoangioblasts
- MSCs:
-
Mesenchymal stem cells
- Myf-5:
-
Myogenic factor-5
- NCCs:
-
Neural crest cells
- NF-kB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- NG-2:
-
Neural/glial antigen 2
- p75NTR:
-
Neurotrophine receptor p75
- PAD:
-
Peripheral arterial disease
- PAX7:
-
Paired box protein 7
- PDGFR-β:
-
Platelet-derived growth factor receptor β
- PEG:
-
Polyethylene glycol
- α-SG:
-
A-sarcoglycan
- PACs:
-
Pro-angiogenic cells
- PKC:
-
Protein kinase C
- SCs:
-
Satellite cells
- SMPs:
-
Skeletal muscle pericytes
- SHP1:
-
Src homology region 2 domain-containing phosphatase-1
- TGF-β:
-
Transforming growth factor-β
- T1D:
-
Type 1 diabetes
- T2DM:
-
Type 2 diabetes mellitus
- VCAF:
-
Vascular calcification associated factor
- VEGF:
-
Vascular endothelial growth factor
- VEZF1:
-
Vascular endothelial zinc finger-1
- VSMCs:
-
Vascular smooth muscle cells
References
Abou-Khalil R, Le Grand F, Pallafacchina G et al (2009) Autocrine and paracrine angiopoietin 1/Tie-2 signaling promotes muscle satellite cell self-renewal. Cell Stem Cell. https://doi.org/10.1016/j.stem.2009.06.001
Aicher A, Rentsch M, Sasaki KI et al (2007) Nonbone marrow-derived circulating progenitor cells contribute to postnatal neovascularization following tissue ischemia. Circ Res 100:581–589. https://doi.org/10.1161/01.RES.0000259562.63718.35
Alexander MY, Wilkinson FL, Kirton JP et al (2005) Identification and characterization of vascular calcification-associated factor, a novel gene upregulated during vascular calcification in vitro and in vivo. Arterioscler Thromb Vasc Biol. https://doi.org/10.1161/01.ATV.0000175750.94742.46
Allsopp G, Gamble HJ (1979) An electron microscopic study of the pericytes of the developing capillaries in human fetal brain and muscle. J Anat 128(Pt 1):155–168
Alvino VV, Fernández-Jiménez R, Rodriguez-Arabaolaza I et al (2018) Transplantation of allogeneic pericytes improves myocardial vascularization and reduces interstitial fibrosis in a swine model of reperfused acute myocardial infarction. J Am Heart Assoc 7. https://doi.org/10.1161/JAHA.117.006727
Armulik A, Genové G, Betsholtz C (2011) Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev Cell 21(2):193–215
Avolio E, Meloni M, Spencer HL et al (2015) Combined intramyocardial delivery of human pericytes and cardiac stem cells additively improves the healing of mouse infarcted hearts through stimulation of vascular and muscular repair. Circ Res. https://doi.org/10.1161/CIRCRESAHA.115.306146
Baum O, Bernd J, Becker S et al (2020) Structural microangiopathies in skeletal muscle related to systemic vascular pathologies in humans. Front Physiol. https://doi.org/10.3389/fphys.2020.00028
Benjamin LE, Hemo I, Keshet E (1998) A plasticity window for blood vessel remodelling is defined by pericyte coverage of the preformed endothelial network and is regulated by PDGF-B and VEGF. Development 125:1591–1598
Bergwerff M, Verberne ME, DeRuiter MC et al (1998) Neural crest cell contribution to the developing circulatory system implications for vascular morphology? Circ Res. https://doi.org/10.1161/01.RES.82.2.221
Billaud M, Donnenberg VS, Ellis BW et al (2017) Classification and functional characterization of vasa vasorum-associated perivascular progenitor cells in human aorta. Stem Cell Reports. https://doi.org/10.1016/j.stemcr.2017.04.028
Birbrair A, Wang ZM, Messi ML et al (2011) Nestin-GFP transgene reveals neural precursor cells in adult skeletal muscle. PLoS One 6. https://doi.org/10.1371/journal.pone.0016816
Birbrair A, Zhang T, Wang ZM et al (2013a) Role of pericytes in skeletal muscle regeneration and fat accumulation. Stem Cells Dev. https://doi.org/10.1089/scd.2012.0647
Birbrair A, Zhang T, Wang ZM et al (2013b) Skeletal muscle neural progenitor cells exhibit properties of NG2-glia. Exp Cell Res. https://doi.org/10.1016/j.yexcr.2012.09.008
Birbrair A, Zhang T, Wang ZM et al (2014) Pericytes: multitasking cells in the regeneration of injured, diseased, and aged skeletal muscle. Front Aging Neurosci 6(119):245
Blazquez-Martinez A, Chiesa M, Arnalich F et al (2014) C-kit identifies a subpopulation of mesenchymal stem cells in adipose tissue with higher telomerase expression and differentiation potential. Differentiation 87:147–160. https://doi.org/10.1016/j.diff.2014.02.007
Bloch EH, Iberall AS (1982) Toward a concept of the functional unit of mammalian skeletal muscle. Am J Phys. https://doi.org/10.1152/ajpregu.1982.242.5.r411
Brachvogel B, Moch H, Pausch F et al (2005) Pervascular cells expressing annexin A5 define a novel mesenchymal stem cell-like population with the capacity to differentiate into multiple mesenchymal lineages. Development. https://doi.org/10.1242/dev.01846
Bruns RR, Palade GE (1968) Studies on blood capillaries. I. General organization of blood capillaries in muscle. J Cell Biol. https://doi.org/10.1083/jcb.37.2.244
Campagnolo P, Cesselli D, Al Haj Zen A et al (2010) Human adult vena saphena contains perivascular progenitor cells endowed with clonogenic and proangiogenic potential. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.109.899252
Campagnolo P, Hong X, Di Bernardini E et al (2015a) Resveratrol-induced vascular progenitor differentiation towards endothelial lineage via miR-21/Akt/β-catenin is protective in vessel graft models. PLoS One. https://doi.org/10.1371/journal.pone.0125122
Campagnolo P, Tsai TN, Hong X et al (2015b) C-kit+ progenitors generate vascular cells for tissue-engineered grafts through modulation of the Wnt/Klf4 pathway. Biomaterials. https://doi.org/10.1016/j.biomaterials.2015.04.055
Campagnolo P, Gormley AJ, Chow LW et al (2016) Pericyte seeded dual peptide scaffold with improved endothelialization for vascular graft tissue engineering. Adv Healthc Mater. https://doi.org/10.1002/adhm.201600699
Caporali A, Pani E, Horrevoets AJG et al (2008) Neurotrophin p75 receptor (p75NTR) promotes endothelial cell apoptosis and inhibits angiogenesis: implications for diabetes-induced impaired neovascularization in ischemic limb muscles. Circ Res. https://doi.org/10.1161/CIRCRESAHA.108.177386
Caporali A, Meloni M, Völlenkle C et al (2011) Deregulation of microRNA-503 contributes to diabetes mellitus-induced impairment of endothelial function and reparative angiogenesis after limb ischemia. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.110.952325
Caporali A, Meloni M, Nailor A et al (2015) P75NTR-dependent activation of NF-κB regulates microRNA-503 transcription and pericyte-endothelial crosstalk in diabetes after limb ischaemia. Nat Commun. https://doi.org/10.1038/ncomms9024
Carmeille R, Bouvet F, Tan S et al (2016) Membrane repair of human skeletal muscle cells requires Annexin-A5. Biochim Biophys Acta, Mol Cell Res. https://doi.org/10.1016/j.bbamcr.2016.06.003
Carrabba M, De Maria C, Oikawa A et al (2016) Design, fabrication and perivascular implantation of bioactive scaffolds engineered with human adventitial progenitor cells for stimulation of arteriogenesis in peripheral ischemia. Biofabrication. https://doi.org/10.1088/1758-5090/8/1/015020
Castrechini NM, Murthi P, Gude NM et al (2010) Mesenchymal stem cells in human placental chorionic villi reside in a vascular niche. Placenta 31:203–212. https://doi.org/10.1016/j.placenta.2009.12.006
Ceriello A (2012) The emerging challenge in diabetes: the “metabolic memory”. Vasc Pharmacol 57(5–6):133–138
Chang HN, Pang JHS, Chen CPC et al (2012) The effect of aging on migration, proliferation, and collagen expression of tenocytes in response to ciprofloxacin. J Orthop Res 30(5):764–768
Chen Y, Wong MM, Campagnolo P et al (2013) Adventitial stem cells in vein grafts display multilineage potential that contributes to neointimal formation. Arterioscler Thromb Vasc Biol 33:1844–1851. https://doi.org/10.1161/ATVBAHA.113.300902
Clark ER, Clark EL (1925) A. The development of adventitial (Rouget) cells on the blood capillaries of amphibian larvae. Am J Anat. https://doi.org/10.1002/aja.1000350205
Corselli M, Chen CW, Sun B et al (2012) The tunica adventitia of human arteries and veins as a source of mesenchymal stem cells. Stem Cells Dev. https://doi.org/10.1089/scd.2011.0200
Covas DT, Piccinato CE, Orellana MD et al (2005) Mesenchymal stem cells can be obtained from the human saphena vein. Exp Cell Res 309:340–344. https://doi.org/10.1016/j.yexcr.2005.06.005
Crisan M, Yap S, Casteilla L et al (2008) A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell. https://doi.org/10.1016/j.stem.2008.07.003
D’Souza DM, Al-Sajee D, Hawke TJ (2013) Diabetic myopathy: impact of diabetes mellitus on skeletal muscle progenitor cells. Front Physiol 88:158–164
Davie NJ, Crossno JT, Frid MG et al (2004) Hypoxia-induced pulmonary artery adventitial remodeling and neovascularization: contribution of progenitor cells. Am J Physiol Lung Cell Mol Physiol 286:668–678. https://doi.org/10.1152/ajplung.00108.2003
De Palma M, Venneri MA, Galli R et al (2005) Tie2 identifies a hematopoietic lineage of proangiogenic monocytes required for tumor vessel formation and a mesenchymal population of pericyte progenitors. Cancer Cell 8:211–226. https://doi.org/10.1016/j.ccr.2005.08.002
Deasy BM, Schugar RC, Chirieleison SM et al (2009) High harvest yield, high expansion, and phenotype stability of CD146 mesenchymal stromal cells from whole primitive human umbilical cord tissue. J Biomed Biotechnol 2009. https://doi.org/10.1155/2009/789526
Dellavalle A, Sampaolesi M, Tonlorenzi R et al (2007) Pericytes of human skeletal muscle are myogenic precursors distinct from satellite cells. Nat Cell Biol. https://doi.org/10.1038/ncb1542
Dellavalle A, Maroli G, Covarello D et al (2011) Pericytes resident in postnatal skeletal muscle differentiate into muscle fibres and generate satellite cells. Nat Commun. https://doi.org/10.1038/ncomms1508
Devana SK, Kelley BV, McBride OJ et al (2018) Adipose-derived human perivascular stem cells may improve achilles tendon healing in rats. Clin Orthop Relat Res. https://doi.org/10.1097/CORR.0000000000000461
Díaz-Flores L, Gutiérrez R, Madrid JF et al (2009) Pericytes. Morphofunction, interactions and pathology in a quiescent and activated mesenchymal cell niche. Histol Histopathol 24(7):909–969
Doherty MJ, Ashton BA, Walsh S et al (1998) Vascular pericytes express osteogenic potential in vitro and in vivo. J Bone Miner Res 13:828–838. https://doi.org/10.1359/jbmr.1998.13.5.828
Dulmovits BM, Herman IM (2012) Microvascular remodeling and wound healing: a role for pericytes. Int J Biochem Cell Biol 44(11):1800–1812
Faglia E, Clerici G, Clerissi J et al (2006) Early and five-year amputation and survival rate of diabetic patients with critical limb ischemia: data of a cohort study of 564 patients. Eur J Vasc Endovasc Surg. https://doi.org/10.1016/j.ejvs.2006.03.006
Fang S, Wei J, Pentinmikko N et al (2012) Generation of functional blood vessels from a single c-kit+ adult vascular endothelial stem cell. PLoS Biol 10. https://doi.org/10.1371/journal.pbio.1001407
Farrington-Rock C, Crofts NJ, Doherty MJ et al (2004) Chondrogenic and adipogenic potential of microvascular pericytes. Circulation. https://doi.org/10.1161/01.CIR.0000144457.55518.E5
Faulkner A, Tamiato A, Cathery W, Rampin A, Garcia EJ, Allen S, Mellor H, Lisa Heather GS, PM (2020) 2-oxoglutarate improves redox balance and mitochondrial function in muscle pericytes of type-2 diabetic patients. Diabetologia. https://doi.org/10.1007/s00125-020-05230-4
Fuoco C, Sangalli E, Vono R et al (2014) 3D hydrogel environment rejuvenates aged pericytes for skeletal muscle tissue engineering. Front Physiol. https://doi.org/10.3389/fphys.2014.00203
Garoffolo G, Ruiter MS, Piola M et al (2020) Coronary artery mechanics induces human saphenous vein remodelling via recruitment of adventitial myofibroblast-like cells mediated by thrombospondin-1. Theranostics 10:2597–2611. https://doi.org/10.7150/thno.40595
Geraldes P, Hiraoka-Yamamoto J, Matsumoto M et al (2009) Activation of PKC-and SHP-1 by hyperglycemia causes vascular cell apoptosis and diabetic retinopathy. Nat Med. https://doi.org/10.1038/nm.2052
Gitiaux C, Kostallari E, Lafuste P et al (2013) Whole microvascular unit deletions in dermatomyositis. Ann Rheum Dis. https://doi.org/10.1136/annrheumdis-2012-201822
Hirsch AT, Haskal ZJ, Hertzer NR et al (2006) ACC/AHA 2005 practice guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic). Circulation 113(11):e463–e654
Hoehn K, Marieb EN (2012) Human anatomy & physiology. Benjamin-Cummings Publ Co, San Francisco. https://doi.org/10.1007/BF00845519
Hoong SL, Blann AD, Aun YC et al (2004) Plasma vascular endothelial growth factor, angiopoietin-1, and angiopoietin-2 in diabetes: implications for cardiovascular risk and effects of multifactorial intervention. Diabetes Care. https://doi.org/10.2337/diacare.27.12.2918
Howson KM, Aplin AC, Gelati M et al (2005) The postnatal rat aorta contains pericyte progenitor cells that form spheroidal colonies in suspension culture. Am J Physiol Cell Physiol. https://doi.org/10.1152/ajpcell.00168.2005
Hu Y, Zhang Z, Torsney E et al (2004) Abundant progenitor cells in the adventitia contribute to atheroscleroses of vein grafts in ApoE-deficient mice. J Clin Invest 113:1258–1265. https://doi.org/10.1172/JCI19628
Ieronimakis N, Hays AL, Janebodin K et al (2013) Coronary adventitial cells are linked to perivascular cardiac fibrosis via TGF$β$1 signaling in the mdx mouse model of Duchenne muscular dystrophy. J Mol Cell Cardiol 63:122–134. https://doi.org/10.1016/j.yjmcc.2013.07.014
Invernici G, Emanueli C, Madeddu P et al (2007) Human fetal aorta contains vascular progenitor cells capable of inducing vasculogenesis, angiogenesis, and myogenesis in vitro and in a murine model of peripheral ischemia. Am J Pathol 170:1879–1892. https://doi.org/10.2353/ajpath.2007.060646
James AW, Zara JN, Corselli M et al (2012a) An abundant perivascular source of stem cells for bone tissue engineering. Stem Cells Transl Med. https://doi.org/10.5966/sctm.2012-0053
James AW, Zara JN, Zhang X et al (2012b) Perivascular stem cells: a prospectively purified mesenchymal stem cell population for bone tissue engineering. Stem Cells Transl Med 1:510–519. https://doi.org/10.5966/sctm.2012-0002
Katare R, Riu F, Mitchell K et al (2011) Transplantation of human pericyte progenitor cells improves the repair of infarcted heart through activation of an angiogenic program involving micro-RNA-132. Circ Res 109:894–906. https://doi.org/10.1161/CIRCRESAHA.111.251546
Khan WS, Tew SR, Adesida AB, Hardingham TE (2008) Human infrapatellar fat pad-derived stem cells express the pericyte marker 3G5 and show enhanced chondrogenesis after expansion in fibroblast growth factor-2. Arthritis Res Ther 10:1–11. https://doi.org/10.1186/ar2448
Kim MS, Dean LS (2011) In-stent restenosis. Cardiovasc Ther 29(3):190–198
Kim SJ, Kim SA, Choi YA et al (2020) Alpha-smooth muscle actin-positive perivascular cells in diabetic retina and choroid. Int J Mol Sci 21. https://doi.org/10.3390/ijms21062158
Klatte-Schulz F, Pauly S, Scheibel M et al (2012) Influence of age on the cell biological characteristics and the stimulation potential of male human tenocyte-like cells. Eur Cells Mater. https://doi.org/10.22203/eCM.v024a06
Klein D, Weißhardt P, Kleff V et al (2011) Vascular wall-resident CD44+ multipotent stem cells give rise to pericytes and smooth muscle cells and contribute to new vessel maturation. PLoS One. https://doi.org/10.1371/journal.pone.0020540
Kostallari E, Baba-Amer Y, Alonso-Martin S et al (2015) Pericytes in the myovascular niche promote post-natal myofiber growth and satellite cell quiescence. Development. https://doi.org/10.1242/dev.115386
Kramann R, Schneider RK, Dirocco DP et al (2015) Perivascular Gli1+ progenitors are key contributors to injury-induced organ fibrosis. Cell Stem Cell 16:51–66. https://doi.org/10.1016/j.stem.2014.11.004
Kramann R, Goettsch C, Wongboonsin J et al (2016) Adventitial MSC-like cells are progenitors of vascular smooth muscle cells and drive vascular calcification in chronic kidney disease. Cell Stem Cell 19:628–642. https://doi.org/10.1016/j.stem.2016.08.001
Kumar A, D’Souza SS, Moskvin OV et al (2017) Specification and diversification of pericytes and smooth muscle cells from mesenchymoangioblasts. Cell Rep. https://doi.org/10.1016/j.celrep.2017.05.019
Larson DM, Carson MP, Haudenschild CC (1987) Junctional transfer of small molecules in cultured bovine brain microvascular endothelial cells and pericytes. Microvasc Res. https://doi.org/10.1016/0026-2862(87)90052-5
Lavasani M, Thompson SD, Pollett JB et al (2014) Human muscle-derived stem/progenitor cells promote functional murine peripheral nerve regeneration. J Clin Invest. https://doi.org/10.1172/JCI44071
Lepper C, Partridge TA, Fan CM (2011) An absolute requirement for pax7-positive satellite cells in acute injury-induced skeletal muscle regeneration. Development. https://doi.org/10.1242/dev.067595
Leszczynska A, O’Doherty A, Farrell E et al (2016) Differentiation of vascular stem cells contributes to ectopic calcification of atherosclerotic plaque. Stem Cells. https://doi.org/10.1002/stem.2315
Li Z, Yan G, Diao Q et al (2019) Transplantation of human endometrial perivascular cells with elevated CYR61 expression induces angiogenesis and promotes repair of a full-thickness uterine injury in rat. Stem Cell Res Ther 10:1–16. https://doi.org/10.1186/s13287-019-1272-3
Lusis AJ (2000) Atherosclerosis. Nature 407(6801):233–234
Maier CL, Shepherd BR, Yi T, Pober JS (2010) Explant outgrowth, propagation and characterization of human pericytes. Microcirculation 17:367–380. https://doi.org/10.1111/j.1549-8719.2010.00038.x
Majesky MW, Horita H, Ostriker A et al (2017) Differentiated smooth muscle cells generate a subpopulation of resident vascular progenitor cells in the adventitia regulated by Klf4. Circ Res 120:296–311. https://doi.org/10.1161/CIRCRESAHA.116.309322
Mazurek R, Dave JM, Chandran RR et al (2017) Vascular cells in blood vessel wall development and disease. In: Advances in pharmacology, APHA. Academic, London
Mekala SR, Wörsdörfer P, Bauer J et al (2018) Generation of cardiomyocytes from vascular adventitia-resident stem cells. Circ Res 123:686–699. https://doi.org/10.1161/CIRCRESAHA.117.312526
Michelis KC, Nomura-Kitabayashi A, Lecce L et al (2018) CD90 identifies adventitial mesenchymal progenitor cells in adult human medium- and large-sized arteries. Stem Cell Reports 11:242–257. https://doi.org/10.1016/j.stemcr.2018.06.001
Montani D, Perros F, Gambaryan N et al (2011) C-kit-positive cells accumulate in remodeled vessels of idiopathic pulmonary arterial hypertension. Am J Respir Crit Care Med 184:116–123. https://doi.org/10.1164/rccm.201006-0905OC
Montemurro T, Andriolo G, Montelatici E et al (2011) Differentiation and migration properties of human foetal umbilical cord perivascular cells: potential for lung repair. J Cell Mol Med 15:796–808. https://doi.org/10.1111/j.1582-4934.2010.01047.x
Morgan JE, Pagel CN, Sherrratt T, Partridge TA (1993) Long-term persistence and migration of myogenic cells injected into pre-irradiated muscles of mdx mice. J Neurol Sci. https://doi.org/10.1016/0022-510X(93)90224-M
Nehls V, Drenckhahn D (1991) Heterogeneity of microvascular pericytes for smooth muscle type alpha-actin. J Cell Biol. https://doi.org/10.1083/jcb.113.1.147
Nehls V, Drenckhahn D (1993) The versatility of microvascular pericytes: from mesenchyme to smooth muscle? Histochemistry 99:1–12
Nehls V, Denzer K, Drenckhahn D (1992) Pericyte involvement in capillary sprouting during angiogenesis in situ. Cell Tissue Res. https://doi.org/10.1007/BF00645048
Ni Z, Deng J, Potter CMF et al (2019) Recipient c-kit lineage cells repopulate smooth muscle cells of transplant arteriosclerosis in mouse models. Circ Res 125:223–241. https://doi.org/10.1161/CIRCRESAHA.119.314855
Ozerdem U, Grako KA, Dahlin-Huppe K et al (2001) NG2 proteoglycan is expressed exclusively by mural cells during vascular morphogenesis. Dev Dyn. https://doi.org/10.1002/dvdy.1200
Pasquinelli G, Tazzari PL, Vaselli C et al (2007) Thoracic aortas from multiorgan donors are suitable for obtaining resident angiogenic mesenchymal stromal cells. Stem Cells. https://doi.org/10.1634/stemcells.2006-0731
Passman JN, Dong XR, Wu SP et al (2008) A sonic hedgehog signaling domain in the arterial adventitia supports resident Sca1+ smooth muscle progenitor cells. Proc Natl Acad Sci U S A 105:9349–9354. https://doi.org/10.1073/pnas.0711382105
Paulson M (1992) Basement membrane proteins: structure, assembly, and cellular interactions. Crit Rev Biochem Mol Biol. https://doi.org/10.3109/10409239209082560
Prattichizzo F, Giuliani A, De Nigris V et al (2016) Extracellular microRNAs and endothelial hyperglycaemic memory: a therapeutic opportunity? Diabetes Obes Metab 18(9):855–867
Psaltis PJ, Puranik AS, Spoon DB et al (2014) Characterization of a resident population of adventitial macrophage progenitor cells in postnatal vasculature. Circ Res 115:364–375. https://doi.org/10.1161/CIRCRESAHA.115.303299
Robin C, Bollerot K, Mendes S et al (2009) Human placenta is a potent hematopoietic niche containing hematopoietic stem and progenitor cells throughout development. Cell Stem Cell 5:385–395. https://doi.org/10.1016/j.stem.2009.08.020
Romeo G, Liu WH, Asnaghi V et al (2002) Activation of nuclear factor-κB induced by diabetes and high glucose regulates a proapoptotic program in retinal pericytes. Diabetes. https://doi.org/10.2337/diabetes.51.7.2241
Rouget C (1873) Memoire sur le developpement, la structure et les proprietes physiologiques des capillaries sanguins et lymphatiques. Arch Physiol Norm Pathol 5:603–663
Russell JS, Brown JM (2014) Circulating mouse Flk1+/c-kit+/CD45- cells function as endothelial progenitors cells (EPCs) and stimulate the growth of human tumor xenografts. Mol Cancer 13:1–16. https://doi.org/10.1186/1476-4598-13-177
Saeedi P, Petersohn I, Salpea P et al (2019) Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res Clin Pract. https://doi.org/10.1016/j.diabres.2019.107843
Sainz J, Zen AAH, Caligiuri G et al (2006) Isolation of “side population” progenitor cells from healthy arteries of adult mice. Arterioscler Thromb Vasc Biol 26:281–286. https://doi.org/10.1161/01.ATV.0000197793.83391.91
Sallum AME, Varsani H, Holton JL et al (2013) Morphometric analyses of normal pediatric brachial biceps and quadriceps muscle tissue. Histol Histopathol. https://doi.org/10.14670/HH-28.525
Sambasivan R, Yao R, Kissenpfennig A et al (2011) Pax7-expressing satellite cells are indispensable for adult skeletal muscle regeneration. Development. https://doi.org/10.1242/dev.067587
Sampaolesi M, Torrente Y, Innocenzi A et al (2003) Cell therapy of α-sarcoglycan null dystrophic mice through intra-arterial delivery of mesoangioblasts. Science (80-). https://doi.org/10.1126/science.1082254
Sarugaser R, Lickorish D, Baksh D et al (2005) Human umbilical cord perivascular (HUCPV) cells: a source of mesenchymal progenitors. Stem Cells 23:220–229. https://doi.org/10.1634/stemcells.2004-0166
Schwab KE, Gargett CE (2007) Co-expression of two perivascular cell markers isolates mesenchymal stem-like cells from human endometrium. Hum Reprod 22:2903–2911. https://doi.org/10.1093/humrep/dem265
Shepro D, Morel NM (1993) Pericyte physiology. FASEB J. https://doi.org/10.1096/fasebj.7.11.8370472
Shi Y, O’Brien JE, Fard A et al (1996) Adventitial myofibroblasts contribute to neointimal formation in injured porcine coronary arteries. Circulation. https://doi.org/10.1161/01.CIR.94.7.1655
Spinetti G, Fortunato O, Caporali A et al (2013) MicroRNA-15a and MicroRNA-16 impair human circulating proangiogenic cell functions and are increased in the proangiogenic cells and serum of patients with critical limb ischemia. Circ Res. https://doi.org/10.1161/CIRCRESAHA.111.300418
Spinetti G, Sangalli E, Tagliabue E et al (2020) microRNA-21/PDCD4 proapoptotic signaling from circulating CD34 + cells to vascular endothelial cells: a potential contributor to adverse cardiovascular outcomes in patients with critical limb ischemia. Diabetes Care. https://doi.org/10.2337/dc19-2227
Tedesco FS, Gerli MFM, Perani L et al (2012) Transplantation of genetically corrected human iPSC-derived progenitors in mice with limb-girdle muscular dystrophy. Sci Transl Med. https://doi.org/10.1126/scitranslmed.3003541
Tigges U, Komatsu M, Stallcup WB (2013) Adventitial pericyte progenitor/mesenchymal stem cells participate in the restenotic response to arterial injury. J Vasc Res 50:134–144. https://doi.org/10.1159/000345524
Tilton RG, Kilo C, Williamson JR (1979) Pericyte-endothelial relationships in cardiac and skeletal muscle capillaries. Microvasc Res. https://doi.org/10.1016/0026-2862(79)90041-4
Tilton RG, Faller AM, Burkhardt JK et al (1985) Pericyte degeneration and acellular capillaries are increased in the feet of human diabetic patients. Diabetologia. https://doi.org/10.1007/BF00703132
Toledo-Flores D, Williamson A, Schwarz N et al (2019) Vasculogenic properties of adventitial Sca-1 + CD45 + progenitor cells in mice: a potential source of vasa vasorum in atherosclerosis. Sci Rep 9:1–15. https://doi.org/10.1038/s41598-019-43765-8
Torsney E, Mandal K, Halliday A et al (2007) Characterisation of progenitor cells in human atherosclerotic vessels. Atherosclerosis 191:259–264. https://doi.org/10.1016/j.atherosclerosis.2006.05.033
Tsai TN, Kirton JP, Campagnolo P et al (2012) Contribution of stem cells to neointimal formation of decellularized vessel grafts in a novel mouse model. Am J Pathol 181:362–373. https://doi.org/10.1016/j.ajpath.2012.03.021
Tsang WP, Shu Y, Kwok PL et al (2013) CD146+ human umbilical cord perivascular cells maintain stemness under hypoxia and as a cell source for skeletal regeneration. PLoS One 8:1–13. https://doi.org/10.1371/journal.pone.0076153
Tsukada T, Tippens D, Gordon D et al (1987) HHF35, a muscle-actin-specific monoclonal antibody. I. Immunocytochemial and biochemical characterization. Am J Pathol 126(1):51–60
Van Dijk CGM, Nieuweboer FE, Pei JY et al (2015) The complex mural cell: pericyte function in health and disease. Int J Cardiol 190:75–89
Varu VN, Hogg ME, Kibbe MR (2010) Critical limb ischemia. J Vasc Surg 51(1):230–241
Vono R, Fuoco C, Testa S et al (2016) Activation of the pro-oxidant PKCβIIp66Shc signaling pathway contributes to pericyte dysfunction in skeletal muscles of patients with diabetes with critical limb ischemia. Diabetes. https://doi.org/10.2337/db16-0248
Wang G, Jacquet L, Karamariti E, Xu Q (2015) Origin and differentiation of vascular smooth muscle cells. J Physiol. https://doi.org/10.1113/JP270033
Wang Y, Xu J, Chang L et al (2019) Relative contributions of adipose-resident CD146+ pericytes and CD34+ adventitial progenitor cells in bone tissue engineering. NPJ Regen Med 4. https://doi.org/10.1038/s41536-018-0063-2
White RB, Biérinx AS, Gnocchi VF, Zammit PS (2010) Dynamics of muscle fibre growth during postnatal mouse development. BMC Dev Biol. https://doi.org/10.1186/1471-213X-10-21
Wilkinson FL, Liu Y, Rucka AK et al (2007) Contribution of VCAF-positive cells to neovascularization and calcification in atherosclerotic plaque development. J Pathol. https://doi.org/10.1002/path.2114
Wong MM, Winkler B, Karamariti E et al (2013) Sirolimus stimulates vascular stem/progenitor cell migration and differentiation into smooth muscle cells via epidermal growth factor receptor/extracellular signal-regulated kinase/$β$-catenin signaling pathway. Arterioscler Thromb Vasc Biol 33:2397–2406. https://doi.org/10.1161/ATVBAHA.113.301595
Yokota T, Ma RC, Park JY et al (2003) Role of protein kinase C on the expression of platelet-derived growth factor and endothelin-1 in the retina of diabetic rats and cultured retinal capillary pericytes. Diabetes. https://doi.org/10.2337/diabetes.52.3.838
Zengin E, Chalajour F, Gehling UM et al (2006) Vascular wall resident progenitor cells: a source for postnatal vasculogenesis. Development. https://doi.org/10.1242/dev.02315
Zhang S, Ba K, Wu L et al (2015) Adventitial cells and perictyes support chondrogenesis through different mechanisms in 3-dimensional cultures with or without nanoscaffolds. J Biomed Nanotechnol. https://doi.org/10.1166/jbn.2015.2112
Zimmerlin L, Donnenberg VS, Pfeifer ME et al (2010) Stromal vascular progenitors in adult human adipose tissue. Cytom Part A 77:22–30. https://doi.org/10.1002/cyto.a.20813
Zimmermann KW (1923) Der feinere Bau der Blutcapillaren. Z Anat Entwicklungsgesch. https://doi.org/10.1007/bf02593544
Acknowledgments
We thank Dr. Sadie Slater, University of Bristol, for proofreading the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Ethics declarations
Disclosure of Interests: All authors declare they have no conflict of interest.
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Rampin, A., Campagnolo, P., Mastrullo, V., Madeddu, P., Spinetti, G. (2021). Adventitial and Skeletal Muscle Pericytes in Health and Ischemic Tissue Regeneration. In: Birbrair, A. (eds) Biology of Pericytes – Recent Advances. Stem Cell Biology and Regenerative Medicine, vol 68. Humana, Cham. https://doi.org/10.1007/978-3-030-62129-2_11
Download citation
DOI: https://doi.org/10.1007/978-3-030-62129-2_11
Published:
Publisher Name: Humana, Cham
Print ISBN: 978-3-030-62128-5
Online ISBN: 978-3-030-62129-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)