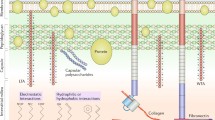
Abstract
In this chapter, an overview of current medical implant devices and infection problems associated with implantation is provided, bridging the gap between material engineering and clinical practice. The pathogenesis, common pathogens, and infection sites are listed, alongside the details of up-to-date strategies and guidelines for diagnosis and treatment of biomaterials-associated infections. Through the combined understanding of microbial pathogenicity, drug resistance, patients’ immune response processes, and current clinical practices, we can tackle the problem of biomaterials-associated infection via multidisciplinary approaches. To meet the clinical demands and challenges in future, strategic design of intelligent biomaterials is in need to reduce implantation device-caused infections, improving the patient’s quality of life.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Biomaterial industry in China. CCM Data & Business Intelligence 2016
Nana A, Nelson SB, McLaren A, Chen AF (2016) What’s new in musculoskeletal infection: update on biofilms. JBJS 98(14):1226–1234
Europe, Health & Medicine, Pharmaceuticals & Biotech (2019) Asia pacific orthopedic biomaterial market 2019, industry analysis, size, share, trends, product price, profit, forecast to 2023
Patent application trend survey report summary (artificial organs). https://www.jpo.go.jp/shiryou/pdf/gidou-houkoku/h28/28_06.pdf. Accessed 8 May 2019
Kheir MM, Tan TL, Shohat N, Foltz C, Parvizi J (2018) Routine diagnostic tests for periprosthetic joint infection demonstrate a high false-negative rate and are influenced by the infecting organism. JBJS 100(23):2057–2065
US Disease Control and Prevention (CDC). Appropriate antibiotic use in hospitals and long-term care. https://www.cdc.gov/antibiotic-use/healthcare/index.html. Accessed 26 April 2019
Use of International Standard ISO 10993-1. Biological evaluation of medical devices—part 1: evaluation and testing within a risk management process. https://www.iso.org/standard/44908.html. Accessed 26 April 2019
Holzapfel BM, Reichert JC, Schantz JT, Gbureck U, Rackwitz L, Noth U, Jakob F, Rudert M, Groll J, Hutmacher DW (2013) How smart do biomaterials need to be? A translational science and clinical point of view. Adv Drug Deliv Rev 65(4):581–603
Singh B (2018) NanoBioMaterials: nanobiomaterials. CRC Press
Yamamoto R (2017) Changes and prospects of implant materials from the viewpoint of biocompatibility. Materia 56(3):225–228. (in Japanese)
Gristina AG, Naylor P, Myrvik Q (1989) Infections from biomaterials and implants-a race for the surface. Med Prog Technol 14(3–4):205–224
Busscher HJ, van der Mei HC, Subbiahdoss G, Jutte PC, van den Dungen JJ, Zaat SA, Schultz MJ, Grainger DW (2012) Biomaterial-associated infection: locating the finish line in the race for the surface. Sci Transl Med 4(153):153rv10
Kapadia BH, Berg RA, Daley JA, Fritz J, Bhave A, Mont MA (2016) Periprosthetic joint infection. Lancet 387(10016):386–394
Kannan MB, Walter R, Yamamoto A (2015) Biocompatibility and in vitro degradation behavior of magnesium–calcium alloy coated with calcium phosphate using an unconventional electrolyte. ACS Biomater Sci Eng 2(1):56–64
Baidya AK, Bhattacharya S, Dubey GP, Mamou G, Ben-Yehuda S (2018) Bacterial nanotubes: a conduit for intercellular molecular trade. Curr Opin Microbiol 42:1–6
Bryers JD (2018) Modeling biofilm accumulation. Physiological models in microbiology volume II, Chapter 11
de la Fuente-Nunez C, Reffuveille F, Fernandez L, Hancock REW (2013) Bacterial biofilm development as a multicellular adaptation: antibiotic resistance and new therapeutic strategies. Curr Opin Microbiol 16(5):580–589
Flemming HC, Wingender J (2010) The biofilm matrix. Nat Rev Microbiol 8(9):623–633
Romling U, Balsalobre C (2012) Biofilm infections, their resilience to therapy and innovative treatment strategies. J Intern Med 272(6):541–561
Sasaki K (2016) Polysaccharide intercellular adhesion (PIA) process also happens which involves staphylococcal surface protein. J Jpn Soc Poweder Powder Metallurgy 63(8). (in Japanese)
Guidelines for Biomaterial Research That Supports Medical Care (2017) Annual meeting of Japan biomaterial engineering committee. (in Japanese)
Wolfmeier H, Pletzer D, Mansour SC, Hancock REW (2018) New perspectives in biofilm eradication. ACS Infect Dis 4(2):93–106
Guilhen C, Forestier C, Balestrino D (2017) Biofilm dispersal: multiple elaborate strategies for dissemination of bacteria with unique properties. Mol Microbiol 105(2):188–210
Guo Y, Shao HY. Characteristics and challenges of human implant infection (in Chinese). Beijing-Hong Kong infection Forum 2018
Osmon DR, Berbari EF, Berendt AR, Lew D, Zimmerli W, Steckelberg JM, Rao N, Hanssen A, Wilson WR (2013) Diagnosis and management of prosthetic joint infection: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis 56(1):e1–e25
Elkins JM, Kates S, Lange J, Lange J, Lichstein P, Otero J, Soriano A, Wagner C, Wouthuyzen-Bakker M (2019) General assembly, diagnosis, definitions: proceedings of international consensus on orthopedic infections. J Arthroplast 34(2):S181–S185
Parvizi J, Della Valle CJ (2010) AAOS Clinical Practice guideline: diagnosis and treatment of periprosthetic joint infections of the hip and knee. J Am Acad Orthop Surg 18(12):771–772
Drago L, Lidgren L, Bottinelli E, Villafañe JH, Berjano P, Banfi G, Romanò CL, Sculco TP (2016) Mapping of microbiological procedures by the members of the International Society of Orthopaedic Centers (ISOC) for diagnosis of periprosthetic infections. J Clin Microbiol 54(5):1402–1403
Higuera CA, Zmistowski B, Malcom T, Barsoum WK, Sporer SM, Mommsen P, Kendoff D, Della Valle CJ, Parvizi J (2017) Synovial fluid cell count for diagnosis of chronic periprosthetic hip infection. JBJS 99(9):753–759
Johns Hopkins ABX Guide. 2018
Guidelines for clinical application of antimicrobial agents (2015 edition, in Chinese). National Health and Family Planning Commission
Thoendel M, Jeraldo P, Greenwood-Quaintance KE, Chia N, Abdel MP, Steckelberg JM, Osmon DR, Patel R (2017) A novel prosthetic joint infection pathogen, Mycoplasma salivarium, identified by metagenomic shotgun sequencing. Clin Infect Dis 65(2):332–335
Cabrita HB, Croci AT, de Camargo OP, de Lima A (2007) Prospective study of the treatment of infected hip arthroplasties with or without the use of an antibiotic-loaded cement spacer. Clinics 62(2):99–108
Del Pozo JL, Patel R (2009) Infection associated with prosthetic joints. N Engl J Med 361(8):787–794
Wang Y, Cheng LI, Helfer DR, Ashbaugh AG, Miller RJ, Tzomides AJ, Thompson JM, Ortines RV, Tsai AS, Liu H, Dillen CA, Archer NK, Cohen TS, Tkaczyk C, Stover CK, Sellman BR, Miller LS (2017) Mouse model of hematogenous implant-related Staphylococcus aureus biofilm infection reveals therapeutic targets. Proc Natl Acad Sci U S A 114(26):E5094–E5102
Wang Z, de la Fuente-Nunez C, Shen Y, Haapasalo M, Hancock REW (2015) Treatment of oral multispecies biofilms by an anti-biofilm peptide. PLoS One 10(7):e0132512
Gao Q, Yu M, Su Y, Xie M, Zhao X, Li P, Ma PX (2017) Rationally designed dual functional block copolymers for bottlebrush-like coatings: in vitro and in vivo antimicrobial, antibiofilm, and antifouling properties. Acta Biomater 51:112–124
Gross MS, Phillips EA, Carrasquillo RJ, Thornton A, Greenfield JM, Levine LA, Alukal JP, Conners WP, Glina S, Tanrikut C, Honig SC, Becher EF, Bennett NE, Wang R, Perito PE, Stahl PJ, Gaya MR, Barbara MR, Cedeno JD, Gheiler EL, Kalejaiye O, Ralph DJ, Kohler TS, Stember DS, Carrion RE, Maria PP, Brant WO, Bickell MW, Garber BB, Pineda M, Burnett AL, Eid JF, Henry GD, Munarriz RM (2017) Multicenter investigation of the micro-organisms involved in penile prosthesis infection: an analysis of the efficacy of the AUA and EAU guidelines for penile prosthesis prophylaxis. J Sex Med 14(3):455–463
Habous M, Tal R, Tealab A, Soliman T, Nassar M, Mekawi Z, Mahmoud S, Abdelwahab O, Elkhouly M, Kamr H, Remeah A, Binsaleh S, Ralph D, Mulhall J (2018) Defining a glycated haemoglobin (HbA1c) level that predicts increased risk of penile implant infection. BJU Int 121(2):293–300
Scholz HC, Nockler K, Gollner C, Bahn P, Vergnaud G, Tomaso H, Al Dahouk S, Kampfer A, Cloeckaert M, Maquart MS, Zygmunt AM, Whatmore M, Pfeffer B, Huber P, Busse HJ, De BK (2010) Brucella inopinata sp nov., isolated from a breast implant infection. Int J Syst Evol Microbiol 60:801–808
Hoque J, Haldar J (2017) Direct synthesis of dextran-based antibacterial hydrogels for extended release of biocides and eradication of topical biofilms. ACS Appl Mater Interfaces 9(19):15975–15985
Mao X, Cheng R, Zhang H, Bae J, Cheng L, Zhang L, Deng L, Cui W, Zhang Y, Santos HA, Sun X (2019) Self-healing and injectable hydrogel for matching skin flap regeneration. Adv Sci (Weinh) 6(3):1801555
Hoque J, Adhikary U, Yadav V, Samaddar S, Konai MM, Prakash RG, Pararnanandham K, Shome BR, Sanyal K, Haldar J (2016) Chitosan derivatives active against multidrug-resistant bacteria and pathogenic fungi: in vivo evaluation as topical antimicrobials. Mol Pharm 13(10):3578–3589
Moran GJ, Krishnadasan A, Gorwitz RJ, Fosheim GE, McDougal LK, Carey RB, Talan DA, Grp EMINS (2006) Methicillin-resistant S-aureus infections among patients in the emergency department. N Engl J Med 355(7):666–674
Scott VCS, Haake DA, Churchill BM, Justice SS, Kim JH (2015) Intracellular bacterial communities: a potential etiology for chronic lower urinary tract symptoms. Urology 86(3):425–431
Minassian AM, Newnham R, Kalimeris E, Bejon P, Atkins BL, Bowler IC (2014) Use of an automated blood culture system (BD BACTEC™) for diagnosis of prosthetic joint infections: easy and fast. BMC Infect Dis 14(1):233
Heudorf U, Albert-Braun S, Hunfeld KP, Birne FU, Schulze J, Strobel K, Petscheleit K, Kempf VA, Brandt C (2016) Multidrug-resistant organisms in refugees: prevalences and impact on infection control in hospitals. GMS Hyg Infect Control 11
US Public Law 105-230 105th Congress, Biomaterials Access Assurance Act of 1998. https://www.biomaterials.org/sites/default/files/docs/2014/biomaterials_access_assurance_act_of_1998.pdf. Accessed 26 April 2019
Acknowledgments
This work was financially supported by the National Key R&D Program of China (2018YFC1105402 and 2017YFA0207202), the National Natural Science Foundation of China (21875189 and 81601553), Key R&D Program of Jiangsu Province (BE2017740), the Natural Science Foundation of Zhejiang Province (LGF19H200005), and the Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Bai, D., Chen, J., Li, P., Huang, W. (2020). Perspectives on Biomaterial-Associated Infection: Pathogenesis and Current Clinical Demands. In: Li, B., Moriarty, T., Webster, T., Xing, M. (eds) Racing for the Surface. Springer, Cham. https://doi.org/10.1007/978-3-030-34475-7_4
Download citation
DOI: https://doi.org/10.1007/978-3-030-34475-7_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-34474-0
Online ISBN: 978-3-030-34475-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)