Abstract
Corticomuscular and intermuscular coherence are established methods to study connectivity between activity of neurons in sensorimotor cortex measured with electroencephalography (EEG) and muscle measured with electromyography (EMG), or between muscles, in a variety of neurological conditions. However, there is a debate on the importance of EMG signal rectification before coherence estimation. This paper studies the effects of EMG rectification in corticomuscular and intermuscular coherence estimation from SCA2 patients and prodromal SCA2 gene mutation carriers in comparison to healthy controls. EEG and EMG were recorded from 20 SCA2 patients, 16 prodromal SCA2 gene mutation carriers and 26 healthy control subjects during a motor task in upper or lower limbs. Coherence estimations were carried out using the non-rectified raw EMG signal vs. the rectified EMG signal. The results showed that EMG rectification impairs the level of significance of the differences in corticomuscular and intermuscular coherence between SCA2 patients and prodromal SCA2 gene mutation carriers vs. healthy controls in the beta-band, and also results in overall lower coherence values.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Spinocerebellar ataxia type 2 (SCA2) is caused by an abnormal expansion of the CAG trinucleotide in the ataxin-2 gen [1, 2]. This disease includes a wide range of clinical manifestation like cerebellar syndrome, saccadic slowing, cognitive decline, sensory neuropathy and corticospinal tract damage [3,4,5,6,7,8,9]. Signs of corticospinal tract dysfunction had been found on clinical (ex. hyperreflexia and spasticity), anatomic (ex. degeneration of the motor cortex) and electrophysiology studies of SCA2 patients and prodromal SCA2 gene mutation carriers [7, 10,11,12]. Electrophysiological studies had demonstrated the increase of resting motor thresholds (RMT) and central motor conduction time (CMCT) by using transcranial magnetic stimulation (TMS) [13, 14]. Corticomuscular coherence (CMC) and intermuscular coherence (IMC) estimation are well-established methods to study connectivity between activity of neurons in sensorimotor cortex (EEG or MEG) and muscle (EMG) or between muscles [15,16,17,18]. These measures have been reported in a variety of neurological condition such as stroke [17], Parkinson disease [19], sleep behavior disorder [20] and recently in SCA2 [10, 21] as a marker of corticospinal tract dysfunction.
However, there has been a discussion about the processing steps to estimate the CMC and IMC [22,23,24,25]. The discussion mainly focused on the appropriateness of EMG signal rectification prior to coherence estimation. The main concern is about CMC estimation being a frequency domain method used to demonstrate a linear coupling between rhythmic activity from sensorimotor cortex and the activity of voluntarily contracting muscles [26, 27], but rectification is a non-linear operator that changes the frequency components of the signal to which it is applied [24]. Theoretical, simulation and experimental studies had been conducted to study the influence of the rectification on the EMG signal spectrum to understand its effect on neural connectivity [28]. Some authors claimed that the rectification is a necessary step [29,30,31,32], while others indicated that rectification alters characterization of oscillatory input to muscle, consequently affecting identification of corticomuscular coherence [33, 34].
In [10, 21], estimation of CMC and IMC was performed to study neuro-degeneration of the corticospinal tract in SCA2 patients and prodromal SCA2 gene mutation carriers. Findings revealed a significant CMC and IMC reduction in the beta-band in SCA2 patients and prodromal SCA2 gene mutation carriers in comparison to healthy controls. In the present paper we focus on CMC and IMC estimation, using the same data as in [10, 21] to study the effects of EMG rectification. Also, the previous CMC and IMC analyses are extended to the theta, alpha and gamma frequency bands.
2 Materials and Methods
2.1 Data Description
The study was approved by the institutional ethics committee and conducted according to the declaration of Helsinki. The experimental design and inclusion criteria have been reported elsewhere [10, 21]. Written informed consent was obtained from all subjects prior to participation. Twenty SCA2 patients (mean age (±SD) 45.2 ± 9.8 years, age range 32–64 years) and sixteen prodromal SCA2 gene mutation carriers (mean age (±SD) 41.8 ± 11.8 years; range 26–72 years) were admitted to the Center for Research and Rehabilitation of Hereditary Ataxias in Holguín to participate in this study. Twenty-six healthy non-paid volunteers from Holguin province (mean age (±SD) 43.8 ± 10.4 years, age range 20–66 years) served as age-matched control group.
All subjects performed a motor task as follows: Subjects were seated comfortably in a chair with the examined limbs placed in their natural resting positions for 1 min. Then, repeated simultaneous flexion movements of fingers and wrist, for the upper limb, or dorsal extension movements of the foot, for the lower limb, were executed over a period of 10 min. Onset and offset of each contraction were signaled by a computer-generated tone. The contraction cycles consisted of 3 s contraction and 2 s rest. The force level was kept at 30% of the maximum voluntary force, as determined by using a hand digital dynamometer (Smedley Hand dynamometer, China) for the upper limb. For the lower limb task, the force level was not measured, but previously reported quantitative EMG analysis showed that there was no difference in task-related EMG activity between SCA2 patients, prodromal SCA2 gene mutation carriers and healthy controls [10, 21].
2.2 Signals Recording
EMG and EEG recordings were performed using a Medicid 5 amplifier with Ag/AgCl surface electrodes, electrodes impedances were <5 kΩ throughout the experiment. Bipolar EMG signals were band-pass filtered (2 Hz–100 Hz), amplified (gain, 500–5000) and digitized at 1 kHz. EEG channels were referenced to the left ear lobe, amplified (gain, 1000), band-pass filtered (2 Hz–100 Hz) and digitized at 1 kHz.
EMG was recorded from the first dorsal interosseous (IOD), flexor digitorum superficialis (FDS) and extensor digitorum communis (EDC) muscles of the right upper limb, and from the extensor digitorum brevis (EDB), tibialis anterior (TA) and medial gastrocnemius (MG) muscles of the right lower limb. The EEG was recorded from the sensorimotor cortex of the left hemisphere. For the upper limb, electrode FC3 (placed 30 mm lateral and 20 mm anterior relative to the vertex) and CP3 (30 mm lateral and 20 mm posterior relative to the vertex) according to the International 10/20 system were used to recorded the EEG signal. For the lower limb, EEG electrodes were placed over vertex (Cz) and 20 mm anterior to vertex (Cz´). The electrode position criteria have been reported elsewhere [10, 21].
2.3 Coherence Estimation
The coherence of two signals was defined by Eq. 1
where Sxy is the cross spectral density between the x(t) and y(t) signals and Sxx and Syy are their respective auto-spectral density functions. The coherence value is bounded between 0 and 1 and provides a measure of the correlation of the time series of the two signals in the frequency domain [26]. The corticomuscular coherence was calculated between the EEG data from the FC3 and CP3 electrodes and the EMG data from the IOD, FDS and EDC electrodes for the upper limb, and between the Cz and Cz’ EEG data and the EDB, TA and MG EMG data for the lower limb. Intermuscular coherence was calculated within-limb between the respective three EMG signals from the upper and lower limb. Two coherence estimations were carried out, using the raw EMG signal vs. the rectified EMG.
Data analysis and spectral estimation were performed using the FieldTrip Toolbox [35]. Signals were segmented as 2.6 s trials, defined as the central part of the 3 s contraction periods, in order to avoid unstable EMG during the onset and offset of the contractions [15]. Auto-spectra and cross-spectra were calculated using the multi-taper method based on Hanning tapers with a spectral smoothing of 3 Hz through multi-tapering [36, 37]. Spectral analyses were performed between 5 and 55 Hz, bounded by the EEG interest band and the effects of the line frequency in the coherence at 60 Hz. The spectra were averaged across all trials in a given participant. Statistical significance of differences in CMC and IMC between SCA2 patients vs. healthy controls and prodromal SCA2 gene mutation vs. healthy controls were tested using a non-parametric cluster-based permutation test [38].
3 Results and Discussion
3.1 Corticomuscular Coherence
Previous research [10, 21] reported a CMC increase in the beta-band in healthy controls which is not present in SCA2 patients or prodromal SCA2 gene mutation carriers. This finding has been considered as an early neurodegenerative affection of the corticospinal tract [10, 15, 17]. However, those differences disappear if the rectified EMG is used to estimate the spectral densities and coherence.
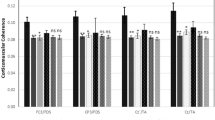
Figure 1 shows the mean CMC values in the beta-band (15–30 Hz for the EEG-EMG channels FC3-FDS, CP3-FDS, Cz’-TA and Cz-TA [10, 21] using the non-rectified raw EMG vs. rectified EMG data. In the case of the rectified EMG the differences were not significant (p > 0.05). The significant differences of CMC to upper and lower limb in SCA2 patients and prodromal SCA2 gene mutation carriers compared to healthy controls were vanish if the rectified EMG is used (see also Figs. 2 and 3).
Upper limb CMC group averages (solid lines) and standard error of the mean (shades areas), left: non-rectified raw EMG, right: rectified EMG, red: SCA2 patients, blue: prodromal SCA2 carriers, black: healthy controls. Pink dashed lines represent the 95% confidence limits of significant CMC. * denote significant differences at single frequencies binned at 0.30 Hz, red: SCA2 patients vs. healthy controls, blue: prodromal SCA2 carriers vs. healthy controls (Color figure online).
Lower limb CMC group averages (solid lines) and standard error of the mean (shades areas), left: non-rectified raw EMG, right: rectified EMG, red: SCA2 patients, blue: prodromal SCA2 carriers, black: healthy controls. Pink dashed lines represent the 95% confidence limits of significant CMC. * denote significant differences at single frequencies binned at 0.30 Hz, red: SCA2 patients vs. healthy controls, blue: prodromal SCA2 carriers vs. healthy controls. (Color figure online)
In other frequency ranges, i.e., the theta (5–7 Hz) and alpha bands (8–12 Hz), and the low gamma band (30–55 Hz), significant differences were not previously reported between SCA2 patients or prodromal SCA2 gene carriers and healthy controls [10, 21]. EMG rectification had no impact on these nil findings.
3.2 Intermuscular Coherence
IMC analysis showed lower values with the rectified EMG data than the non-rectified raw EMG data. Figure 4 shows the mean IMC in the beta-band. Significant differences between SCA2 patients and healthy controls were found for the raw EMG data for the upper limb in EMG channels IOD-FDS, FDS-EDC, and for the lower limb in EMG channels EDB-TA, EDB-GNM. Between prodromal SCA2 gene mutation carriers and healthy controls, there was a significant difference only in the upper limb EMG channels IOD-FDS. Significant differences were found for the same EMG channel combinations for the rectified EMG data, but the p-values were less significant than for the non-rectified raw EMG data (see Table 1). There was an exception for IOD-FDS IMC where the difference was not significant for the rectified data.
Figures 5 and 6 show the mean IMC of the three groups for the EMG channel combinations of the upper and lower limb as indicated above, plotted over the analyzed full frequency range. Qualitatively, rectification of the EMG did not produce any important change in the theta, alpha or gamma band coherence, but resulted in less significant differences compared to the analysis of the non-rectified EMG. In general the results do not support the use of EMG rectification as processing step to identify IMC differences between the studied groups.
Upper limb IMC group averages (solid lines) and standard error of the mean (shades areas), left: non-rectified raw EMG, right: rectified EMG, red: SCA2 patients, blue: prodromal SCA2 carriers, black: healthy controls. Pink dashed lines represent the 95% confidence limits of significant CMC. * denote significant differences at single frequencies binned at 0.30 Hz, red: SCA2 patients vs. healthy controls, blue: prodromal SCA2 carriers vs. healthy controls. (Color figure online)
Lower limb CMC group averages (solid lines) and standard error of the mean (shades areas), left: non-rectified raw EMG, right: rectified EMG, red: SCA2 patients, blue: prodromal SCA2 carriers, black: healthy controls. Pink dashed lines represent the 95% confidence limits of significant CMC. * denote significant differences at single frequencies binned at 0.30 Hz, red: SCA2 patients vs. healthy controls, blue: prodromal SCA2 carriers vs. healthy controls (Color figure online).
4 Discussion
The CMC results suggest that the disappearance of the statistically significant differences in the rectified EMG in the beta-band can largely be attributed to a CMC decrease in the healthy controls which is not present in the SCA2 patients and prodromal SCA2 gene mutations carriers (Fig. 1). The SCA2 patients and prodromal SCA2 gene mutations carriers already had a reduced value of CMC coherence that was not affected by the rectification process, perhaps due to the corticospinal tract damage caused by the disease [7, 14, 15, 39]. However in healthy controls, with an intact corticospinal tract, EMG rectification could have obscured genuine coherence because rectification may have eliminated frequencies that exist in raw signals or introduce power peaks at false frequencies [32, 34].
EMG rectification is a not linear procedure often used to augments the neural information of the EMG. This procedure can introduce power peaks at frequencies not present in the non-rectified raw EMG making possible overestimation of CMC coherence or impose significant CMC at inappropriate frequency bands, but the other way around is not possible [25, 30, 34]. When the rectified EMG is used, it becomes difficult to discern whether a change in CMC values are relates to a genuine physiological change or simply reflects the variable effect of rectification.
In [28, 30] the authors demonstrated that common oscillatory inputs are presents on rectified EMG and non-rectified raw EMG. Also, common oscillatory inputs may be stronger in rectified EMG only at low level of amplitude cancellation [23] and amplitude cancellation changes with contraction level, fatigue, noise level, or across subjects and muscles. Nevertheless non-rectified raw EMG spectrum is less influenced by amplitude cancellation, so the coherence estimation [28, 30] consequently, analysis with the non-rectified raw EMG will be able to detect higher IMC value irrespective of the amplitude cancellation effects.
5 Conclusions
In general, for our data, the rectification process resulted in lower a CMC and IMC value which impairs the significant differences in CMC and IMC between SCA2 patients and prodromal SCA2 gene mutation carriers vs. healthy controls. Based on former arguments and our data reported here, we recommend that future CMC and IMC studies will be carry out on non-rectified raw EMG data.
References
Giunti, P., et al.: The role of the SCA2 trinucleotide repeat expansion in 89 autosomal dominant cerebellar ataxia families. Frequency, clinical and genetic correlates. Brain J. Neurol. 121(Pt 3), 459–467 (1998)
Velázquez-Pérez, L., Rodríguez-Labrada, R., García-Rodríguez, J.C., Almaguer-Mederos, L.E., Cruz-Mariño, T., Laffita-Mesa, J.M.: A comprehensive review of spinocerebellar ataxia type 2 in cuba. Cerebellum 10(2), 184–198 (2011)
Velázquez-Pérez, L., et al.: Saccade velocity is reduced in presymptomatic spinocerebellar ataxia type 2. Clin. Neurophysiol. 120(3), 632–635 (2009)
Velázquez Pérez, L., Rodríguez Labrada, R.: Manifestaciones tempranas de la Ataxia Espinocerebelosa tipo 2. Ediciones Holguin, Cuba (2012)
Rodríguez-Labrada, R., et al.: Saccadic latency is prolonged in spinocerebellar ataxia type 2 and correlates with the frontal-executive dysfunctions. J. Neurol. Sci. 306(1–2), 103–107 (2011)
Rodríguez-Labrada, R., et al.: Subtle rapid eye movement sleep abnormalities in presymptomatic spinocerebellar ataxia type 2 gene carriers. Mov. Disord. 26(2), 347–350 (2011)
Velázquez-Pérez, L., et al.: Abnormal corticospinal tract function and motor cortex excitability in non-ataxic SCA2 mutation carriers: a TMS study. Clin. Neurophysiol. 127(8), 2713–2719 (2016)
Linnemann, C., et al.: Peripheral neuropathy in spinocerebellar ataxia type 1, 2, 3, and 6. Cerebellum 15(2), 165–173 (2016)
Velázquez-Pérez, L., et al.: Sleep disorders in spinocerebellar ataxia type 2 patients. Neurodegener. Dis. 8(6), 447–454 (2011)
Velázquez-Pérez, L., et al.: Early corticospinal tract damage in prodromal SCA2 revealed by EEG-EMG and EMG-EMG coherence. Clin. Neurophysiol. 128(12), 2493–2502 (2017)
Velázquez-Pérez, L., et al.: Comprehensive study of early features in spinocerebellar ataxia 2: delineating the prodromal stage of the disease. Cerebellum 13(5), 568–579 (2014)
Velázquez-Pérez, L., Rodríguez-Labrada, R., García-Rodríguez, J.C., Almaguer-Mederos, L.E., Cruz-Mariño, T., Laffita-Mesa, J.M.: A comprehensive review of spinocerebellar ataxia type 2 in Cuba. Cerebellum Lond. Engl. 10(2), 184–198 (2011)
Velázquez-Pérez, L., et al.: Central motor conduction time as prodromal biomarker in spinocerebellar ataxia type 2. Mov. Disord. Off. J. Mov. Disord. Soc. 31(4), 603–604 (2016)
Velázquez-Pérez, L., et al.: Progression of corticospinal tract dysfunction in pre-ataxic spinocerebellar ataxia type 2: a two-years follow-up TMS study. Clin. Neurophysiol. 129(5), 895–900 (2018)
Fisher, K.M., Zaaimi, B., Williams, T.L., Baker, S.N., Baker, M.R.: Beta-band intermuscular coherence: a novel biomarker of upper motor neuron dysfunction in motor neuron disease. Brain 135(9), 2849–2864 (2012)
Bowyer, S.M.: Coherence a measure of the brain networks: past and present. Neuropsychiatr. Electrophysiol. 2(1), 1 (2016)
Mima, T., Toma, K., Koshy, B., Hallett, M.: Coherence between cortical and muscular activities after subcortical stroke. Stroke 32(11), 2597–2601 (2001)
Baker, M.R., Baker, S.N.: The effect of diazepam on motor cortical oscillations and corticomuscular coherence studied in man. J. Physiol. 546(3), 931–942 (2003)
Caviness, J.N., Shill, H.A., Sabbagh, M.N., Evidente, V.G.H., Hernandez, J.L., Adler, C.H.: Corticomuscular coherence is increased in the small postural tremor of parkinson’s disease: postural tremor in PD. Mov. Disord. 21(4), 492–499 (2006)
Jung, K.-Y., et al.: Increased corticomuscular coherence in idiopathic REM sleep behavior disorder. Front. Neurol. 3, 60 (2012)
Velázquez-Pérez, L., et al.: Corticomuscular coherence: a novel tool to assess the pyramidal tract dysfunction in spinocerebellar ataxia type 2. Cerebellum 16(2), 602–606 (2017)
Farina, D., Merletti, R., Enoka, R.M.: The extraction of neural strategies from the surface EMG. J. Appl. Physiol. 96(4), 1486–1495 (2004)
Farina, D., Merletti, R., Enoka, R.M.: The extraction of neural strategies from the surface EMG: an update. J. Appl. Physiol. 117(11), 1215–1230 (2014)
Myers, L.J., et al.: Rectification and non-linear pre-processing of EMG signals for cortico-muscular analysis. J. Neurosci. Methods 124(2), 157–165 (2003)
McClelland, V.M., Cvetkovic, Z., Mills, K.R.: Inconsistent effects of EMG rectification on coherence analysis. J. Physiol. 592(1), 249–250 (2014)
Rosenberg, J.R., Amjad, A.M., Breeze, P., Brillinger, D.R., Halliday, D.M.: The fourier approach to the identification of functional coupling between neuronal spike trains. Prog. Biophys. Mol. Biol. 53(1), 1–31 (1989)
Random Data: Analysis and Measurement Procedures, 4th edn. Wiley.com. https://www.wiley.com/en-us/Random+Data%3A+Analysis+and+Measurement+Procedures%2C+4th+Edition-p-9780470248775. Accessed 20 November 2018
Negro, F., Keenan, K., Farina, D.: Power spectrum of the rectified EMG: when and why is rectification beneficial for identifying neural connectivity? J. Neural Eng. 12(3), 036008 (2015)
Mima, T., Hallett, M.: Electroencephalographic analysis of cortico-muscular coherence: reference effect, volume conduction and generator mechanism. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 110(11), 1892–1899 (1999)
Farina, D., Negro, F., Jiang, N.: Identification of common synaptic inputs to motor neurons from the rectified electromyogram. J. Physiol. 591(10), 2403–2418 (2013)
Halliday, D.M., Farmer, S.F.: On the need for rectification of surface EMG. J. Neurophysiol. 103(6), 3547 (2010)
Yao, B., Salenius, S., Yue, G.H., Brown, R.W., Liu, J.Z.: Effects of surface EMG rectification on power and coherence analyses: an EEG and MEG study. J. Neurosci. Methods 159(2), 215–223 (2007)
Neto, O.P., Christou, E.A.: Rectification of the EMG signal impairs the identification of oscillatory input to the muscle. J. Neurophysiol. 103(2), 1093–1103 (2010)
McClelland, V.M., Cvetkovic, Z., Mills, K.R.: Rectification of the EMG is an unnecessary and inappropriate step in the calculation of corticomuscular coherence. J. Neurosci. Methods 205(1), 190–201 (2012)
Oostenveld, R., Fries, P., Maris, E., Schoffelen, J.-M.: FieldTrip: open source software for advanced analysis of MEG, EEG, and invasive electrophysiological data. Comput. Intell. Neurosci. 2011, 1–9 (2011)
Thomson, D.J.: Spectrum Estimation and Harmonic Analysis. IEEE Proc. 70, 1055–1096 (1982)
Press, W.H., Teukolsky, S.A., Vetterling, W.T., Flannery, B.P.: Numerical Recipes: The Art of Scientific Computing, 3rd edn. Cambridge University Press, Cambridge (2007)
Maris, E., Oostenveld, R.: Nonparametric statistical testing of EEG- and MEG-data. J. Neurosci. Methods 164(1), 177–190 (2007)
Farmer, S.F., Swash, M., Ingram, D.A., Stephens, J.A.: Changes in motor unit synchronization following central nervous lesions in man. J. Physiol. 463(1), 83–105 (1993)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this paper
Cite this paper
Ruiz-Gonzalez, Y., Velázquez-Pérez, L., Rodríguez-Labrada, R., Torres-Vega, R., Ziemann, U. (2019). Role of EMG Rectification for Corticomuscular and Intermuscular Coherence Estimation of Spinocerebellar Ataxia Type 2 (SCA2). In: Nyström, I., Hernández Heredia, Y., Milián Núñez, V. (eds) Progress in Pattern Recognition, Image Analysis, Computer Vision, and Applications. CIARP 2019. Lecture Notes in Computer Science(), vol 11896. Springer, Cham. https://doi.org/10.1007/978-3-030-33904-3_28
Download citation
DOI: https://doi.org/10.1007/978-3-030-33904-3_28
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-33903-6
Online ISBN: 978-3-030-33904-3
eBook Packages: Computer ScienceComputer Science (R0)