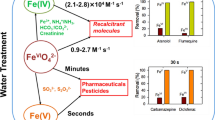
Abstract
Over the past decades, soluble compounds with iron in high oxidation state(s) (so-called ferrates) have turned out to be a highly promising and “green” oxidants deployed in water treatment technologies. Ferrates(IV, V, VI) have good oxidation properties, as indicated by the magnitude of the oxidation-reduction potential (an acidic reaction with an oxidation-reduction potential of 2.20 V, in the basic environment it is 0.72 V), making them interesting for oxidation of inorganic compounds including metals, the decomposition of broad range of organic pollutants including various types of micropollutants found in wastewater (pesticides, pharmaceuticals, and drugs), and for disinfection. The important advantage of ferrates is also their minimal environmental load as the reactions result in the formation of benign iron hydroxides. The only limitation of using ferrates in practice is their demanding production as well as their instability due to the high electron affinity of hexavalent iron.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Barışçı S, Särkkä H, Sillanpää M, Dimoglo A (2016) The treatment of greywater from a restaurant by electrosynthesized ferrate (VI) ion. Desalin Water Treat 57(24):11375–11385. https://doi.org/10.1080/19443994.2015.1038740
Birošová L, Mackul’ak T, Bodík I, Ryba J, Škubák J, Grabic R (2014) Pilot study of seasonal occurrence and distribution of antibiotics and drug resistant bacteria in wastewater treatment plants in Slovakia. Sci Total Environ 490:440–444. https://doi.org/10.1016/j.scitotenv.2014.05.030
Casbeer EM, Sharma VK, Zajickova Z, Dionysiou DD (2013) Kinetics and mechanism of oxidation of tryptophan by ferrate(VI). Environ Sci Technol 47(9):4572–4580. https://doi.org/10.1021/es305283k
Faridmarandi S, Naja GM (2014) Phosphorus and water budgets in an agricultural basin. Environ Sci Technol 48(15):8481–8490. https://doi.org/10.1021/es500738v
Feng MB, Wang XH, Chen J, Qu RJ, Sui YX, Cizmas L, Wang ZY, Sharma VK (2016) Degradation of fluoroquinolone antibiotics by ferrate(VI): effects of water constituents and oxidized products. Water Res 103:48–57. https://doi.org/10.1016/j.watres.2016.07.014
Feng M, Wang Z, Sharma VK (2017) Synergetic effect of the oxidation of fluoroquionolone antibiotics by a combined use of ferrate(VI) and peroxymonosulfate. Paper presented at the 253rd American Chemical Society National Meeting & Exposition, San Francisco, April 2–6, 2017
Filip J, Yngard RA, Siskova K, Marusak Z, Ettler V, Sajdl P, Sharma VK, Zboril R (2011) Mechanisms and efficiency of the simultaneous removal of metals and cyanides by using ferrate(VI): crucial roles of nanocrystalline iron(III) oxyhydroxides and metal carbonates. Chem Eur J 17(36):10097–10105. https://doi.org/10.1002/chem.201100711
Han Q, Dong W, Wang H, Liu T, Tian Y, Song X (2018) Degradation of tetrabromobisphenol A by ferrate(VI) oxidation: performance, inorganic and organic products, pathway and toxicity control. Chemosphere 198:92–102. https://doi.org/10.1016/j.chemosphere.2018.01.117
Hightower SM, Lorenz BB, Bernard JG, Johnson MD (2012) Oxidation of phosphorus centers by ferrate(VI): spectral observation of an intermediate. Inorg Chem 51(12):6626–6632. https://doi.org/10.1021/ic3001812
Híveš J, Benová M, Bouzek K, Sitek J, Sharma VK (2008) The cyclic voltammetric study of ferrate(VI) formation in a molten Na/K hydroxide mixture. Electrochim Acta 54(2):203–208. https://doi.org/10.1016/j.electacta.2008.08.009
Híveš J, Gál M, Kerekeš K (2014) High oxidation state of iron in molten hydroxides. Chem Eng Trans 41:49–54. https://doi.org/10.3303/CET1441009
Híveš J, Gál M, Kerekeš K, Kubiňáková E, Mackuľak T (2016) Electrochemical ferrates(VI) preparation and wastewater treatment. In: Ferrites and ferrates: chemistry and applications in sustainable energy and environmental remediation, ACS Symposium Series, vol 1238. American Chemical Society, Washington, DC, pp 221–240. https://doi.org/10.1021/bk-2016-1238.ch008
Hrnčiariková L, Kerekeš K, Híveš J, Gál M (2013) The influence of anode composition on the electrochemical ferrate (VI) production in molten KOH. Int J Electrochem Sci 8(6):7768–7778
Hu L, Martin HM, Arce-Bulted O, Sugihara MN, Keating KA, Strathmann TJ (2009) Oxidation of carbamazepine by Mn(VII) and Fe(VI): reaction kinetics and mechanism. Environ Sci Technol 43(2):509–515. https://doi.org/10.1021/es8023513
Jain A, Sharma VK, Mbuya OS (2009) Removal of arsenite by Fe(VI), Fe(VI)/Fe(III), and Fe(VI)/Al(III) salts: effect of pH and anions. J Hazard Mater 169(1):339–344. https://doi.org/10.1016/j.jhazmat.2009.03.101
Järup L (2003) Hazards of heavy metal contamination. Br Med Bull 68(1):167–182. https://doi.org/10.1093/bmb/ldg032
Jiang J-Q (2014) Advances in the development and application of ferrate(VI) for water and wastewater treatment. J Chem Technol Biotechnol 89(2):165–177. https://doi.org/10.1002/jctb.4214
Jiang J-Q, Lloyd B (2002) Progress in the development and use of ferrate(VI) salt as an oxidant and coagulant for water and wastewater treatment. Water Res 36(6):1397–1408. https://doi.org/10.1016/S0043-1354(01)00358-X
Jiang J-Q, Panagoulopoulos A, Bauer M, Pearce P (2006) The application of potassium ferrate for sewage treatment. J Environ Manag 79(2):215–220. https://doi.org/10.1016/j.jenvman.2005.06.009
Kamachi T, Kouno T, Yoshizawa K (2005) Participation of multioxidants in the pH dependence of the reactivity of ferrate(VI). J Org Chem 70(11):4380–4388. https://doi.org/10.1021/jo050091o
Kazama F (1995) Viral inactivation by potassium ferrate. Water Sci Technol 31(5–6):165–168. https://doi.org/10.1016/0273-1223(95)00259-P
Kerekeš K, Hrnčiariková L, Híveš J, Gál M (2014) On the mechanism of electrochemical transpassive dissolution of Fe-based anodes in binary hydroxide media. J Electrochem Soc 161(1):C62–C68. https://doi.org/10.1149/2.075401jes
Kolář M, Novák P, Šišková KM, Machala L, Malina O, Tuček J, Sharma VK, Zbořil R (2016) Impact of inorganic buffering ions on the stability of Fe(vi) in aqueous solution: role of the carbonate ion. Phys Chem Chem Phys 18(6):4415–4422. https://doi.org/10.1039/C5CP07543B
Kolařík J, Prucek R, Tuček J, Filip J, Sharma VK, Zbořil R (2018) Impact of inorganic ions and natural organic matter on arsenates removal by ferrate(VI): understanding a complex effect of phosphates ions. Water Res 141:357–365. https://doi.org/10.1016/j.watres.2018.05.024
Kralchevska RP, Prucek R, Kolařík J, Tuček J, Machala L, Filip J, Sharma VK, Zbořil R (2016a) Remarkable efficiency of phosphate removal: ferrate(VI)-induced in situ sorption on core-shell nanoparticles. Water Res 103:83–91. https://doi.org/10.1016/j.watres.2016.07.021
Kralchevska RP, Sharma VK, Machala L, Zboril R (2016b) Ferrates(FeVI, FeV, and FeIV) oxidation of iodide: formation of triiodide. Chemosphere 144:1156–1161. https://doi.org/10.1016/j.chemosphere.2015.09.091
Kubiňáková E, Gál M, Kerekeš K, Híveš J (2015a) Electrochemical preparation of ferrates in NaOH melts. Chem List 109(9):714–717
Kubiňáková E, Kerekeš K, Gál M, Híveš J (2015b) Electrolytic ferrate preparation in various hydroxide molten media. J Appl Electrochem 45(9):1035–1042. https://doi.org/10.1007/s10800-015-0841-0
Kubiňáková E, Híveš J, Gál M, Fašková A (2017) Effect of ferrate on green algae removal. Environ Sci Pollut Res 24(27):21894–21901. https://doi.org/10.1007/s11356-017-9846-z
Kümmerer K (2009) Antibiotics in the aquatic environment – a review – part II. Chemosphere 75(4):435–441. https://doi.org/10.1016/j.chemosphere.2008.12.006
Lan B, Wang Y, Wang X, Zhou X, Kang Y, Li L (2016) Aqueous arsenic (As) and antimony (Sb) removal by potassium ferrate. Chem Eng J 292:389–397. https://doi.org/10.1016/j.cej.2016.02.019
Lee Y, Um I-h, Yoon J (2003) Arsenic(III) oxidation by iron(VI) (ferrate) and subsequent removal of arsenic(V) by iron(III) coagulation. Environ Sci Technol 37(24):5750–5756. https://doi.org/10.1021/es034203+
Lee Y, Zimmermann SG, Kieu AT, von Gunten U (2009) Ferrate (Fe(VI)) application for municipal wastewater treatment: a novel process for simultaneous micropollutant oxidation and phosphate removal. Environ Sci Technol 43(10):3831–3838. https://doi.org/10.1021/es803588k
Lee Y, Kissner R, von Gunten U (2014) Reaction of ferrate(VI) with ABTS and self-decay of ferrate(VI): kinetics and mechanisms. Environ Sci Technol 48(9):5154–5162. https://doi.org/10.1021/es500804g
Lubello C, Gori R (2004) Membrane bio-reactor for advanced textile wastewater treatment and reuse. Water Sci Technol 50(2):113–119. https://doi.org/10.2166/wst.2004.0102
Machala L, Zboril R, Sharma VK, Filip J, Schneeweiss O, Homonnay Z (2007) Mössbauer characterization and in situ monitoring of thermal decomposition of potassium ferrate(VI), K2FeO4 in static air conditions. J Phys Chem B 111(16):4280–4286. https://doi.org/10.1021/jp068272x
Machala L, Zboril R, Sharma VK, Filip J, Jancik D, Homonnay Z (2009) Transformation of solid potassium ferrate(VI) (K2FeO4): mechanism and kinetic effect of air humidity. Eur J Inorg Chem 2009(8):1060–1067. https://doi.org/10.1002/ejic.200801068
Machala L, Filip J, Prucek R, Tucek J, Frydrych J, Sharma VK, Zboril R (2015a) Potassium ferrite (KFeO2): synthesis, decomposition, and application for removal of metals. Sci Adv Mater 7(3):579–587. https://doi.org/10.1166/sam.2015.2143
Machala L, Procházka V, Miglierini M, Sharma VK, Marušák Z, Wille H-C, Zbořil R (2015b) Direct evidence of Fe(v) and Fe(iv) intermediates during reduction of Fe(vi) to Fe(iii): a nuclear forward scattering of synchrotron radiation approach. Phys Chem Chem Phys 17(34):21787–21790. https://doi.org/10.1039/C5CP03784K
Machalová Šišková K, Jančula D, Drahoš B, Machala L, Babica P, Alonso PG, Trávníček Z, Tuček J, Maršálek B, Sharma VK, Zbořil R (2016) High-valent iron (FeVI, FeV, and FeIV) species in water: characterization and oxidative transformation of estrogenic hormones. Phys Chem Chem Phys 18(28):18802–18810. https://doi.org/10.1039/C6CP02216B
Mackul’ak T, Birošová L, Bodík I, Grabic R, Takáčová A, Smolinská M, Hanusová A, Híveš J, Gál M (2016) Zerovalent iron and iron(VI): effective means for the removal of psychoactive pharmaceuticals and illicit drugs from wastewaters. Sci Total Environ 539:420–426. https://doi.org/10.1016/j.scitotenv.2015.08.138
Mácová Z, Bouzek K, Híveš J, Sharma VK, Terryn RJ, Baum JC (2009) Research progress in the electrochemical synthesis of ferrate(VI). Electrochim Acta 54(10):2673–2683. https://doi.org/10.1016/j.electacta.2008.11.034
Mura S, Malfatti L, Greppi G, Innocenzi P (2017) Ferrates for water remediation. Rev Environ Sci Biotechnol 16(1):15–35. https://doi.org/10.1007/s11157-016-9416-8
Noorhasan N, Patel B, Sharma VK (2010) Ferrate(VI) oxidation of glycine and glycylglycine: kinetics and products. Water Res 44(3):927–935. https://doi.org/10.1016/j.watres.2009.10.003
Petrov VG, Perfiliev YD, Dedushenko SK, Kuchinskaya TS, Kalmykov SN (2016) Radionuclide removal from aqueous solutions using potassium ferrate(VI). J Radioanal Nucl Chem 310(1):347–352. https://doi.org/10.1007/s10967-016-4867-5
Prucek R, Tuček J, Kolařík J, Filip J, Marušák Z, Sharma VK, Zbořil R (2013) Ferrate(VI)-induced arsenite and arsenate removal by in situ structural incorporation into magnetic iron(III) oxide nanoparticles. Environ Sci Technol 47(7):3283–3292. https://doi.org/10.1021/es3042719
Prucek R, Tuček J, Kolařík J, Hušková I, Filip J, Varma RS, Sharma VK, Zbořil R (2015) Ferrate(VI)-prompted removal of metals in aqueous media: mechanistic delineation of enhanced efficiency via metal entrenchment in magnetic oxides. Environ Sci Technol 49(4):2319–2327. https://doi.org/10.1021/es5048683
Rai PK, Lee J, Kailasa SK, Kwon EE, Tsang YF, Ok YS, Kim K-H (2018) A critical review of ferrate(VI)-based remediation of soil and groundwater. Environ Res 160:420–448. https://doi.org/10.1016/j.envres.2017.10.016
Sharma VK (2002) Potassium ferrate(VI): an environmentally friendly oxidant. Adv Environ Res 6(2):143–156. https://doi.org/10.1016/S1093-0191(01)00119-8
Sharma VK (2010) Oxidation of nitrogen-containing pollutants by novel ferrate(VI) technology: a review. J Environ Sci Health, Part A: Tox Hazard Subst Environ Eng 45(6):645–667. https://doi.org/10.1080/10934521003648784
Sharma VK (2013) Ferrate(VI) and ferrate(V) oxidation of organic compounds: kinetics and mechanism. Coord Chem Rev 257(2):495–510. https://doi.org/10.1016/j.ccr.2012.04.014
Sharma VK, Mishra SK, Nesnas N (2006) Oxidation of sulfonamide antimicrobials by ferrate(VI) [FeVIO4 2−]. Environ Sci Technol 40(23):7222–7227. https://doi.org/10.1021/es060351z
Sharma VK, Liu F, Tolan S, Sohn M, Kim H, Oturan MA (2013) Oxidation of β-lactam antibiotics by ferrate(VI). Chem Eng J 221:446–451. https://doi.org/10.1016/j.cej.2013.02.024
Sharma VK, Zboril R, Varma RS (2015) Ferrates: greener oxidants with multimodal action in water treatment technologies. Acc Chem Res 48(2):182–191. https://doi.org/10.1021/ar5004219
Sharma VK, Chen L, Zboril R (2016a) Review on high valent FeVI (ferrate): a sustainable green oxidant in organic chemistry and transformation of pharmaceuticals. ACS Sustain Chem Eng 4(1):18–34. https://doi.org/10.1021/acssuschemeng.5b01202
Sharma VK, Tolan S, Bumbálek V, Macova Z, Bouzek K (2016b) Stability of ferrate(VI) in 14 M NaOH-KOH mixtures at different temperatures. In: Ferrites and ferrates: chemistry and applications in sustainable energy and environmental remediation, ACS Symposium Series, vol 1238. American Chemical Society, Washington, DC, pp 241–253. https://doi.org/10.1021/bk-2016-1238.ch009
Sun XH, Zhang Q, Liang H, Ying L, Meng XX, Sharma VK (2016) Ferrate(VI) as a greener oxidant: electrochemical generation and treatment of phenol. J Hazard Mater 319:130–136. https://doi.org/10.1016/j.jhazmat.2015.12.020
Sun S, Liu Y, Ma J, Pang S, Huang Z, Gu J, Gao Y, Xue M, Yuan Y, Jiang J (2018) Transformation of substituted anilines by ferrate(VI): kinetics, pathways, and effect of dissolved organic matter. Chem Eng J 332:245–252. https://doi.org/10.1016/j.cej.2017.08.116
Yngard RA, Sharma VK, Filip J, Zboril R (2008) Ferrate(VI) oxidation of weak-acid dissociable cyanides. Environ Sci Technol 42(8):3005–3010. https://doi.org/10.1021/es0720816
Yu XW, Licht S (2008) Advances in electrochemical Fe(VI) synthesis and analysis. J Appl Electrochem 38(6):731–742. https://doi.org/10.1007/s10800-008-9536-0
Yu H, Chen JW, Xie HB, Ge P, Kong QW, Luo Y (2017) Ferrate(VI) initiated oxidative degradation mechanisms clarified by DFT calculations: a case for sulfamethoxazole. Environ Sci Process Impacts 19(3):370–378. https://doi.org/10.1039/c6em00521g
Zajíček P, Kolář M, Prucek R, Ranc V, Bednář P, Varma RS, Sharma VK, Zbořil R (2015) Oxidative degradation of triazine- and sulfonylurea-based herbicides using Fe(VI): the case study of atrazine and iodosulfuron with kinetics and degradation products. Sep Purif Technol 156:1041–1046. https://doi.org/10.1016/j.seppur.2015.08.024
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Machala, L., Zajíček, P., Kolařík, J., Mackuľak, T., Filip, J. (2020). Ferrates as Powerful Oxidants in Water Treatment Technologies. In: Filip, J., Cajthaml, T., Najmanová, P., Černík, M., Zbořil, R. (eds) Advanced Nano-Bio Technologies for Water and Soil Treatment. Applied Environmental Science and Engineering for a Sustainable Future. Springer, Cham. https://doi.org/10.1007/978-3-030-29840-1_8
Download citation
DOI: https://doi.org/10.1007/978-3-030-29840-1_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-29839-5
Online ISBN: 978-3-030-29840-1
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)