Abstract
In aquaponics, nutrients originate mainly from the fish feed and water inputs in the system. A substantial part of the feed is ingested by the fish and either used for growth and metabolism or excreted as soluble and solid faeces, while the rest of any uneaten feed decays in the tanks. While the soluble excretions are readily available for the plants, the solid faeces need to be mineralised by microorganisms in order for its nutrient content to be available for plant uptake. It is thus more challenging to control the available nutrient concentrations in aquaponics than in hydroponics. Furthermore, many factors, amongst others pH, temperature and light intensity, influence the nutrient availability and plant uptake. Until today, most studies have focused on the nitrogen and phosphorus cycles. However, to ensure good crop yields, it is necessary to provide the plants with sufficient levels of all key nutrients. It is therefore essential to better understand and control nutrient cycles in aquaponics.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Aquaponic systems offer various advantages when it comes to producing food in an innovative and sustainable way. Besides the synergistic effects of increased aerial CO2 concentration for greenhouse crops and decreased total heat energy consumption when cultivating fish and crops in the same space (Körner et al. 2017), aquaponics has two main advantages for nutrient cycling. First, the combination of a recirculating aquaculture system with hydroponic production avoids the discharge of aquaculture effluents enriched in dissolved nitrogen and phosphorus into already polluted groundwater (Buzby and Lin 2014; Guangzhi 2001; van Rijn 2013), and second, it allows for the fertilisation of the soilless crops with what can be considered an organic solution (Goddek et al. 2015; Schneider et al. 2004; Yogev et al. 2016) instead of using fertilisers of mineral origin made from depleting natural resources (Schmautz et al. 2016; Chap. 2). Furthermore, aquaponics yields comparable plant growth as compared with conventional hydroponics despite the lower concentrations of most nutrients in the aquaculture water (Graber and Junge 2009; Bittsanszky et al. 2016; Delaide et al. 2016), and production can be even better than in soil (Rakocy et al. 2004). Increased CO2 concentrations in the aerial environment and changes in the biomes of the root zone are thought to be main reasons for this. In addition, the mineral content and the nutritional quality of tomatoes grown aquaponically have been reported to be equivalent or superior to the mineral content of conventionally grown ones (Schmautz et al. 2016).
Despite having two attractive assets (i.e. the recycling of aquaculture effluents and the use of organic fertilisers), the use of aquaculture effluents increases the challenge of monitoring the nutrients within the solution. Indeed, it is harder to control the composition of a solution where the nutrients originate from a biological degradation of organic matter than to follow the evolution of the nutrients’ concentration in a precisely dosed hydroponic solution based on mineral compounds (Bittsanszky et al. 2016; Timmons and Ebeling 2013). Moreover, a plant’s nutritional needs vary during the growth period in accordance with physiological stages, and it is necessary to meet these needs to maximise yields (Bugbee 2004; Zekki et al. 1996; Chap. 4).
In order to recycle aquaculture effluents to produce plant biomass, it is necessary to optimise the recycling rates of phosphorus and nitrogen (Goddek et al. 2016; Graber and Junge 2009; Chap. 1). Several factors can influence this, such as the fish species, fish density, water temperature, the type of plants and the microbial community (ibid.). Therefore, it is of prime importance to understand the functioning of the nutrient cycles in aquaponics (Seawright et al. 1998). This chapter aims at explaining the origins of the nutrients in an aquaponic system, describing the nutrient cycles and analysing the causes of nutrient losses.
2 Origin of Nutrients
The major sources of nutrients in an aquaponic system are the fish feed and the water added (containing Mg, Ca, S) (see Sect. 9.3.2.) into the system (Delaide et al. 2017; Schmautz et al. 2016) as further elaborated in Chap. 13. With respect to fish feed, there are two main types: fishmeal-based and plant-based feed. Fishmeal is the classic type of feed used in aquaculture where lipids and proteins rely on fish meal and fish oil (Geay et al. 2011). However, for some time now, concerns regarding the sustainability of such feed have been raised and attention drawn towards plant-based diets (Boyd 2015; Davidson et al. 2013; Hua and Bureau 2012; Tacon and Metian 2008). A meta-analysis conducted by Hua and Bureau (2012) revealed that the use of plant proteins in fish feed can influence the growth of fish if incorporated in high proportions. Indeed, plant proteins can have an impact on the digestibility and levels of anti-nutritional factors of the feed. In particular, phosphorus originating from plants and thus in the form of phytates does not benefit, for example, salmon, trout and several other fish species (Timmons and Ebeling 2013). It is not surprising that this observation is highly dependent on the fish species and on the quality of the ingredients (Hua and Bureau 2012). However, little is known of the impact of varying fish feed composition on crop yields (Yildiz et al. 2017).
Classical fish feed is composed of 6–8 macro ingredients and contains 6–8% organic nitrogen, 1.2% organic phosphorus and 40–45% organic carbon (Timmons and Ebeling 2013) with around 25% protein for herbivorous or omnivorous fish and around 55% protein for carnivorous fish (Boyd 2015). Lipids can be fish or plant based as well (Boyd 2015).
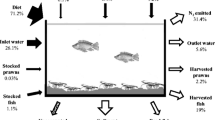
Once fish feed is added into the system, a substantial part of it is eaten by the fish and either used for growth and metabolism or excreted as soluble and solid faeces, while the rest of the given feed decays in the tanks (Goddek et al. 2015; Schneider et al. 2004) (Fig. 9.1). In this case, the feed leftovers and metabolic products are partly dissolved in the aquaponic water, thus enabling the plants to uptake nutrients directly from the aquaponic solution (Schmautz et al. 2016).
Environmental flow of nitrogen and phosphorus in % for (a) Nile tilapia cage production (after Neto and Ostrensky 2015) and (b) RAS production (from a variety of sources)
In most cultivation systems (Chaps. 7 and 8), nutrients can be added to complement the aquaponic solution and ensure a better matching with the plants’ needs (Goddek et al. 2015). Indeed, even when the system is coupled, it is possible to add iron or potassium (which are often lacking) without harming the fish (Schmautz et al. 2016).
2.1 Fish Feed Leftovers and Fish Faeces
Ideally, all the given feed should be consumed by the fish (Fig. 9.1). However, a small part (less than 5% (Yogev et al. 2016)) is often left to decompose in the system and contributes to the nutrient load of the water (Losordo et al. 1998; Roosta and Hamidpour 2013; Schmautz et al. 2016), thus consuming dissolved oxygen and releasing carbon dioxide and ammonia (Losordo et al. 1998), amongst other things. The composition of fish feed leftovers depends on the composition of the feed.
Logically enough, the composition of fish faeces depends on the fish’s diet which also has an impact on the water quality (Buzby and Lin 2014; Goddek et al. 2015). However, the nutrient retention in fish biomass is highly dependent on fish species, feeding levels, feed composition, fish size and system temperature (Schneider et al. 2004). At higher temperatures, for example, fish metabolism is accelerated and thus results in more nutrients contained in the solid fraction of the faeces (Turcios and Papenbrock 2014). The proportion of excreted nutrients also depends on the quality and digestibility of the diet (Buzby and Lin 2014). The digestibility of the fish feed, the size of the faeces and the settling ratio should be carefully considered to ensure a good balance in the system and to maximise crop yields (Yildiz et al. 2017). Indeed, while it is a priority that fish feed should carefully be chosen to suit fish needs, the feed components could also be selected to suit plant’s requirements when it makes no difference to fish (Goddek et al. 2015; Licamele 2009; Seawright et al. 1998).
3 Microbiological Processes
3.1 Solubilisation
Solubilisation consists of the breaking down of the complex organic molecules composing fish waste and feed leftovers into nutrients in the form of ionic minerals which plants can absorb (Goddek et al. 2015; Somerville et al. 2014). In both aquaculture (Sugita et al. 2005; Turcios and Papenbrock 2014) and aquaponics, solubilisation is conducted mainly by heterotrophic bacteria (van Rijn 2013; Chap. 6) which have not yet been fully identified (Goddek et al. 2015). Some studies have started deciphering the complexity of these bacteria communities (Schmautz et al. 2017). In current aquaculture, the most commonly observed bacteria are Rhizobium sp., Flavobacterium sp., Sphingobacterium sp., Comamonas sp., Acinetobacter sp., Aeromonas sp. and Pseudomonas sp. (Munguia-Fragozo et al. 2015; Sugita et al. 2005). An example of the major role of bacteria in aquaponics could be the transformation of insoluble phytates into phosphorus (P) made available for plant uptake through the production of phytases which are particularly present in γ-proteobacteria (Jorquera et al. 2008). (More research needs to be done in this area). Other nutrients than P can also be trapped as solids and evacuated from the system with the sludge. Efforts are thus being made to remineralise this sludge with UASB-EGSB reactors in order to reinject nutrients into the aquaponic system (Delaide 2017; Goddek et al. 2016; Chap. 10). Furthermore, different minerals are not released at the same rate, depending on the composition of the feed (Letelier-Gordo et al. 2015), thus leading to more complicated monitoring of their concentration in the aquaponic solution (Seawright et al. 1998).
3.2 Nitrification
The main nitrogen source in an aquaponic system is the fish feed and the proteins it contains (Goddek et al. 2015; Ru et al. 2017; Wongkiew et al. 2017; Yildiz et al. 2017). Ideally, 100% of this feed should be eaten by the fish. However, is has been observed that fish only use about 30% of the nitrogen contained in the given feed (Rafiee and Saad 2005). The ingested feed is partly used for assimilation and metabolism (Wongkiew et al. 2017), while the rest is excreted either through the gills or as urine and faeces (Ru et al. 2017). The nitrogen which is excreted through the gills is mainly in the form of ammonia, NH3 (Wongkiew et al. 2017; Yildiz et al. 2017), while urine and faeces are composed of organic nitrogen (Wongkiew et al. 2017) which is transformed into ammonia by proteases and deaminases (Sugita et al. 2005). In general, the fish excrete nitrogen under the form of TAN, i.e. NH3 and NH4 +. The balance between NH3 and NH4 + depends mostly on the pH and temperature. Ammonia is the major waste produced by fish catabolism of the feed proteins (Yildiz et al. 2017).
Nitrification is a two-step process during which the ammonia NH3 or ammonium NH4 + excreted by the fish is transformed first into nitrite NO2 − and then into nitrate NO3 − by specific aerobic chemosynthetic autotrophic bacteria. A high availability of dissolved oxygen is required as nitrification consumes oxygen (Carsiotis and Khanna 1989; Madigan and Martinko 2007; Shoda 2014). The first step of this transformation is carried out by ammonia-oxidising bacteria (AOB) such as Nitrosomonas, Nitrosococcus, Nitrosospira, Nitrosolobus and Nitrosovibrio. The second step is conducted by nitrite-oxidising bacteria (NOB) such as Nitrobacter, Nitrococcus, Nitrospira and Nitrospina (Rurangwa and Verdegem 2013; Timmons and Ebeling 2013; Wongkiew et al. 2017). Nitrospira is currently deduced to be a complete nitrifier, i.e. to be involved in the production of both nitrite and nitrate (Daims et al. 2015). The same bacteria can be found both in aquaculture and aquaponic systems (Wongkiew et al. 2017). These bacteria are mainly found in biofilms fixed to the media composing the biofilter but can also be observed in the other compartments of the system (Timmons and Ebeling 2013).
Nitrification is of prime importance in aquaponics as ammonia and nitrite are quite toxic for fish: 0.02–0.07 mg/L of ammonia–nitrogen are sufficient to observe damage in warm water fish, and nitrite–nitrogen should be kept under 1 mg/L (Losordo et al. 1998; Timmons and Ebeling 2013). Ammonia affects the central nervous system of the fish (Randall and Tsui 2002; Timmons and Ebeling 2013), while nitrite induces problems with oxygen fixation (Losordo et al. 1998). Nitrate–nitrogen is, however, tolerated by the fish up to 150–300 mg/L (Goddek et al. 2015; Graber and Junge 2009; Yildiz et al. 2017).
Nitrification mostly takes place in biofilters (Losordo et al. 1998; Timmons and Ebeling 2013). Therefore, when starting a system, it is recommended to run the system without fish in order to allow the slowly growing population of nitrifying bacteria to establish (Timmons and Ebeling 2013; Wongkiew et al. 2017). It is also necessary to avoid, as far as possible, the presence of organic matter in the biofilters in order to prevent the growth of highly competitive heterotrophic bacteria (Timmons and Ebeling 2013). Alternatively, commercial mixes of nitrifying bacteria can be added to the system, prior to stocking, to hasten the colonisation process (Kuhn et al. 2010). Nevertheless, small aquaponic systems without biofilter also exist. In these systems, nitrifying bacteria form biofilms of the available surfaces (e.g. hydroponic compartment walls, inert media when using the media bed technique) (Somerville et al. 2014).
4 Mass Balance: What Happens to Nutrients once They Enter into the Aquaponic System?
4.1 Context
The functioning of aquaponic systems is based on a dynamic equilibrium of the nutrient cycles (Somerville et al. 2014). It is therefore necessary to understand these cycles in order to optimise the management of the systems. Plants growing hydroponically have specific requirements, which should be met during their various growing stages (Resh 2013). Therefore, nutrient concentrations in the different compartments of the system must be closely monitored, and nutrients should be supplemented to prevent deficiencies (Resh 2013; Seawright et al. 1998) either in the system water or via foliar application (Roosta and Hamidpour 2011). According to Delaide et al. (2016), in some cases, supplementing an aquaponic solution with mineral nutrients in order to reach the same nutrient concentrations as in hydroponics could lead to higher yields than those achieved in hydroponics. The first step to take towards a balanced system is the correct design and relative sizing of the compartments (Buzby and Lin 2014). If the hydroponic compartment is too small compared to the fish tanks, then the nutrients will accumulate in the water and could reach toxic levels. The feed rate ratio (i.e. the amount of fish feed in the system based on the plant-growing surface and the plant type) is often used for the first sizing of the system (Rakocy et al. 2006; Somerville et al. 2014). However, according to Seawright et al. (1998), it is not possible to reach a plant/fish ratio which will enable an optimal match of plants’ needs if only fish feed is used as an input. To make sure that the system is well balanced and functions properly, monitoring methods are usually based on the nitrogen cycle (Cerozi and Fitzsimmons 2017; Somerville et al. 2014), but to ensure optimal functioning of the system, it is necessary to monitor more closely the balance of the other macronutrients (N, P, K, Ca, Mg, S) and micronutrients (Fe, Zn, B, Mn, Mo, Cu) (Resh 2013; Somerville et al. 2014; Sonneveld and Voogt 2009) as well. Recent studies (Delaide et al. (2017), Schmautz et al. (2015, 2016)) have started tackling this topic. Schmautz et al. (2015, 2016) compared the impact of three different hydroponic layouts (i.e. nutrient film technique (NFT), floating raft and drip irrigation) on the nutrient uptake of aquaponic tomatoes. Drip irrigation was the system which produced slightly better yields with tomatoes. The mineral content of the fruits (P, K, Ca, Mg) was equivalent to the conventional values even though the iron and zinc contents were higher. The leaves however had lower levels of P, K, S, Ca, Mg, Fe, Cu and Zn than in conventional agriculture. Delaide et al. (2016) followed the cycles of macro- and micronutrients in a coupled aquaponic system. They observed that K, P, Fe, Cu, Zn, Mn and Mo were lacking in their aquaponic solution, while N, Ca, B and Na were quickly accumulated. Graber and Junge (2009) noted that their aquaponic solution contained three times less nitrogen and ten times less phosphorus than a hydroponic solution. As for potassium (K), it was 45 times lower compared to hydroponics. Nevertheless, they obtained yields as similar yields even though the quality was poorer due to a lack of potassium (K).
Factors Influencing the Nutrient Cycles
Light intensity, root zone temperature, air temperature, nutrient availability, growth stage and growth rate all influence a plant’s nutrient uptake (Buzby and Lin 2014). Experiments conducted by Schmautz et al. (2016) and Lennard and Leonard (2006) showed that the hydroponic method could also play a role in a plant’s nutrient uptake capacity, and it is therefore necessary to match the growing system to the type of vegetables being grown. NFT and DWC (deep water culture – raft) are thus suitable for leafy greens, whereas drip irrigation on rockwool slabs is more suitable for fruity vegetables (Resh 2013).
4.2 Macronutrient Cycles
Carbon (C)
Carbon is provided to the fish via the feed (Timmons and Ebeling 2013) and to the plants via CO2 fixation. Fish can use 22% of the carbon contained in the fish feed for biomass increase and metabolism. The rest of the ingested carbon is either expirated under the form of CO2 (52%) or excreted in a dissolved (0.7–3%) and solid (25%) form (Timmons and Ebeling 2013). The expirated CO2 can be used by plants for their own carbon source as well (Körner et al. 2017). The uneaten part of the feed carbon is left to decompose in the system. The type of carbohydrates found in fish feed (e.g. starch or non-starch polysaccharides) can also influence the digestibility of the feed and the biodegradability of the waste in an aquaculture or aquaponic system (Meriac et al. 2014).
Nitrogen (N)
Nitrogen is absorbed by the plants either in the nitrate or ammonium form (Sonneveld and Voogt 2009; Xu et al. 2012) depending on the concentration and plant’s physiology (Fink and Feller 1998 cited by Wongkiew et al. 2017). Associations between plants and microorganisms should not be overlooked as plants affect the presence of the microorganisms in aquaponics, and microorganisms can play a significant role in the nitrogen uptake capacity of plants (Wongkiew et al. 2017). The uptake of nitrogen by plants is affected by the ambient carbon dioxide concentration (Zhang et al. 2008 cited by Wongkiew et al. 2017).
Phosphorus (P)
Phosphorus is one of the essential elements for plant growth and can be absorbed under its ionic orthophosphate form (H2PO4 −, HPO4 2−, PO4 3−) (Prabhu et al. 2007; Resh 2013). Little is known about the dynamics of phosphorus in aquaponics. The main input of phosphorus in the system is the fish feed (Cerozi and Fitzsimmons 2017; Delaide et al. 2017; Schmautz et al. 2015), and in un-supplemented systems (Chap. 7), phosphorus tends to be limiting and thus can impede plant growth (Graber and Junge 2009; Seawright et al. 1998). According to Rafiee and Saad (2005), fish can use up to 15% of the phosphorus contained in the feed. In a system growing lettuce, Cerozi and Fitzsimmons (2017) noticed that the amount of phosphorus provided by the fish feed can be sufficient or insufficient depending on the growth stage. Up to 100% of phosphorus present in the fish water can be recycled in the plant biomass, depending on the design of the system. Graber and Junge (2009) observed a 50% recycling, while Schmautz et al. (2015) reported that 32% of the phosphorus could be found in the fruit and 28% in the leaves. The solubility of phosphorus depends on the pH, and a higher pH will foster the precipitation of phosphorus, thus rendering it unavailable for the plants (Yildiz et al. 2017). Phosphorus can precipitate as struvite (magnesium ammonium phosphate) (Le Corre et al. 2005) and/or hydroxyapatite (Cerozi and Fitzsimmons 2017; Goddek et al. 2015). These insoluble complexes are removed via solid fish sludge from the system. Schneider et al. (2004) reported that 30–65% of the phosphorus contained in the fish feed remains unavailable to plants as it is fixed in the solid excretions which are then removed through mechanical filtration. Yogev et al. (2016) estimated that this loss can be up to 85%. One option to prevent this massive loss of P via solid sludge is to add a digestion compartment to the aquaponic system. During aerobic or anaerobic digestion, the P is released into the digestate and could be re-introduced into the circulating water (Goddek et al. 2016).
Potassium (K)
Delaide et al. (2017) found that the major source of K in their system was the fish feed. Fish can use up to 7% of the K contained in the fish feed (Rafiee and Saad 2005). However, potassium is not necessary for fish which leads to a low potassium composition of the fish feed and to even lower levels of potassium available for the plants (Graber and Junge 2009; Seawright et al. 1998; Suhl et al. 2016). To supply potassium, a KOH pH buffer is often used as the pH often decreases in aquaponics due to nitrification (Graber and Junge 2009). In an aquaponic system planted with tomatoes, potassium accumulated mainly in the fruits (Schmautz et al. 2016).
Magnesium (Mg), Calcium (Ca) and Sulphur (S)
The main source for Mg, Ca and S is tap water which facilitates the absorption by the plants as the nutrients are already available (Delaide et al. 2017). Calcium is however present in insufficient levels in aquaponics (Schmautz et al. 2015; Seawright et al. 1998) and is added under the form of calcium hydroxide Ca(OH)2 (Timmons and Ebeling 2013). According to Rafiee and Saad (2005), fish can use on average 26.8% of the calcium and 20.3% of the magnesium present in the feed. Sulphur is often at low levels in aquaponic systems (Graber and Junge 2009; Seawright et al. 1998).
4.3 Micronutrient Cycles
Iron (Fe), manganese (Mn) and zinc (Zn) derive mainly from the fish feed, while boron (B) and copper (Cu) derive from the tap water (Delaide et al. 2017). In aquaponics, key micronutrients are often present but at too low levels (Delaide et al. 2017), and supplementation from external sources of nutrients is then necessary (Chap. 8). Iron deficiencies occur very often in aquaponics (Schmautz et al. 2015; Seawright et al. 1998; Fitzsimmons and Posadas; 1997 cited by Licamele 2009), mostly because of the non-availability of the ferric ion form. This deficiency can be solved by the use of bacterial siderophore (i.e. organic iron-chelating compounds) produced by genera such as Bacillus or Pseudomonas (Bartelme et al. 2018) or by iron supplementation with chemical chelated iron to avoid iron precipitation.
4.4 Nutrient Losses
Reducing nutrient loss is a constant challenge facing aquaponics practitioners. Nutrient loss occurs in several ways, e.g. the settlement of the sludge (37% of faeces and 18% of uneaten feed) (Neto and Ostrensky 2015), water losses, denitrification, ammonia volatilisation, etc. (Wongkiew et al. 2017). As an example, Rafiee and Saad (2005) note that 24% of the iron, 86% of the manganese, 47% of the zinc, 22% of the copper, 16% of the calcium, 89% of the magnesium, 6% of the nitrogen, 6% of the potassium and 18% of the phosphorus contained in the fish feed were contained in the sludge. The sludge can hold up to 40% of the nutrients present in the feed input (Yogev et al. 2016).
Denitrification can lead to a loss of 25–60% of the nitrogen (Hu et al. 2015; Zou et al. 2016). Denitrification is also linked to anoxic conditions (Madigan and Martinko 2007; van Lier et al. 2008) and low carbon levels and is responsible for the transformation of nitrate into nitrite, nitric oxide (NO), nitrous oxide (N2O) and eventually nitrogen gas (N2) with flows into the atmosphere (ibid.). Denitrification is conducted by several facultative heterotrophic bacteria such as Achromobacter, Aerobacter, Acinetobacter, Bacillus, Brevibacterium, Flavobacterium, Pseudomonas, Proteus and Micrococcus sp. (Gentile et al. 2007; Michaud et al. 2006; Wongkiew et al. 2017). Some bacteria can perform both nitrification and denitrification if dissolved oxygen levels are below 0.3 mg/L (Fitzgerald et al. 2015; Wongkiew et al. 2017). The loss of nitrogen can also occur via anaerobic ammonium oxidation (ANAMMOX), i.e. the oxidation of ammonium into dinitrogen gas in the presence of nitrite (Hu et al. 2011).
Another important loss of nitrogen which should be available for the plants is the consumption of the nitrogen by the heterotrophic aerobic bacteria present in the aquaponic systems. Indeed, the nitrogen used by these bacteria is lost to nitrifying bacteria, and nitrification is thus impeded (Blancheton et al. 2013). These bacteria are particularly present when the C/N ratio increases as they are more competitive and more able to colonise the media than the autotrophic nitrifying bacteria (Blancheton et al. 2013; Wongkiew et al. 2017).
4.5 Nutrient Balance Systems Dynamics
The nutrient concentration of the two major subsystems in an aquaponic system, i.e. the fish tanks (aquaculture) and the hydroponic solution, needs to be balanced for each of their needs. In closed aquaponic systems, nutrients are transported from fish to plants more or less directly through filters (usually a drum filter or a settler and then a biofilter) for nitrification. However, the nutrient needs of crops and the supplied nutrients from the aquaponic subsystem are not in balance. In multi-loop and decoupled systems (Chap. 8), it is easier to provide optimal conditions for both, fish and plant sections. Through modelling of the systems (Chap. 11), the optimal size of the hydroponic area to the fish tanks, biofilters and other equipment can be calculated (Goddek and Körner 2019). This is particularly important with decoupled multi-loop systems which comprise various types of equipment. For example, UASB (upflow anaerobic sludge blanket) (Goddek et al. 2018) or desalination units (Goddek and Keesman 2018) need to be sized carefully as discussed in Chap. 8. The basic mismatch of nutrients supplied by the fish environment and the needs of the crops needs to be rectified and balanced. For the purpose of up-concentrating the nutrients, Goddek and Keesman (2018) have described an appropriate desalination unit (Chap. 8). This approach, however, only solves part of the problem, as the perfectly balanced system is driven by a non-dynamic evaporation rate achievable only in closed chambers and perfectly working plants. The reality, however, is that the evapotranspiration of the crop (ETc) in greenhouse-based aquaponics systems is highly dependent on multiple factors such as physical climate and biological variables. ETc is calculated per area of the ground surface covered by the crop and is calculated for different levels in the canopy (z) by integrating irradiative net fluxes, boundary layer resistance, stomata resistance and the vapour pressure deficit, in the canopy (Körner et al. 2007) using the Penman–Monteith equation. This equation, nevertheless, only calculates the water flux through the crop. Nutrient uptake can either be calculated simply by assuming that all diluted nutrients in the water are taken up by the crop. In reality though, uptake of nutrients is a highly complicated matter. Different nutrients have different states, changing with parameters such as pH. Meanwhile nutrient availability to the plants strongly depends on pH and the relationships of nutrients to each other (e.g. K/Ca availability). In addition, the microbiome in the root zone plays an important role (Orozco-Mosque et al. 2018) which is not yet implemented in models although some models differentiate between phloem and xylem pathways. Thus, the vast amount of nutrients is not modelled in detail for aquaponic nutrient balancing and sizing of systems. The easiest way to estimate nutrient uptake is the assumption that nutrients are taken up/absorbed as dissolved in irrigation water and apply to the above-explained ETc calculation approach and assuming that no element-specific chemical, biological or physical resistances exist. Consequentially, to maintain equilibrium, all nutrients taken up by the crop as contained in the nutrient solution need to be added back to the hydroponic system.
5 Conclusions
5.1 Current Drawbacks of Nutrient Cycling in Aquaponics
In hydroponics, the nutrient solution is accurately determined and the nutrient input into the system is well understood and controlled. This makes it relatively easy to adapt the nutrient solution for each plant species and for each growth stage. In aquaponics, according to the definition (Palm et al. 2018), the nutrients have to originate at least at 50% from uneaten fish feed, fish solid faeces and fish soluble excretions, thus making the monitoring of the nutrient concentrations available for plant uptake more difficult. A second drawback is the loss of nutrients through several pathways such as sludge removal, water renewal or denitrification. Sludge removal induces a loss of nutrients as several key nutrients such as phosphorus often precipitate and are then trapped in the evacuated solid sludge. Water renewal, which has to take place even if in small proportions, also adds to the loss of nutrients from the aquaponic circuit. Finally, denitrification happens because of the presence of denitrifying bacteria and conditions favourable to their metabolisms.
5.2 How to Improve Nutrient Cycling?
To conclude, nutrient cycling still needs to be improved in order to optimise plant growth in aquaponics. Several options are therefore currently explored in Chap. 8. To avoid losing the nutrients captured in the sludge, sludge remineralisation units have been developed (Chap. 10). The aim of these units is to extract the nutrients captured in solid form in the sludge and to reinject these into the system under a form which the plants can absorb (Delaide 2017). A further technique to reduce nutrient loss would be to foster plant uptake through the concentration of the aquaponic solution (i.e. the removal of a fraction of the water to keep the same amount of nutrients but in a lesser water volume). Such a concentration could be achieved via the addition of a desalination unit as part of the aquaponic system (Goddek and Körner 2019; Goddek and Keesman 2018). Finally, the use of decoupled/multi-loop systems enables optimal living and growing conditions for all fish, plants and microorganisms. While some research has been undertaken in this field, more research should be conducted to better understand nutrient cycling in aquaponics. Indeed, more information concerning the exact cycles of each macronutrient (what form, how it can be transformed or not by microorganisms, how it is taken up by plants in aquaponics) or the influence of the plant and fish species and water parameters on the nutrient cycles could greatly help the understanding of aquaponic systems.
References
Bartelme RP, Oyserman BO, Blom JE, Sepulveda-Villet OJ, Newton RJ (2018) Stripping away the soil: plant growth promoting microbiology opportunities in aquaponics. Front Microbiol 9(8). https://doi.org/10.3389/fmicb.2018.00008
Bittsanszky A, Uzinger N, Gyulai G, Mathis A, Junge R, Villarroel M, Kotzen B, Komives T (2016) Nutrient supply of plants in aquaponic systems. Ecocycles 2(1720). https://doi.org/10.19040/ecocycles.v2i2.57
Blancheton JP, Attramadal KJK, Michaud L, d’Orbcastel ER, Vadstein O (2013) Insight into bacterial population in aquaculture systems and its implication. Aquac Eng 53:30–39
Boyd CE (2015) Overview of aquaculture feeds: global impacts of ingredient use. Feed Feed Pract Aquac 3–25. https://doi.org/10.1016/B978-0-08-100506-4.00001-5
Bugbee B (2004) Nutrient management in recirculating hydroponic culture. Acta Hortic 648:99–112. https://doi.org/10.17660/ActaHortic.2004.648.12
Buzby KM, Lin LS (2014) Scaling aquaponic systems: balancing plant uptake with fish output. Aquac Eng 63:39–44. https://doi.org/10.1016/j.aquaeng.2014.09.002
Carsiotis M, Khanna S (1989) Genetic engineering of microbial nitrification. United States Environmental Protection Agency, Risk Reduction Engineering Laboratory, Cincinnati
Cerozi BS, Fitzsimmons K (2017) Phosphorus dynamics modeling and mass balance in an aquaponics system. Agric Syst 153:94–100. https://doi.org/10.1016/j.agsy.2017.01.020
Daims H, Lebedeva EV, Pjevac P, Han P, Herbold C, Albertsen M, Jehmlich N, Palatinszky M, Vierheilig J, Bulaev A, Kirkegaard RH, Von Bergen M, Rattei T, Bendinger B, Nielsen H, Wagner M (2015) Complete nitrification by Nitrospira bacteria. Nature 528:504. https://doi.org/10.1038/nature16461
Davidson J, Good C, Barrows FT, Welsh C, Kenney PB, Summerfelt ST (2013) Comparing the effects of feeding a grain- or a fish meal-based diet on water quality, waste production, and rainbow trout Oncorhynchus mykiss performance within low exchange water recirculating aquaculture systems. Aquac Eng 52:45–57. https://doi.org/10.1016/J.AQUAENG.2012.08.001
Delaide B(2017) A study on the mineral elements available in aquaponics, their impact on lettuce productivity and the potential improvement of their availability. PhD thesis. Gembloux Agro- Bio Tech, University of Liege
Delaide B, Goddek S, Gott J, Soyeurt H, Haissam Jijakli M, Lalman J, Junge R (2016) Lettuce (Lactuca sativa L. var. Sucrine) growth performance in complemented aquaponic solution outperforms hydroponics. Water 8. https://doi.org/10.3390/w8100467
Delaide B, Delhaye G, Dermience M, Gott J, Soyeurt H, Jijakli MH (2017) Plant and fish production performance, nutrient mass balances, energy and water use of the PAFF box, a small-scale aquaponic system. Aquac Eng 78:130–139. https://doi.org/10.1016/j.aquaeng.2017.06.002
Fitzgerald CM, Camejo P, Oshlag JZ, Noguera DR (2015) Ammonia-oxidizing microbial communities in reactors with efficient nitrification at low-dissolved oxygen. Water Res 70:38–51. https://doi.org/10.1016/J.WATRES.2014.11.041
Geay F, Ferraresso S, Zambonino-Infante JL, Bargelloni L, Quentel C, Vandeputte M, Kaushik S, Cahu CL, Mazurais D (2011) Effects of the total replacement of fish-based diet with plant-based diet on the hepatic transcriptome of two European sea bass (Dicentrarchus labrax) half-sibfamilies showing different growth rates with the plant-based diet. BMC Genomics 12:522. https://doi.org/10.1186/1471-2164-12-522
Gentile ME, Lynn Nyman J, Criddle CS (2007) Correlation of patterns of denitrification instability in replicated bioreactor communities with shifts in the relative abundance and the denitrification patterns of specific populations. ISME J 1:714–728. https://doi.org/10.1038/ismej.2007.87
Goddek S, Keesman KJ (2018) The necessity of desalination technology for designing and sizing multi-loop aquaponics systems. Desalination 428:76–85. https://doi.org/10.1016/J.DESAL.2017.11.024
Goddek S, Körner O (2019) A fully integrated simulation model of multi-loop aquaponics: a case study for system sizing in different environments. Agric Syst 171:143
Goddek S, Delaide B, Mankasingh U, Ragnarsdottir KV, Jijakli H, Thorarinsdottir R (2015) Challenges of sustainable and commercial aquaponics. Sustainability 7:4199–4224. https://doi.org/10.3390/su7044199
Goddek S, Schmautz Z, Scott B, Delaide B, Keesman K, Wuertz S, Junge R (2016) The effect of anaerobic and aerobic fish sludge supernatant on hydroponic lettuce. Agronomy 6:37. https://doi.org/10.3390/agronomy6020037
Goddek S, Delaide BPL, Joyce A, Wuertz S, Jijakli MH, Gross A, Eding EH, Bläser I, Reuter M, Keizer LCP et al (2018) Nutrient mineralization and organic matter reduction performance of RAS-based sludge in sequential UASB-EGSB reactors. Aquac Eng 83:10–19
Graber A, Junge R (2009) Aquaponic systems: nutrient recycling from fish wastewater by vegetable production. Desalination 246:147–156. https://doi.org/10.1016/j.desal.2008.03.048
Guangzhi G (2001) Mass balance and water quality in aquaculture tanks. The United Nations University, Fisheries Training Programme, Reyjavik
Hu B, Shen L, Xu X, Zheng P (2011) Anaerobic ammonium oxidation (anammox) in different natural ecosystems. Biochem Soc Trans 39:1811–1816. https://doi.org/10.1042/BST20110711
Hu Z, Lee JW, Chandran K, Kim S, Brotto AC, Khanal SK (2015) Effect of plant species on nitrogen recovery in aquaponics. Bioresources 188:92–98. https://doi.org/10.1016/j.biortech.2015.01.013
Hua K, Bureau DP (2012) Exploring the possibility of quantifying the effects of plant protein ingredients in fish feeds using meta-analysis and nutritional model simulation-based approaches. Aquaculture 356–357:284–301. https://doi.org/10.1016/J.AQUACULTURE.2012.05.003
Jorquera M, Martínez O, Maruyama F, Marschner P, de la Luz Mora M (2008) Current and future biotechnological applications of bacterial Phytases and phytase-producing Bacteria. Microbes Environ 23:182–191. https://doi.org/10.1264/jsme2.23.182
Körner O, Aaslyng JM, Andreassen AU, Holst N (2007) Modelling microclimate for dynamic greenhouse climate control. Hortscience 42:272–279
Körner O, Gutzmann E, Kledal PR (2017) A dynamic model simulating the symbiotic effects in aquaponic systems. Acta Hortic 1170:309–316
Kuhn DD, Drahos DD, Marsh L, Flick GJ (2010) Evaluation of nitrifying bacteria product to improve nitrification efficacy in recirculating aquaculture systems. Aquac Eng 43:78–82. https://doi.org/10.1016/J.AQUAENG.2010.07.001
Le Corre KS, Valsami-Jones E, Hobbs P, Parsons SA (2005) Impact of calcium on struvite crystal size, shape and purity. J Cryst Growth 283:514–522. https://doi.org/10.1016/J.JCRYSGRO.2005.06.012
Lennard WA, Leonard BV (2006) A comparison of three different hydroponic sub-systems (gravel bed, floating and nutrient film technique) in an Aquaponic test system. Aquac Int 14:539–550. https://doi.org/10.1007/s10499-006-9053-2
Letelier-Gordo CO, Dalsgaard J, Suhr KI, Ekmann KS, Pedersen PB (2015) Reducing the dietary protein:energy (P:E) ratio changes solubilization and fermentation of rainbow trout (Oncorhynchus mykiss) faeces. Aquac Eng 66:22–29
Licamele J (2009) Biomass production and nutrient dynamics in an aquaponics system. PhD thesis. Department of Agriculture and biosystems engineering, University of Arizona
Losordo TM, Masser MP, Rakocy J (1998) Recirculating aquaculture tank production systems: an overview of critical considerations. SRAC No. 451, pp 18–31
Madigan MT, Martinko JM (2007) Biologie des micro-organismes, 11th edn. Pearson Education France, Paris
Meriac A, Eding EH, Schrama J, Kamstra A, Verreth JAJ (2014) Dietary carbohydrate composition can change waste production and biofilter load in recirculating aquaculture systems. Aquaculture 420–421:254–261. https://doi.org/10.1016/j.aquaculture.2013.11.018
Michaud L, Blancheton JP, Bruni V, Piedrahita R (2006) Effect of particulate organic carbon on heterotrophic bacterial populations and nitrification efficiency in biological filters. Aquac Eng 34:224–233. https://doi.org/10.1016/j.aquaeng.2005.07.005
Munguia-Fragozo P, Alatorre-Jacome O, Rico-Garcia E, Torres-Pacheco I, Cruz-Hernandez A, Ocampo-Velazquez RV, Garcia-Trejo JF, Guevara-Gonzalez RG (2015) Perspective for aquaponic systems: “omic” technologies for microbial community analysis. Biomed Res Int 2015:1. https://doi.org/10.1155/2015/480386
Neto MR, Ostrensky A (2015) Nutrient load estimation in the waste of Nile tilapia Oreochromis niloticus (L.) reared in cages in tropical climate conditions. Aquac Res 46:1309–1322. https://doi.org/10.1111/are.12280
Orozco-Mosque MC, Rocha-Granados MC, Glick BR, Santoyo G (2018) Microbiome engineering to improve biocontrol and plant growth-promoting mechanism. Microbiol Res. In Press
Palm HW, Knaus U, Appelbaum S, Goddek S, Strauch SM, Vermeulen T, Haїssam Jijakli M, Kotzen B (2018) Towards commercial aquaponics: a review of systems, designs, scales and nomenclature. Aquac Int 26:813–842
Prabhu AS, Fageria NK, Berni RF, Rodrigues FA (2007) Phosphorus and plant disease. In: Datnoff LE, Elmer WH, Huber DM (eds) Mineral nutrition and plant disease. The American Phytopathological Society, St. Paul, pp 45–55
Rafiee G, Saad CR (2005) Nutrient cycle and sludge production during different stages of red tilapia (Oreochromis sp.) growth in a recirculating aquaculture system. Aquaculture 244:109–118. https://doi.org/10.1016/J.AQUACULTURE.2004.10.029
Rakocy JE, Shultz RC, Bailey DS, Thoman ES (2004) Aquaponic production of tilapia and basil: comparing a batch and staggered cropping system. Acta Hortic 648:63–69. https://doi.org/10.17660/ActaHortic.2004.648.8
Rakocy JE, Masser MP, Losordo TM (2006) Recirculating aquaculture tank production systems: aquaponics- integrating fish and plant culture. SRAC Publ South Reg Aquac Cent 16. https://doi.org/454
Randall D, Tsui TK (2002) Ammonia toxicity in fish. Mar Pollut Bull 45:17–23. https://doi.org/10.1016/S0025-326X(02)00227-8
Resh HM (2013) Hydroponic food production: a definitive guidebook for the advanced home gardener and the commercial hydroponic grower, 7th edn. CRC Press, Boca Raton
Roosta HR, Hamidpour M (2011) Effects of foliar application of some macro- and micro-nutrients on tomato plants in aquaponic and hydroponic systems. Sci Hortic 129:396–402. https://doi.org/10.1016/J.SCIENTA.2011.04.006
Roosta HR, Hamidpour M (2013) Mineral nutrient content of tomato plants in Aquaponic and hydroponic systems: effect of foliar application of some macro- and micro-nutrients. J Plant Nutr 36:2070–2083. https://doi.org/10.1080/01904167.2013.821707
Ru D, Liu J, Hu Z, Zou Y, Jiang L, Cheng X, Lv Z (2017) Improvement of aquaponic performance through micro- and macro-nutrient addition. Environ Sci Pollut Res 24:16328–16335. https://doi.org/10.1007/s11356-017-9273-1
Rurangwa E, Verdegem MCJ (2013) Microorganisms in recirculating aquaculture systems and their management. Rev Aquac 7:117–130. https://doi.org/10.1111/raq.12057
Schmautz Z, Graber A, Mathis A, Griessler Bulc T, Junge R (2015) Tomato production in aquaponic system: mass balance and nutrient recycling (abstract)
Schmautz Z, Loeu F, Liebisch F, Graber A, Mathis A, Bulc TG, Junge R (2016) Tomato productivity and quality in aquaponics: comparison of three hydroponic methods. Water 8:1–22. https://doi.org/10.3390/w8110533
Schmautz Z, Graber A, Jaenicke S, Goesmann A, Junge R, Smits THM (2017) Microbial diversity in different compartments of an aquaponics system. Arch Microbiol 199:613. https://doi.org/10.1007/s00203-016-1334-1
Schneider O, Sereti V, Eding EH, Verreth JAJ (2004) Analysis of nutrient flows in integrated intensive aquaculture systems. Aquac Eng 32:379–401. https://doi.org/10.1016/j.aquaeng.2004.09.001
Seawright DE, Stickney RR, Walker RB (1998) Nutrient dynamics in integrated aquaculture–hydroponics systems. Aquaculture 160:215–237
Shoda M (2014) Heterotrophic nitrification and aerobic denitrification by Alcaligenes faecalis. J Biosci Bioeng 117:737–741. https://doi.org/10.5772/68052
Somerville C, Stankus A, Lovatelli A (2014) Small-scale aquaponic food production. Integrated fish and plant farming. Food and Agriculture Organisation of the United Nations, Rome
Sonneveld C, Voogt W (2009) Plant nutrition of greenhouse crops. Springer, Dordrecht/Heidelberg/London/New York
Sugita H, Nakamura H, Shimada T (2005) Microbial communities associated with filter materials in recirculating aquaculture systems of freshwater fish. Aquaculture 243:403. https://doi.org/10.1016/j.aquaculture.2004.09.028
Suhl J, Dannehl D, Kloas W, Baganz D, Jobs S, Scheibe G, Schmidt U (2016) Advanced aquaponics: evaluation of intensive tomato production in aquaponics vs. conventional hydroponics. Agric Water Manag 178:335–344. https://doi.org/10.1016/j.agwat.2016.10.013
Tacon AGJ, Metian M (2008) Global overview on the use of fish meal and fish oil in industrially compounded aquafeeds: trends and future prospects. Aquaculture 285:146–158. https://doi.org/10.1016/j.aquaculture.2008.08.015
Timmons MB, Ebeling JM (2013) Recirculating aquaculture. Ithaca Publishing, New York
Turcios AE, Papenbrock J (2014) Sustainable treatment of aquaculture effluents—what can we learn from the past for the future? Sustainability 6:836–856. https://doi.org/10.3390/su6020836
van Lier JB, Mahmoud N, Zeeman G (2008) In: Henze M, van Loosdrecht MCM, Ekama GA, Brdjanovic D (eds) Anaerobic wastewater treatment, in: biological wastewater treatment: principles, modelling and design. IWA Publishing, London. ISBN: 9781843391883
van Rijn J (2013) Waste treatment in recirculating aquaculture systems. Aquac Eng 53:49–56. https://doi.org/10.1016/J.AQUAENG.2012.11.010
Wongkiew S, Hu Z, Chandran K, Lee JW, Khanal SK (2017) Nitrogen transformations in aquaponic systems: a review. Aquac Eng 76:9–19. https://doi.org/10.1016/j.aquaeng.2017.01.004
Xu G, Fan X, Miller AJ (2012) Plant nitrogen assimilation and use efficiency. Annu Rev Plant Biol 63:153–182. https://doi.org/10.1146/annurev-arplant-042811-105532
Yildiz HY, Robaina L, Pirhonen J, Mente E, Domínguez D, Parisi G (2017) Fish welfare in Aquaponic systems: its relation to water quality with an emphasis on feed and Faeces—a review. Water 9. https://doi.org/10.3390/w9010013
Yogev U, Barnes A, Gross A (2016) Nutrients and energy balance analysis for a conceptual model of a three loops off grid, aquaponics. Water 8. https://doi.org/10.3390/w8120589
Zekki H, Gauthier L, Gosselin A (1996) Growth, productivity, and mineral composition of hydroponically cultivated greenhouse tomatoes, with or without nutrient solution recycling. J Am Soc Hortic Sci 121:1082–1088
Zou Y, Hu Z, Zhang J, Xie H, Guimbaud C, Fang Y (2016) Effects of pH on nitrogen transformations in media-based aquaponics. Bioresour Technol 210:81–87. https://doi.org/10.1016/J.BIORTECH.2015.12.079
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2019 The Author(s)
About this chapter
Cite this chapter
Eck, M., Körner, O., Jijakli, M.H. (2019). Nutrient Cycling in Aquaponics Systems. In: Goddek, S., Joyce, A., Kotzen, B., Burnell, G.M. (eds) Aquaponics Food Production Systems. Springer, Cham. https://doi.org/10.1007/978-3-030-15943-6_9
Download citation
DOI: https://doi.org/10.1007/978-3-030-15943-6_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-15942-9
Online ISBN: 978-3-030-15943-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)