Abstract
Coupled aquaponics is the archetype form of aquaponics. The technical complexity increases with the scale of production and required water treatment, e.g. filtration, UV light for microbial control, automatic controlled feeding, computerization and biosecurity. Upscaling is realized through multiunit systems that allow staggered fish production, parallel cultivation of different plants and application of several hydroponic subsystems. The main task of coupled aquaponics is the purification of aquaculture process water through integration of plants which add economic benefits when selecting suitable species like herbs, medicinal plants or ornamentals. Thus, coupled aquaponics with closed water recirculation systems has a particular role to fulfil.
Under fully closed recirculation of nutrient enriched water, the symbiotic community of fish, plants and bacteria can result in higher yields compared with stand-alone fish production and/or plant cultivation. Fish and plant choices are highly diverse and only limited by water quality parameters, strongly influenced by fish feed, the plant cultivation area and component ratios that are often not ideal. Carps, tilapia and catfish are most commonly used, though more sensitive fish species and crayfish have been applied. Polyponics and additional fertilizers are methods to improve plant quality in the case of growth deficiencies, boosting plant production and increasing total yield.
The main advantages of coupled aquaponics are in the most efficient use of resources such as feed for nutrient input, phosphorous, water and energy as well as in an increase of fish welfare. The multivariate system design approach allows coupled aquaponics to be installed in all geographic regions, from the high latitudes to arid and desert regions, with specific adaptation to the local environmental conditions. This chapter provides an overview of the historical development, general system design, upscaling, saline and brackish water systems, fish and plant choices as well as management issues of coupled aquaponics especially in Europe.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The combination of fish and plant cultivation in coupled aquaponics dates back to the first design by Naegel (1977) in Germany, using a 2000 L hobby scale system (Fig. 7.1) located in a controlled environment greenhouse. This system was developed in order to verify the use of nutrients from fish waste water under fully controlled water recirculating conditions intended for plant production including a dual sludge system (aerobic/anaerobic wastewater treatment). Naegel based his concept on the open pond aquaponic system of the South Carolina Agricultural Experiment Station, in the USA, where excess nutrients from the fishponds, stocked with channel catfish (Ictalurus punctatus), were eliminated by the hydroponic production of water chestnuts (Eleocharis dulcis) (Loyacano and Grosvenor 1973). By including nitrification and denitrification tanks to increase the nitrate concentration inside his system, Naegel (1977) attempted a complete oxidation of all nitrogenous compounds, reaching nitrate concentrations of 1200 mg/L, and demonstrating the effectiveness of the nitrification step. Although the system was stocked at a low density (20 kg/m3 each) using tilapia (Tilapia mossambica) and carp (Cyprinus carpio), the tomatoes (Lycopersicon esculentum) and iceberg lettuce (Lactuca scariola) grew well and produced harvestable yield. These first research results led to the concept of coupled aquaponic systems, in which the plants eliminate the waste produced by the fish, creating adequate growth, demonstrating highly efficient water use in both units. The principle of coupled aquaponics was first described by Huy Tran at the World Aquaculture Conference in 2015 (Tran 2015).
Diagram of the first system by Naegel (1977) growing tilapia and common carp in combination with lettuce and tomatoes in a closed recirculation system
Coupled aquaponic systems do not necessarily use mechanical particulate filtering in the classical sense and keep consistent nutrient flow between the aquaculture and hydroponic units. The main challenge is how to manage the faecal load in the coupled aquaponic system where the plants absorb the nutrients and particulate waste can be removed from the system by filter presses or geotextiles.
The development of modern agriculture, human population growth and shrinking resources worldwide, has promoted the development of coupled aquaponic systems. Since fish farming is considerably more efficient in protein production and water use compared with other farmed animals and since closed systems are largely site-independent, coupled aquaponic systems have been able to develop worldwide (Graber and Junge 2009), under arid conditions (Kotzen and Appelbaum 2010; Appelbaum and Kotzen 2016) and even in urban settings (König et al. 2016). Most described systems belong to domestic, small-scale and semi-commercial installations (Palm et al. 2018) that are driven by hobby aquarists, enthusiasts or smaller start-up companies. New research results, summarized in this chapter, demonstrate both the potentials and constraints regarding the continued development of these systems into commercial aquaponics, being capable of making a significant contribution to future food production.
2 Historical Development of Coupled Aquaponics
Most original research efforts on coupled aquaponic systems took place in the USA with an increasing presence in the EU partly initiated by COST Action FA1305, The EU Aquaponics Hub and in other European research centres. Nowadays, fully recirculating aquaponic system designs almost completely dominate the American aquaponics industry, with estimates that over 90% of the existing aquaponic systems in the USA are of a fully recirculating design (Lennard, pers. comm.). The first American coupled aquaponics research was undertaken at the Illinois Fisheries and Aquaculture Center (formerly the SIU Cooperative Fisheries Research Laboratory) and the Department of Zoology, focusing on coupled aquaponic systems stocked with channel catfish (Ictalurus punctatus) in combination with tomatoes (Lycopersicon esculentum) (Lewis et al. 1978). The authors noted that an optimal plant growth is only possible when all the essential macro- and micronutrients are available in the process water, and thus nutrient supplementation is required in the event of nutrient deficiencies. The authors also demonstrated a deficiency in plant-available iron, constraining plant growth, which could be solved through iron-chelate supplementation. Other early studies in the USA focused on analysing technological functionality and the quality of the harvested channel catfish and tomatoes (Lewis et al. 1978; Sutton and Lewis 1982). Laboratory-scale aquaponic systems examined parameters, such as resource efficiencies with regard to materials, costs, water and energy consumption, and examined the use of other fish species such as Tilapia spp. in the US Virgin Islands (UVI) (Watten and Busch 1984). Dr. James Rakocy at the UVI developed the first commercial coupled aquaponic system, a raft system that combined the production of Nile tilapia (Oreochromis niloticus) and lettuce (Lactuca sativa), and later investigated the production of further plant species (Rakocy 1989, 2012; Rakocy et al. 2000, 2003, 2004, 2006, 2011). This medium scale commercial installation took advantage of the local climate where greenhouses were not necessary and the market conditions of the Virgin Islands to generate profit. The UVI aquaponic system was subsequently adopted in different countries with respect to the respective needs of different plants and the appropriateness of the technology, e.g. in Canada by Savidov (2005) and in Saudi Arabia by Al-Hafedh et al. (2008). This is the case in Europe as well, where coupled aquaponic systems have evolved from the original UVI design, e.g. the vertical aquaponic system at the Aquaponics Research Lab., University of Greenwich (Khandaker and Kotzen 2018). Several other research departments investigated the technological feasibility of closed – or ‘coupled’ – aquaponics production using various fish and plant species as well as hydroponic subsystems to increase yields and reducing different emission parameters (Graber and Junge 2009). For example, at Rostock University (Germany), the research focused on the stability of backyard systems (Palm et al. 2014a), combining different fish species, African catfish (Clarias gariepinus) and Nile tilapia (Oreochromis niloticus), with different plants (Palm et al. 2014b, 2015). In 2015, a modern experimental semi-commercial scale aquaponic system, the ‘FishGlassHouse’, was built on the campus of the University of Rostock (Palm et al. 2016). However, the system was designed allowing both coupled and decoupled operations. Other notable facilities were built at the Zürich University of Applied Sciences (ZHAW) at Waedenswil in Switzerland (Graber and Junge 2009; Graber et al. 2014), both coupled and decoupled research facility of the Icelandic company Svinna-verkfraedi Ltd. (Thorarinsdottir 2014; Thorarinsdottir et al. 2015), the cold water aquaponic system NIBIO Landvik at Grimstad (Skar et al. 2015; Thorarinsdottir et al. 2015), the PAFF Box (Plant And Fish Farming Box) one loop aquaponic system at Gembloux Agro-Bio Tech − University of Liège, in Gembloux, Belgium (Delaide et al. 2017), the combined living wall and vertical farming aquaponic system at the University of Greenwich (Khandaker and Kotzen 2018), as well as the research-domestic coupled aquaponic system (changed from decoupled to coupled in 2018, Morgenstern and Dapprich 2018, pers. comm.) at the South Westphalia University of Applied Sciences, i.GREEN Institute for Green Technology & Rural Development.
3 Coupled Aquaponics: General System Design
The coupled aquaponics principle combines three classes of organisms: (1) aquatic organisms, (2) bacteria and (3) plants that benefit from each other in a closed recirculated water body. The water serves as a medium of nutrient transport, mainly from dissolved fish waste, which is converted into nutrients for plant growth by bacteria. These bacteria (e.g. Nitrosomonas spec., Nitrobacter spec.) oxidize ammonium to nitrite and finally to nitrate. Therefore, it is necessary for the bacteria to receive substantial amounts of ammonium and nitrite to stabilize colony growth and the quantity of nitrate production. Consequently, in a coupled aquaponic system, volumes are critically important, i) the aquaculture unit following the principles of recirculating aquaculture systems (RAS), ii) the bacterial growth substrate and iii) the space for the plant units and the amount of plants to be cultivated. Together, they form the aquaponics unit (Fig. 7.2).
The specific biological-chemical components of the process water have particular importance for coupled aquaponic systems. With food or uneaten feed particles, the organic fish waste and the bacteria inside the process water, an emulsion of nutrients combined with enzymes and digestive bacteria support the growth of fish and plants. There is evidence that compared to stand-alone systems such as aquaculture (fish) and hydroponics (plants), the growth of aquatic organisms and crops in a coupled aquaponics can be similar or even higher. Rakocy (1989) described a slightly higher yield of tilapia (Tilapia nilotica, 46.8 kg) in coupled aquaponics in contrast to stand-alone fish culture (41.6 kg) and slight increases in Summer Bibb lettuce yield (385.1 kg) compared to vegetable hydroponic production (380.1 kg). Knaus et al. (2018b) recorded that aquaponics increased biomass growth of O. basilicum, apparently due to increased leaf generation of the plants (3550 leaves in aquaponics) compared to conventional hydroponics (2393 leaves). Delaide et al. (2016) demonstrated that aquaponic and hydroponic treatments of lettuce exhibited similar plant growth, whereas the shoot weight of the complemented aquaponic solution with nutrients performed best. Similar observations have been made by Goddek and Vermeulen (2018). Lehmonen and Sireeni (2017) observed an increased root weight, leaf area and leaf colour in Batavia salad (Lactuca sativa var. capitata) and iceberg lettuce (L. sativa) with aquaponics process water from C. gariepinus combined with additional fertilizer. Certain plants such as lettuce (Lactuca sativa), cucumbers (Cucumis sativus) or tomatoes (Solanum lycopersicum) can consume nutrients faster, and as a result flower earlier in aquaponics compared with hydroponics (Savidov 2005). Also, Saha et al. (2016) reported a higher plant biomass yield in O. basilicum in combination with crayfish Procambarus spp. and a low start-up fertilization of the aquaponic system.
The basic system design of coupled aquaponics consists of one or more fish tanks, a sedimentation unit or clarifier, substrates for the growth of bacteria or suitable biofilters and a hydroponic unit for plant growth (Fig. 7.3). These units are connected by pipes to form a closed water cycle. Often, after the mechanical filtration and the biofilter, a pump sump is used (one pump or one loop system) which, as the deepest point of the system, pumps the water back to the fish tanks from where it flows by gravity to the hydroponic unit.
Coupled aquaponic systems are used in different scales. The closed-loop principle can be used in domestic systems (mini/hobby/backyard-coupled), demonstration units (e.g. living walls coupled), commercial aquaponics and aquaponics farming (with soil) ranging from small/semi-commercial to large-scale systems (Palm et al. 2018). A recent development in aquaponics has included partial fertilization, which is dependent on the tolerance of the fish species. This, however, can result in a short-term nutrient peak in the system but can be compensated through the nutrient retention by the plants. In coupled aquaponics, an optimal ratio of the production area (or fish volumes) of the aquaculture unit with the resulting feed demand as well as an adequate amount of plants to be cultured in the hydroponic unit (plant production area) must be achieved. (For discussions on the role of evapotranspiration and solar radiation within the systems, see Chaps. 8 and 11). For gravel aquaponics, Rakocy (2012) as a first attempt suggested ‘component ratio principles’, with a fish-rearing volume of 1 m3 of fish tank volume to 2 m3 hydroponic media of 3 to 6 cm pea gravel as a rule of thumb. Ultimately, the amount of fish determines the yield of crops in coupled aquaponics. Additionally, the technical conditions of the fish-rearing unit must be adapted according to the needs of the cultivated aquatic species.
4 Aquaculture Unit
The fish-rearing tanks (size, numbers and design) are selected depending on the scale of production and fish species in use. Rakocy et al. (2006) used four large fish-rearing tanks for the commercial production of O. niloticus in the UVI aquaponic system (USA). With the production of omnivorous or piscivorous fish species, such as C. gariepinus, several tanks should be used due to the sorting of the size classes and staggered production (Palm et al. 2016). Fish tanks should be designed so that the solids that settle at the bottom of the tanks can effectively be removed through an effluent at the bottom. This solid waste removal is the first crucial water treatment step in coupled aquaponics as is the case in aquaculture and decoupled aquaponics. The waste originates from uneaten feed, fish faeces, bacterial biomass and flocculants produced during aquaculture production, increasing BOD and reducing water quality and oxygen availability with respect to both the aquaculture and hydroponic units. In aquaculture, the solid waste consists to a large extent of organic carbon, which is used by heterotrophic bacteria to produce energy through oxygen consumption. The better the solid waste removal, the better the general performance of the system for both fish and plants, i.e. with optimal oxygenation levels and no accumulation of particles in the rhizosphere inhibiting nutrient uptake, and with round or oval tanks proving to be particularly efficient (Knaus et al. 2015).
Fish production in coupled aquaponics in the FishGlassHouse in Germany was tested at different scales in order to ascertain cost effectiveness. This was done effectively as extensive (max. 50 kg, 35 fish m−3) or intensive (max. 200 kg, 140 fish m−3) African catfish production. The semi-intensive production (max. 100 kg, 70 fish m−3) cannot be recommended due to a negative cost benefit balance. In the semi-intensive production mode, system maintenance, labour and feed input were as much as under intensive production but with reduced fish and plant biomass output, and any economic gains in the aquaculture unit did not pay off (Palm et al. 2017). This resulted from the high biochemical oxygen demands (BOD), high denitrification because of the reduced oxygen availability, relatively high water exchange rates, predominantly anaerobic mineralization with distinct precipitation, low P and K-levels as well as a low pH-values with much less fish output compared with the intensive conditions. In contrast, the extensive fish production allowed higher oxygen availability with less water exchange rates and better nutrient availability for plant growth. Thus, under the above conditions, a RAS fish production unit for coupled aquaponics therefore either functions under extensive or intensive fish production conditions, and intermediate conditions should be avoided.
4.1 Filtration
Clarifiers, sometimes also called sedimenters or swirl separators (also see Chap. 3), are the most frequently used devices for the removal of solid waste in coupled aquaponics (Rakocy et al. 2006; Nelson and Pade 2007; Danaher et al. 2013, Fig. 7.4). Larger particulate matters must be removed from the system to avoid anoxic zones with denitrifying effects or the development of H2S. Most clarifiers use lamella or plate inserts to assist in solids removal. Conical bottoms support sludge concentration at the bottom during operation and cleaning, whereas flat bottoms require large quantities of water to flush out and remove the sludge. During operation, the solids sink to the bottom of the clarifier to form sludge. Depending on the feed input and retention time, this sludge can build up to form relatively thick layers. The microbial activity inside the sludge layers gradually shifts towards anaerobic conditions, stimulating microbial denitrification. This process reduces plant available nitrate and should be avoided, especially if the process water is to be used for hydroponic plant production. Consequently, denitrification can be counterproductive in coupled aquaponics.
The density of the solid waste removed by the clarifier is rather low, compared with other technologies, maintenance is time-consuming, and cleaning the clarifier with freshwater is responsible for the main water loss of the entire system. The required amount of water is affected by its general design, the bottom shape and the accessibility of the PVC baffles to flushing water (Fig. 7.4a, b). Increasing fish stocking densities require higher quantities of water exchange (every day in the week under intensive conditions) to maintain optimal water quality for fish production, which can result in the loss of large amounts of process water, also losing substantial amounts of nutrients required for plant growth. Furthermore, replacement with freshwater introduces calcium and magnesium carbonates which may then precipitate with phosphates. Therefore, the use of such manually operated clarifiers makes predictions on process water composition with respect to optimal plant growth nearly impossible (Palm et al. 2019). It would be more effective to follow Naegle’s (1977) example of separating aerobic and anaerobic sludge and gaseous nitrogen discharge with a dual sludge system.
More effective solid waste removal can be achieved by automatic drum- or disk-filters which provide mechanical barriers that hold back solids, which are then removed through rinsing. New developments aim to reduce the use of rinse water through vacuum cleaning technologies, allowing the concentration of total solids in the sludge up to 18% (Dr. Günther Scheibe, PAL-Aquakultur GmbH, Germany, personal communication, Fig. 7.4c). Such effective waste removal has a positive influence on the sludge composition, improving effluent water control in order to better meet the horticultural requirements. Another option is the application of multiple clarifiers (sedimenters) or sludge-removal components in a row.
Biofilters are another essential part of RAS, as they convert ammonia nitrogen via microbial oxidation to nitrate (nitrification). Even though plant roots and the system itself provide surfaces for nitrifying bacteria, the capability to control the water quality is limited. Systems that do not have biofiltration are restricted to mini or hobby installations with low feed inputs. As soon as the biomass of fish and the feed input increases, additional biofilter capacity is required to maintain adequate water quality for fish culture and to provide sufficient nitrate quantities for plant growth. For domestic and small-scale aquaponics, plant media (gravel or expanded clay for example) can suffice as effective biofilters. However, due to the high potential for clogging and thus the requirement for regular manual cleaning and maintenance, these methods are not suitable for larger-scale commercial aquaponics (Palm et al. 2018). Additionally, Knaus and Palm (2017a) demonstrated that the use of a simple biofilter in a bypass already increased the possible daily feed input in a backyard-coupled aquaponic system by approximately 25%. Modern biofilters that are used in intensive RAS are effective in providing sufficient nitrification capacity for fish and plant production. Because of increased investment costs, such components are more applicable in medium- and larger-scale commercial aquaponic systems.
4.1.1 Hydroponics in Coupled Aquaponics
In coupled aquaponics, a wide range of hydroponic subsystems can be used (also see Chap. 4) depending on the scale of operation (Palm et al. 2018). Unless labour has no significant impact on the yield (or profit) and the system is not too large, different hydroponic subsystems can be used at the same time. This is common in domestic and demonstration aquaponics that often use media bed substrate systems (sand, gravel, perlite, etc.) in ebb and flow troughs, DWC channels (deep water culture or raft systems) and even often self-made nutrient film channels (NFT). Most labour-intensive are media substrate beds (sand/gravel) in ebb and flow troughs, which can clog due to the deposition of detritus and often need to be washed (Rakocy et al. 2006). Due to the handling of the substrates, these systems are usually limited in size. On the other hand, DWC hydroponic subsystems require less labour and are less prone to maintenance, allowing them to be adopted for larger planting areas. For this reason, DWC subsystems are mainly found in domestic to small/semi-commercial systems, however, not usually in large-scale aquaponic systems. For larger commercial aquaponic production, the proportion of labour and maintenance in the DWC system is still seen to be too high. Even the use of water resources and energy for pumping are also unfavourable for large-scale systems.
If closed aquaponic systems are designed for profit-oriented production, the use of labour must decrease whilst the production area must increase. This is only possible by streamlining fish production combined with the application of easy-to-use hydroponic subsystems. The nutrient film technique (NFT) can, at present, be considered the most efficient hydroponic system, combining low labour with large plant cultivation areas and a good ratio of water, energy and investment costs. However, not all aquaponic plants grow well in NFT systems and thus it is necessary to find the right plant choice for each hydroponic subsystem, which in turn correlates with the nutrient supply of a specific fish species integrated in a specific hydroponic subsystem design. For coupled aquaponics, the sometimes higher particle load in the water can be problematic by clogging drips, pipes and valves in NFT installations. Hence, large aquaponic systems have to contain professional water management with effective mechanical filtration to avoid recirculation blockages. When the continuous supply of water is ensured through the pipes, the NFT system can be used in all types of coupled aquaponic systems, but is most recommended for production under small/semi-commercial systems and large-scale systems (Palm et al. 2018).
5 Scaling Coupled Aquaponic Systems
Typical coupled aquaponic system range from small to medium scale and larger sized systems (Palm et al. 2018). Upscaling remains one of the future challenges because it requires careful testing of the possible fish and plant combinations. Optimal unit sizes can be repeated to form multiunit systems, independent of the scale of production. According to Palm et al. (2018), the range of aquaponic systems were categorized into (1) mini, (2) hobby, (3) domestic and backyard, (4) small/semi-commercial and (5) large(r)-scale systems, as described below:
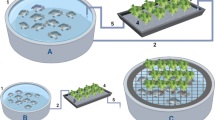
Mini installations (Fig. 7.5) usually consist of a small fish reservoir such as a fish tank or aquarium on which the plants grow on the surface or within a small hydroponic bed. Conventional aquarium filters, aeration and pumps are usually used. Mini systems are usually 2 m2 or less in size (Palm et al. 2018). These small aquaponic systems can be used in the home with only few plants for home consumption and planted with plants such as tomatoes, herbs or ornamentals. Such systems add new values to human living space by adding ‘nature’ back into the family life area which is especially popular in big cities. Some mini systems consist of only a plant vase and one or more fish without filter and pump. However, these systems are only short-term to operate because a regulated filtration is missing.
Principle of a domestic coupled mini aquaponic system (< 2 m2, after Palm et al. 2018) with aeration (a) and a pump (b), the hydroponics (c) act like a biofilter
Hobby aquaponic systems are categorized to reach a maximum size of 10 m2 (Palm et al. 2018). With a higher fish stocking density, more feed and aeration, a mechanical sedimentation unit (sedimenter/clarifier) is necessary (Fig. 7.6). The sedimenter removes particulate matter –‘sludge’ such as faeces and uneaten feed from the system without using energy. The water flows by gravity from the fish tank to the sedimenter and then through the hydroponic tanks and then drops into a sump from where a pump or air lift pumps the water back to the fish tanks. In hobby installations, the plant beds act as a natural microbial filter and often media bed substrates such as sand, fine gravel or perlite are used. Hobby aquaponic systems are more the category of gimmicks that do not target food production. They rather enjoy the functionality of the integrated system. Hobby systems, as the name implies, are usually installed by hobbyists who are interested in growing a variety of aquatic organisms and plants for their own use and for ‘fun’.
Domestic/backyard aquaponics has the purpose of external home use production of fish and plants characterized as having a maximum production area of 50 m2 (Palm et al. 2018). These systems are built by enthusiasts. The construction is technically differentiated with a higher fish production, additional aeration and a higher feed input. The coupled aquaponics principle is applied with the use of one single pump which recirculates the water from a sump (lowest point) to higher standing fish tanks and then by gravity via sedimenter and a biofilter (with aeration and bacteria substrates) to the hydroponic units (Fig. 7.7).
Principle of a coupled domestic backyard aquaponic system, 10–50 m2 (from Palm et al. 2018) with (a) fish tank and aeration, (b) sedimenter or clarifier altered after Nelson and Pade (2007), (c) biofilter with substrates and aeration, (d) hydroponic unit which could consists of combined raft or DWC channels, (e) gravel or sand media substrate system, (f) nutrient film technique NFT-channels and (g) a sump with one pump
For biofiltration, conventional bed filters can also be used as described in Palm et al. (2014a, b, 2015). In backyard aquaponics, hydroponics could consist alone or together of raft or DWC (deep water culture) troughs, substrate subsystems such as coarse gravel/sand ebb and flow boxes or nutrient film technique (NFT) channels. In the northern hemisphere, in outside installations, production is limited to the spring, summer and early autumn periods because of the weather conditions. With this scale of operation, fish and plants can be produced for private consumption (and production can be extended through small greenhouse production), but direct sales in small quantities are also possible.
Small and semi-commercial scale aquaponic systems are characterized by being up to 100 m2 (Palm et al. 2018) with production focused on the retail market. More tanks, often with a higher stocking density, additional filters and water treatment systems and a larger hydroponic area with more diverse designs characterize these systems.
Large(r)-scale commercial operations above 100 m2 (Palm et al. 2018) and reaching many thousands of square metres reach the highest complexity and require careful planning of the water flow and treatment systems (Fig. 7.8). General components are multiple fish tanks, designed as intensive recirculation aquaculture systems (RAS), a water transfer point or a sump allowing water exchange between the fish and plants, and commercial plant production units (aquaponics s.s./s.l.). As fish production is meant for intensive stocking densities, components such as additional filtration with the help of drum filters, oxygen supply, UV light treatments for microbial control, automatic controlled feeding and computerization including automatic water quality control classify these systems.
Schema (supervision) of a large-scale aquaponics module adopted after the FishGlassHouse at University of Rostock (Germany) (1000 m2 total production area, Palm et al. 2018) with (a) independent aquaculture unit, (b) the water transfer system and (c) the independent hydroponic unit; F1-F9 fish tanks, S) sedimenter, P-I pump one (biofilter pump), P-II pump two (aquaculture recirculation pump), T) trickling filter, Su) sump. In the middle, nutrient water transfer system with Wt-I) water transfer tank from the aquaculture unit, P-III) pump three, which pumps the nutrient rich water from aquaculture to C) hydroponics unit on the right with Nu) nutrient tank and an independent hydroponic recirculation system and planting tables (or NFT); P-IV) pump four, which pumps the nutrient low water from the hydroponic unit back to Wt-II) water transfer tank two and to the aquaculture unit for coupled (or decoupled if not used) aquaponic conditions
These systems have a multiunit design capable of upscaling under fully closed water recirculation which also allows for staggered production, parallel cultivation of different plants that require different hydroponic subsystems and better control of the different units in the case of disease outbreak and plant pest control.
6 Saline/Brackish Water Aquaponics
A relatively new field of research is the evaluation of different salinities of the process water for plant growth. Since freshwater worldwide is in continuously increasing demand and at high prices, some attention has been given to the use of saline/brackish water resources for agriculture, aquaculture and also aquaponics. The use of brackish water is significant as many countries such as Israel have underground brackish water resources, and more than half the world’s underground water is saline. Whilst the amount of underground saline water is only estimated as 0.93% of world’s total water resources at 12,870,000 km3, this is more than the underground freshwater reserves (10,530,000 km3), which makes up 30.1% of all freshwater reserves (USGS).
The first published research on the use of brackish water in aquaponics was carried out in 2008–2009 in the Negev Desert of Israel (Kotzen and Appelbaum 2010). The authors studied the potential for brackish water aquaponics that could utilize the estimated 200–300 billion m3 located 550–1000 metres underground in the region. This and additional studies used up to 4708–6800 μS/cm (4000–8000 μS/cm = moderately saline, Kotzen and Appelbaum 2010; Appelbaum and Kotzen 2016) in coupled aquaponic systems with Tilapia sp. (red strain of Nile tilapia Oreochromis niloticus x blue tilapia O. aureus hybrids), combined with deep water culture floating raft and gravel systems. The systems were mirrored with potable water systems as a control. A wide range of herbs and vegetables were grown, with very good and comparative results in both brackish and freshwater systems. In both systems fish health and growth were as good as plant growth of leeks (Allium ampeloprasum), celery (Apium graveolens) (Fig. 7.9), kohlrabi (Brassica oleracea v. gongylodes), cabbage (Brassica oleracea v. capitata), lettuce (Lactuca sativa), cauliflower (Brassica oleracea v. botrytis), Swiss chard (Beta vulgaris vulgaris), spring onion (Allium fistulosum), basil (Ocimum basilicum) and water cress (Nasturtium officinale) (Kotzen and Appelbaum 2010; Appelbaum and Kotzen 2016).
A ‘mission report’ by van der Heijden et al. (2014) on integrating agriculture and aquaculture with brackish water in Egypt suggests that red tilapia (probably red strains of Oreochromis mossambicus) has high potential combined with vegetables such as peas, tomatoes and garlic that can tolerate low to moderate salinity. Plants that are known to have saline tolerance include the cabbage family (Brassicas), such as cabbage (Brassica oleracea), broccoli (Brassica oleracea italica), kale (Brassica oleracea var. sabellica), Beta family, such as Beta vulgaris (beetroot), perpetual spinach (Beta vulgaris subsp. Vulgaris), and bell peppers (Capsicum annuum) and tomatoes (Solanum lycopersicum). An obvious plant candidate for brackish water aquaponics is marsh samphire (Salicornia europaea) and potentially other ‘strand vegetables’ such as sea kale (Crambe maritima), sea aster (Tripolium pannonicum) and sea purslane (Atriplex portulacoides). Gunning (2016) noted that in the most arid regions of the word the cultivation of halophytes as an alternative to conventional crops is gaining significant popularity and Salicornia europea is becoming increasingly popular on the menus of restaurants and the counters of fishmongers and health-food stores across the country. This is similarly the case across the UK and the EU where most of the produce is exported from Israel and now also Egypt. A distinct advantage of growing marsh samphire is that it is a ‘cut and come again’ crop which means it can be harvested at intervals of around 1 month. In its natural environment along saline estuaries Salicornia europaea grows along a saline gradient from saline through brackish (Davy et al. 2001). In trials by Gunning (2016), plants were grown from seed, whereas Kotzen grew his trial plants from cut stems bought at the supermarket fish counter. Further studies under saline conditions were performed by Nozzi et al. (2016), who studied the effects of dinoflagellate (Amyloodinum ocellatum) infection in sea bass (Dicentrarchus labrax) at different salinity levels. Pantanella (2012) studied the growth of the halophyte Salsola soda (salt cabbage) in combination with the flathead grey mullet (Mugil cephalus) under marine conditions of increasing salt contents on an experimental farm at the University of Tuscia (Italy). Marine water resources have also been successful used in coupled aquaponics with the production of European sea bass (Dicentrarchus labrax) and salt-tolerant plants (halophytes) such as Salicornia dolichostachya, Plantago coronopus and Tripolium pannonicum in an inner land marine recirculating aquaculture system (Waller et al. 2015).
7 Fish and Plant Choices
7.1 Fish Production
In larger scale commercial aquaponics fish and plant production need to meet market demands. Fish production allows species variation, according to the respective system design and local markets. Fish choice also depends on their impact onto the system. Problematic coupled aquaponics fish production due to inadequate nutrient concentrations, negatively affecting fish health, can be avoided. If coupled aquaponic systems have balanced fish to plant ratios, toxic nutrients will be absorbed by the plants that are cleaning the water. Since acceptance of toxic substances is species dependent, fish species choice has a decisive influence on the economic success. Therefore, it is important to find the right combination and ratio between the fish and the plants, especially of those fish species with less water polluting activities and plants with high nutrient retention capacity.
The benefits of having a particular fish family in coupled aquaponic systems are not clearly understood with respect to their specific needs in terms of water quality and acceptable nutrient loads. Naegel (1977) found there was no notable negative impact on the fish and fish growth in his use of tilapia (Tilapia mossambica) and common carp (Cyprinus carpio). The channel catfish (Ictalurus punctatus) was also used by Lewis et al. (1978) and Sutton and Lewis (1982) in the USA. It was demonstrated that the quality of the aquaponics water readily met the demands of the different fish species, especially through the use of ‘easy-to-produce’ fish species such as the blue tilapia (Oreochromis aureus, formerly Sarotherodon aurea) in Watten and Busch (1984); Nile tilapia (Oreochromis niloticus), which was often used in studies with different plant species as a model fish species (Rakocy 1989; Rakocy et al. 2003, 2004; Al-Hafedh et al. 2008; Rakocy 2012; Villarroel et al. 2011; Simeonidou et al. 2012; Palm et al. 2014a, 2014b; Diem et al. 2017); and also tilapia hybrids-red strain (Oreochromis niloticus x blue tilapia O. aureus hybrids), that were investigated in arid desert environments (Kotzen and Appelbaum 2010; Appelbaum and Kotzen 2016).
There has been an expansion in the types of fish species used in aquaponics, at least in Europe, which is based on the use of indigenous fish species as well as those that have a higher consumer acceptance. This includes African catfish (Clarias gariepinus) which was grown successfully under coupled aquaponic conditions by Palm et al. (2014b), Knaus and Palm (2017a) and Baßmann et al. (2017) in northern Germany. The advantage of C. gariepinus is a higher acceptance of adverse water parameters such as ammonium and nitrate, as well as there is no need for additional oxygen supply due to their special air breathing physiology. Good growth rates of C. gariepinus under coupled aquaponic conditions were further described in Italy by Pantanella (2012) and in Malaysia by Endut et al. (2009). An expansion of African catfish production under coupled aquaponics can be expected, due to unproblematic production and management, high product quality and increasing market demand in many parts of the world.
In Europe, other fish species with high market potential and economic value have recently become the focus in aquaponic production, with particular emphasis on piscivorous species such as the European pikeperch ‘zander’ (Sander lucioperca). Pikeperch production, a fish species that is relatively sensitive to water parameters, was tested in Romania in coupled aquaponics. Blidariu et al. (2013a, b) showed significantly higher P2O5 (phosphorous pentoxide) and nitrate levels in lettuce (Lactuca sativa) using pikeperch compared to the conventional production, suggesting that the production of pikeperch in coupled aquaponics is possible without negative effects on fish growth by nutrient toxicity. The Cyprinidae (Cypriniformes) such as carp have been commonly used in coupled aquaponics and have generally shown better growth with reduced stocking densities and minimal aquaponic process water flow rates (efficient water use) during experiments in India. The optimal stocking density of koi carp (Cyprinus carpio var. koi) was at 1.4 kg/m (Hussain et al. 2014), and the best weight gain and yield of Beta vulgaris var. bengalensis (spinach) was found with a water flow rate of 1.5 L/min (Hussain et al. 2015). Good fish growth and plant yield of water spinach (Ipomoea aquatica) with a maximum percentage of nutrient removal (NO3-N, PO4-P, and K) was reported at a minimum water flow rate of 0.8 L/min with polycultured koi carp (Cyprinus carpio var. koi) and gold fish (Carassius auratus) by Nuwansi et al. (2016). It is interesting to note that plant growth and nutrient removal in koi (Cyprinus carpio var. koi) and gold fish (Carassius auratus) production (Hussain et al. 2014, 2015) with Beta vulgaris var. bengalensis (spinach) and water spinach (Ipomoea aquatica) increased linearly with a decrease in process water flow between 0.8 L/min and 1.5 L/min. These results suggest that for cyprinid fish culture, lower water flow is recommended as this has no negative impacts on fish growth. In contrast, however, Shete et al. (2016) described a higher flow rate of 500 L h−1 (approx. 8 L/min) for common carp and mint (Mentha arvensis) production, indicating the need for different water flow rates for different plant species. Another cyprinid, the tench (Tinca tinca), was successfully tested by Lobillo et al. (2014) in Spain and showed high fish survival rates (99.32%) at low stocking densities of 0.68 kg m−3 without solids removal devices and good lettuce survival rates (98%). Overall, members of the Cyprinidae family highly contribute to the worldwide aquaculture production (FAO 2017); most likely this would also be true under aquaponic conditions and productivity, but the economic situation should be tested for each country separately.
Other aquatic organisms such as shrimp and crayfish have been introduced into coupled aquaponic production. Mariscal-Lagarda et al. (2012) investigated the influence of white shrimp process water (Litopenaeus vannamei) on the growth of tomatoes (Lycopersicon esculentum) and found good yields in aquaponics with a twofold water sparing effect under integrated production. Another study compared the combined semi-intensive aquaponic production of freshwater prawns (Macrobrachium rosenbergii – the Malaysian shrimp) with basil (Ocimum basilicum) versus traditional hydroponic plant cultivation with a nutrient solution (Ronzón-Ortega et al. 2012). However, basil production in aquaponics was initially less effective (25% survival), but with increasing biomass of the prawns, the plant biomass also increased so that the authors came to a positive conclusion with the production of basil with M. rosenbergii. Sace and Fitzsimmons (2013) reported a better plant growth in lettuce (Lactuca sativa), Chinese cabbage (Brassica rapa pekinensis) and pakchoi (Brassica rapa) with M. rosenbergii in polyculture with the Nile tilapia (O. niloticus). The cultivation with prawns stabilized the system in terms of the chemical-physical parameters, which in turn improved plant growth, although due to an increased pH, nutrient deficiencies occurred in the Chinese cabbage and lettuce. In general, these studies demonstrate that shrimp production under aquaponic conditions is possible and can even exert a stabilizing effect on the closed loop – or coupled aquaponic principle.
7.2 Plant Production
The cultivation of many species of plants, herbs, fruiting crops and leafy vegetables have been described in coupled aquaponics. In many cases, the nutrient content of the aquaponics process water was sufficient for good plant growth. A review by Thorarinsdottir et al. (2015) summarized information on plant production under aquaponic production conditions from various sources. Lettuce (Lactuca sativa) was the main cultivated plant in aquaponics and was often used in different variations such as crisphead lettuce (iceberg), butterhead lettuce (bibb in the USA), romaine lettuce and loose leaf lettuce under lower night (3–12 °C) and higher day temperatures (17–28 °C) (Somerville et al. 2014). Many experiments were carried out with lettuce in aquaponics (e.g. Rakocy 1989) or as a comparison of lettuce growth between aquaponics, hydroponics and complemented aquaponics (Delaide et al. 2016). Romaine lettuce (Lactuca sativa longifolia cv. Jericho) was also investigated by Seawright et al. (1998) with good growth results similar to stand-alone hydroponics and an increasing accumulation of K, Mg, Mn, P, Na and Zn with increasing fish biomass of Nile tilapia (Oreochromis niloticus). Fe and Cu concentrations were not affected. Lettuce yield was insignificant with different stocking densities of fish (151 g, 377 g, 902 g, 1804 g) and plant biomass between 3040 g (151 g fish) and 3780 g (902 g fish). Lettuce was also cultivated, e.g. by Lennard and Leonard (2006) with Murray Cod (Maccullochella peelii peelii), and by Lorena et al. (2008) with the sturgeon ‘bester’ (hybrid of Huso huso female and Acipenser ruthenus male) and by Pantanella (2012) with Nile tilapia (O. niloticus). As a warm water crop, basil (Ocimum basilicum) was reported as a good herb for cultivation under coupled aquaponics and was reported as the most planted crop by 81% of respondents in findings of an international survey (Love et al. 2015). Rakocy et al. (2003) investigated basil with comparable yields under batch and staggered production (2.0; 1.8 kg/m2) in contrast to field cultivation with a comparatively low yield (0.6 kg/m2). Somerville et al. (2014) described basil as one of the most popular herbs for aquaponics, especially in large-scale systems due to its relatively fast growth and good economic value. Different cultivars of basil can be grown under higher temperatures between 20 and 25 °C in media beds, NFT (nutrient film technique) and DWC (deep water culture) hydroponic systems. Basil grown in gravel media beds can reach 2.5-fold higher yield combined with tilapia juveniles (O. niloticus, 0.30 g) in contrast to C. gariepinus (0.12 g) (Knaus and Palm 2017a).
Tomatoes (Lycopersicon esculentum) were described by Somerville et al. (2014) as an ‘excellent summer fruiting vegetable’ in aquaponics and can cope with full sun exposure and temperatures below 40 °C depending on tomato type. However, economic sustainability in coupled aquaponics is disputed due to the reduced competitiveness of aquaponics tomato production compared to high-engineered conventional hydroponic production in greenhouses in, e.g. the Netherlands Improvement Centre of DLV GreenQ in Bleiswijk with tomato yield of 100.6 kg m−2 (Hortidaily 2015), or even higher (Heuvelink 2018). Earlier investigations focused on the cultivation of this plant mostly compared to field production. Lewis et al. (1978) reported nearly double the crop of tomatoes under aquaponics compared to field production and the iron deficiency which occurred was fixed by using ethylene diamine tetra-acetic acid. Tomatoes were also produced in different aquaponic systems over the last decades, by Sutton and Lewis (1982) with good plant yields at water temperatures up to 28 °C combined with Channel catfish (Ictalurus punctatus), by Watten and Busch (1984) combined with tilapia (Sarotherodon aurea) and a calculated total marketable tomato fruit yield of 9.6 kg/m−2, approximately 20% of recorded yields for decoupled aquaponics (47 kg/m2/y, Geelen 2016). McMurtry et al. (1993) combined hybrid tilapia (Oreochromis mossambicus x Oreochromis niloticus) with tomatoes in associated sand biofilters which showed optimal ‘plant yield/high total plant yield’ of 1:1.5 tank/biofilter ratio (sand filter bed) and McMurtry et al. (1997) with increasing total plant fruit yield with increasing biofilter/tank ratio. It must be stated that the production of tomatoes is possible under coupled aquaponics. Following the principle of soilless plant cultivation in aquaponics sensu stricto after Palm et al. (2018), it is advantageous to partially fertilize certain nutrients such as phosphorous, potassium or magnesium to increase yields (see challenges below).
The cultivation of further plant species is also possible and testing of new crops is continuously being reported. In the UK, Kotzen and Khandaker have tested exotic Asian vegetables, with particular success with bitter gourd, otherwise known as kerala or bitter melon (Momordica charantia) (Kotzen pers. comm.). Taro (Colocasia esculenta) is another species which is readily grown with reported success both for its large ‘elephant ear’ like leaves as well as its roots (Kotzen pers. comm.). Somerville et al. (2014) noted that crops such as cauliflower, eggplant, peppers, beans, peas, cabbage, broccoli, Swiss chard and parsley have the potential for cultivation under aquaponics. But there are many more (e.g. celery, broccoli, kohlrabi, chillies, etc.) including plants that prefer to have wet root conditions, including water spinach (Ipomoea aquatica) and mint (Menta sp.) as well as some halophytic plants, such as marsh samphire (Salicornia europaea).
Ornamental plants can also be cultivated, alone or together with other crops (intercropping), e.g. Hedera helix (common ivy) grown at the University of Rostock by Palm & Knaus in a coupled aquaponic system. The trials used 50% less nutrients that would be normally supplied to the plants under normal nursery conditions with a 94.3% success rate (Fig. 7.10).
Besides the chosen plant and variant, there are two major obstacles that concern aquaponics plant production under the two suggested states of fish production, extensive and intensive. Under extensive conditions, nutrient availability inside the process water is much lower than under commercial plant production, nutrients such as K, P and Fe are deficient, and the conductivity is between 1000 and 1500 μS / cm, which is much less than applied under regular hydroponic production of commercial plants regularly between 3000 and 4000 μS / cm. Plants that are deficient in some nutrients can show signs of leaf necroses and have less chlorophyll compared with optimally fertilized plants. Consequently, selective addition of some nutrients increases plant quality that is required to produce competitive products.
In conclusion, commercial plant production of coupled aquaponics under intensive fish production has the difficulty to compete with regular plant production and commercial hydroponics at a large scale. The non-optimal and according to Palm et al. (2019) unpredictable composition of nutrients caused by the fish production process must compete against optimal nutrient conditions found in hydroponic systems. There is no doubt that solutions need to be developed allowing optimal plant growth whilst at the same time providing the water quality required for the fish.
7.3 Fish and Plant Combination Options
Combining fish and plants in closed aquaponics can generate better plant growth (Knaus et al. 2018b) combined with benefits for fish welfare (Baßmann et al. 2017). Inside the process water, large variations in micronutrients and macronutrients may occur with negative effects on plant nutritional needs (Palm et al. 2019). A general analysis of coupled aquaponic systems has shown that there are low nutrient levels within the systems (Bittsanszky et al. 2016) in comparison with hydroponic nutrient solutions (Edaroyati et al. 2017). Plants do not tolerate an under or oversupply of nutrients without effects on growth and quality, and the daily feed input of the aquaponic system needs to be adjusted to the plant’s nutrient needs. This can be achieved by regulating the stocking density of the fish as well as altering the fish feed. Somerville et al. (2014) categorized plants in aquaponics according to their nutrient requirements as follows:
-
1.
Plants with low nutrient requirements (e.g. basil, Ocimum basilicum)
-
2.
Plants with medium nutritional requirements (e.g. cauliflower, Brassica oleracea var. Botrytis)
-
3.
Plants with high nutrient requirements such as fruiting species (e.g. strawberries, Fragaria spec.).
Not all plants can be cultured in all hydroponic subsystems with the same yield. The plant choice depends on the hydroponic subsystem if conventional soilless aquaponic systems (e.g. DWC, NFT, ebb and flow; aquaponics sensu stricto’ – s.s. – in the narrow sense) are used. Under aquaponics farming (‘aquaponics sensu lato’ – s.l. – in a broader sense, Palm et al. 2018), the use of inert soil or with addition of fertilizer applies gardening techniques from horticulture, increasing the possible range of species.
Under hydroponic conditions, the component structures of the subsystems have a decisive influence on plant growth parameters. According to Love et al. (2015), most aquaponic producers used raft and media bed systems and to a smaller amount NFT and vertical towers. Lennard and Leonard (2006) studied the growth of Green oak lettuce (Lactuca sativa) and recorded the relationship Gravel bed > Floating raft > NFT in terms of biomass development and yield in combination with the Murray Cod (Maccullochella peelii peelii) in Australia. Knaus & Palm (2016–2017, unpublished data) have tested different hydroponic subsystems such as NFT, floating raft and gravel substrate on the growth of different plants in the FishGlassHouse in a decoupled aquaponic experimental design, requiring subsequent testing under coupled conditions. With increasing production density of African catfish (C. gariepinus, approx. 20–168 kg/m3), most of the cultured crops such as cucumbers (Cucumis sativus), basil (Ocimum basilicum) and pak choi (Brassica rapa chinensis) tended to grow better, in contrast to Lennard and Leonard (2006), in gravel and NFT aquaponics (GRAVEL > NFT > RAFT; Wermter 2016; Pribbernow 2016; Lorenzen 2017), and Moroccan mint ‘spearmint’ (Mentha spicata) showed the opposite growth performance (RAFT = NFT > GRAVEL) with highest leaf numbers in NFT (Zimmermann 2017). This demonstrates an advantage of gravel conditions and can be used figuratively also in conventional plant pots with soil substrate under coupled aquaponic conditions. This type of aquaponics was designated as ‘horticulture – aquaponics (s.l.)’ due to the use of substrates from the horticultural sector (soil, coco fibre, peat, etc.) (see Palm et al. 2018). This involves all plant cultivation techniques that allow plants to grow in pots, whereby the substrate in the pot itself may be considered equivalent to a classical gravel substrate for aquaponics. Research by Knaus & Palm (unpublished data) showed variance in the quality of commonly grown vegetables and thus their suitability for growing in this type of aquaponics with soil (Fig. 7.11, Table 7.1). In this type of aquaponics, beans, lambs lettuce and radish did well.
The plant choice (species and strain) and especially the hydroponic subsystem and/or substrate, including peat, peat substitutes, coco fibre, composts, clay, etc. or a mix of them (see Somerville et al. 2014), has a significant impact on the economic success of the venture. The efficiency of some substrates must be tested in media bed hydroponic sub-units (e.g. the use of sand (McMurtry et al. 1990, 1997), gravel (Lennard and Leonard 2004) and perlite (Tyson et al. 2008). The use of other media bed substrates such as volcanic gravels or rock (tuff/tufa), limestone gravel, river bed gravel, pumice stone, recycled plastics, organic substrates such as coconut fibre, sawdust, peat moss and rice trunk have been described by Somerville et al. (2014). Qualitative comparative studies with recommendations, however, are very rare and subject of future research.
7.4 Polyponics
The combination of different aquatic organisms in a single aquaponic system can increase total yields. First applied by Naegel (1977), this multispecies production principle was coined from the term polyculture combined with aquaponics in coupled systems as ‘polyponic’ (polyculture + aquaponics) by Knaus and Palm (2017b). Like IMTA (integrated multitrophic aquaculture), polyponics expands the diversity of the production systems. Using multiple species in one system has both advantages and disadvantages as (a) diversification allows the producer to respond to local market demands but (b) on the other hand, focus is spread across a number of products, which requires greater skill and better management. Published information on polyponics is scarce. However, Sace and Fitzsimmons (2013) reported better plant growth of lettuce, Chinese cabbage and pakchoi in polyculture with freshwater shrimp (Macrobrachium rosenbergii) and Nile tilapia (O. niloticus) in coupled aquaponics. Alberts-Hubatsch et al. (2017) described the cultivation of noble crayfish (Astacus astacus), hybrid striped bass (Morone saxatilis x M. chrysops), microalgae (Nannochloropsis limnetica) and watercress (Nasturtium officinale), where crayfish growth was higher than expected, feeding on watercress roots, fish faeces and a pikeperch-designed diet.
Initial investigations at the University of Rostock showed differences in plant growth in two identical 25m2 backyard-coupled aquaponic units with the production of African catfish (Clarias gariepinus) and Nile tilapia (Oreochromis niloticus, Palm et al. 2014b). The plant yields of lettuce (Lactuca sativa) and cucumber fruits (Cucumis sativus) were significantly better in combination with O. niloticus. This effect was also seen by Knaus and Palm (2017a) with a 2.5-fold higher yield in basil (Ocimum basilicum) and two times more biomass of parsley (Petroselinum crispum) combined with O. niloticus. Another comparison between O. niloticus and common carp (Cyprinus carpio) showed a twofold higher gross biomass per plant (g plant−1) of tomatoes (Solanum lycopersicum) with tilapia and a slightly increased gross biomass of cucumbers (Cucumis sativus) with carp, however, with higher cucumber fruit weight in the O. niloticus aquaponic unit (Knaus and Palm 2017b). The yield of mint (Mentha x piperita) was approximately 1.8 times higher in the tilapia unit, but parsley was 2.4 times higher combined with the carp (Knaus et al. 2018a). The results of these experiments followed the order of plant growth: O. niloticus > C. carpio > C. gariepinus, whilst fish growth showed a reverse order with: C. gariepinus > O. niloticus > C. carpio.
According to these results, the fish choice influences the plant yield and a combination of different fish species and their respective growth performance allows adjustment of a coupled aquaponics to optimal fish and plant yields. During consecutive experiments (O. niloticus only, C. gariepinus only), a higher basil (O. basilicum) biomass yield of 20.44% (Plant Growth Difference – PGD) was observed for O. niloticus in contrast to the basil yield with C. gariepinus (Knaus et al. 2018b). Thus, O. niloticus can be used to increase the plant yield in a general C. gariepinus system. This so-called boost effect by tilapia enhances the overall system production output and compensates i) poorer plant growth with high fish growth of C. gariepinus as well as ii) poorer fish growth in O. niloticus with a boost to the plant yield. A first commercial polyponic farm has opened in Bali, Indonesia, producing tilapia combined with Asian catfish (Clarias batrachus) and conventional farm products.
8 System Planning and Management Issues
Coupled aquaponics depends on the nutrients that are provided from the fish units, either a commercial intensive RAS or tanks stocked under extensive conditions in smaller operations. The fish density in the latter is often about 15–20 kg/m3 (tilapia, carp), but extensive African catfish production can be higher up to 50 kg/m3. Such different stocking densities have a significant influence on nutrient fluxes and nutrient availability for the plants, the requirement of water quality control and adjustment as well as appropriate management practices.
The process water quality with respect to nutrient concentrations is primarily dependent on the composition of the feed and the respective turnover rates of the fish. The difference between feed input and feed nutrients, assimilating inside the fish or lost through maintenance of the system, equals the maximum potential of plant available nutrients from aquaculture. As noted above, the nutrient concentrations should be adjusted to levels, which allow the plants to grow effectively. However, not all fish species are able to withstand such conditions. Consequently, resilient fish species such as the African catfish, tilapia or carp are preferred aquaponic candidates. At the University of Rostock, whole catfish and its standard diet as output and input values were analysed to identify the turnover rates of the macronutrients N, P, K, Ca, Mg and S and the micronutrients Fe, Mn, Mo, Cu, Zn and Se. With the exception of P, more than 50% of the feed nutrients given to the fish are not retained in its body and can be considered potentially available as plant nutrients (Strauch et al. 2018; Fig. 7.12). However, these nutrients are not equally distributed inside the process water and the sediments. Especially macronutrients (N, P, K) accumulate in the process water as well as inside the solid fraction whilst the micronutrients, such as iron, disappear in the solid fraction separated by the clarifier. Figure 7.13 shows the nutrient output per clarifier cleaning after 6 days of sludge collection in an intensive African catfish RAS. The proportions of plant essential nutrients that are bound in the solids relative to the respective amounts that appear dissolved are significant: N = 48%, P = 61%, K = 10%, Ca = 48%, Mg = 16%, S = 11%, Fe = 99%, Mn = 86%, Mo = 100%, Zn = 48% and Cu = 55%.
Distribution of macro- and micronutrients inside the process water and the solids. (Data from Strauch et al. (2018))
One key management factor is the availability of oxygen inside the system, which is crucial to keep the concentration of plant available nitrate in the process water high. Conventional clarifiers that are applied in many RAS remove carbon-rich solid wastes from the recirculation but will leave them in contact with the process water until the next cleaning interval of the sedimentation tank. During this time, the carbon-rich organic matter is utilized as a source of energy by denitrifying bacteria, accounting for significant losses of nitrate. It outgasses as nitrogen into the atmosphere and is lost. Under intensive production conditions, large quantities of organic sludge will accumulate inside the sedimentation tanks, with consequences for maintenance, replacement with freshwater and subsequently for the nutrient composition inside the process water. Figure 7.14 illustrates the nutrient concentrations in the holding tanks of African catfish RAS under three different stocking densities (extensive: 35 fish / tank, semi-intensive: 70 fish / tank, intensive: 140 fish/tank). The higher the stocking density and the lower the resulting oxygen content inside the system, the lower is the plant available nitrate per kg feed inside the system.
In general, with increasing fish intensity, the availability of oxygen inside the system decreases because of the consumption of the oxygen by the fish and aerobic sludge digestion inside the clarifier and the hydroponic subsystems. Oxygen levels can be maintained at higher levels, but this requires additional investment for oxygen monitoring and control. This issue is of tremendous importance for coupled aquaponics, right from the beginning of the planning phase of the systems because the different scenarios are decisive for the planned fish production, the resulting quality of the process water for the plant production units, and consequently for economic sustainability. Four principals of coupled aquaponic production systems with management consequences in terms of system design, maintenance procedures and nutrient availability for plant growth, with transitions between them, can be defined as follows:
-
Extensive production, oxygen resilient fish (e.g. tilapia, carp), no oxygen control, O2 above 6 mg/L, little water use with high nutrient concentrations, small investment, low BOD, high nitrate per kg feed.
-
Intensive production, oxygen resilient fish (e.g. African catfish), no oxygen control, O2 below 6 mg/L, high water use, medium investment, high BOD, low nitrate per kg feed, high nutrient concentrations.
-
Extensive production, oxygen demanding fish (e.g. Trout), oxygen control, O2 above 6–8 mg/L, high water use, medium investment, low BOD, high nitrate per kg feed, low nutrient concentrations.
-
Intensive production, oxygen demanding fish (e.g. Trout, pikeperch), oxygen control, O2 above 6–8 mg/L, high water use, high investment, low BOD, medium nitrate per kg feed.
In addition to the stocking density and the average amount of oxygen inside the system, the plant production regime, i.e. batch or staggered cultivation, has consequences for the plant available nutrients inside the process water (Palm et al. 2019). This is the case especially with fast growing fish, where the feed increase during the production cycle can be so rapid that there needs to be a higher water exchange rate and thus nutrient dilution can increase, with consequences for the nutrient composition and management.
The same oxic or anoxic processes that occur in the RAS as a part of the coupled aquaponic system also occur inside the hydroponic subsystems. Therefore, oxygen availability and possibly aeration of the plant water can be crucial in order to optimize the water quality for good plant growth. The oxygen allows the heterotrophic bacteria to convert organic bound nutrients to the dissolved phase (i.e. protein nitrogen into ammonia) and the nitrifying bacteria to convert the ammonia into nitrate. The availability of oxygen in the water also reduces anoxic microbial metabolism (i.e. nitrate- and/or sulphate-reducing bacteria, Comeau 2008), processes which can have tremendous effects on the reduction of nutrient concentrations. The aeration of the roots also has the advantage that water and nutrients are transported to the root surface, and that particles that settle on the root surface are removed (Somerville et al. 2014).
9 Some Advantages and Disadvantages of Coupled Aquaponics
The following discussion reveals a number of key pros and challenges of coupled aquaponics as follows:
-
Pro: Coupled aquaponic systems have many food production benefits, especially saving resources under different production scales and over a wide range of geographical regions. The main purpose of this production principle is the most efficient and sustainable use of scarce resources such as feed, water, phosphorous as a limited plant nutrient and energy. Whilst, aquaculture and hydroponics (as stand-alone), in comparison to aquaponics are more competitive, coupled aquaponics may have the edge in terms of sustainability and thus a justification of these systems especially when seen in the context of, for example, climate change, diminishing resources, scenarios that might change our vision of sustainable agriculture in future.
-
Pro: Small-scale and backyard-coupled aquaponics are meant to support local and community-based food production by households and farmers. They are not able to stem high investment costs and require simple and efficient technologies. This applies for tested fish and plant combinations in coupled aquaponics.
-
Pro: The plants in contemporary coupled aquaponics have the similar role in treating waste as constructed wetlands do in the removal of waste from water (Fig. 7.15). The plants in the hydroponic unit in coupled aquaponics therefore fulfil the task of purifying the water and can be considered a ‘biological advanced unit of water purification’ in order to reduce the environmental impact of aquaculture.
-
Challenge: It has been widely accepted that using only fish feed as the input for plant nutrition is often qualitatively and quantitatively insufficient in comparison to conventional agriculture production systems (e.g. N-P-K hydroponics manure) (Goddek et al. 2016), limiting the growth of certain crops in coupled aquaponics.
-
Pro: Coupled aquaponic systems have a positive influence on fish welfare. Most recent studies demonstrate that in combination with cucumber and basil, the agonistic behaviour of African catfish (C. gariepinus) was reduced (Baßmann et al. 2017, 2018). More importantly, comparing injuries and behavioural patterns with the control, aquaponics with high basil density influenced African catfish even more positively. Plants release substances into the process water like phosphatases (Tarafdar and Claassen 1988; Tarafdar et al. 2001) that are able to hydrolyse biochemical phosphate compounds around the root area and exude organic acids (Bais et al. 2004). Additionally, microorganisms on the root surfaces play an important role through the excretion of organic substances increasing the solubilization of minerals making them available for plant nutrition. It is evident that the environment of the rhizosphere, the ‘root exudate’, consists of many organic compounds such as organic acid anions, phytosiderophores, sugars, vitamins, amino acids, purines, nucleosides, inorganic ions, gaseous molecules, enzymes and root border cells (Dakora and Phillips 2002), which may influence the health of aquatic organisms in coupled aquaponic systems. This symbiotic relationship is not available in either pure aquaculture or decoupled aquaponics. However, considerable research still needs to be undertaken to understand the responsible factors for better fish welfare.
-
Pro: Aquaponics can be considered as an optimized form of the conventional agricultural production especially in those areas where production factors caused by the environmental conditions are particularly challenging, e.g. in deserts or highly populated urban areas (cities). Coupled aquaponic systems can be easily adjusted to the local conditions, in terms of system design and scale of operation.
-
Challenge: Coupled aquaponic also show disadvantages, due to often unsuitable component ratio conditions of the fish and plant production. In order to avoid consequences for fish welfare, coupled aquaponic systems must balance the feed input, stocking density as well as size of the water treatment units and hydroponics. So far knowledge of component ratios in coupled aquaponics is still limited, and modelling to overcome this problem is at the beginning. Rakocy (2012) suggested 57 g of feed/day per square meter of lettuce growing area and a composite ratio of 1 m3 of fish-rearing tank to 2 m3 of pea gravel that allows a production of 60 kg / m3 tilapia. Based on the UVI-system, the size ratios themselves were perceived as a disadvantage since a relatively large ratio of plant growing area to fish surface area of at least 7:3 must be achieved for adequate plant production. On the other hand, system designs of coupled systems are highly variable, often not comparable, and the experiences made cannot be easily transferred to another system or location. Consequently, far more research data is needed in order to identify the best possible production ratios finally also enabling upscaling of coupled aquaponic systems through multiplying optimal designed basic modules (also see Chap. 11).
-
Challenge: Adverse water quality parameters have been stated to negatively affect fish health. As Yavuzcan Yildiz et al. (2017) pointed out, nutrient retention of plants should be maximized to avoid negative effects of water quality on fish welfare. It is important to select adequate fish species that can accept higher nutrient loads, such as the African catfish (C. gariepinus) or the Nile tilapia (O. niloticus,). More sensible species such as the Zander or pikeperch (Sander lucioperca) might be also applied in aquaponics because they prefer nutrient enriched or eutrophic water bodies with higher turbidity (Jeppesen et al. 2000; Keskinen and Marjomäki 2003; [see Sect. 7.7.1. Fish production]). So far, there is scant data allowing precise statements on fish welfare impairments. With plants generally needing high potassium concentrations between 230 and 400 mg/L inside the process water, 200–400 mg/L potassium showed no negative influence on African catfish welfare (Presas Basalo 2017). Similarly, 40 and 80 mg/L ortho-P in the rearing water had no negative impact on growth performance, feed efficiency and welfare traits of juvenile African catfish (Strauch et al. 2019).
-
Challenge: Another issue is the potential transmission of diseases in terms of food safety, to people through the consumption of plants that have been in contact with fish waste. In general, the occurrence of zoonoses is minor because closed aquaponics are fully controlled systems. However, germs can accumulate in the process water of the system components or in the fish gut. Escherichia coli and Salmonella spp. (zoonotic enteric bacteria) were identified as indicators of faecal contamination and microbial water quality, however, they were detected in aquaponics only in very small quantities (Munguia-Fragozo et al. 2015). Another comparison of smooth-textured leafy greens between aquaponics, hydroponics and soil-based production showed no significant differences in aerobic plate counts (APC, aerobic bacteria), Enterobacteriaceae, non-pathogenic E. coli and Listeria, suggesting a comparable contamination level with pathogens (Barnhart et al. 2015). Listeria spp. was most frequent (40%) in hydroponics with de-rooted plants (aquaponic plants with roots 0%, aquaponic plants without roots <10%), but not necessarily the harmful L. monocytogenes species. It was suggested that the source of the bacteria may be due to the lack of hygiene management, with little relevance to aquaponics as such. Another infectious bacterium, Fusobacteria (Cetobacterium), was detected by Schmautz et al. (2017) in the fish faeces with a high prevalence of up to 75%. Representatives of Fusobacteria are responsible for human diseases (hospital germ, abscesses, infections), reproducing in biofilms or as part of the fish intestines. Human infections with Fusobacteria from aquaponics have not yet been recorded but may be possible by neglecting the required hygiene protocols.
In general, there is rather little information about diseases caused by the consumption of fish and plants originating from coupled aquaponic systems. In Wilson (2005), Dr. J.E. Rakocy stated that there was no recorded human disease outbreak in 25 years of coupled aquaponic production. However, a washing procedure of the plant products should be used to reduce the number of bacteria as a precaution. A chlorine bath (100 ppm) followed by a potable water rinse was recommended by Chalmers (2004). If this methodology is used and the contact of the plants or plant products with the recirculating process water is avoided, the likelihood of contamination with human pathogenic bacteria can be strongly reduced. This is a necessary precaution not only for coupled but also for all other forms of aquaponics.
Change history
01 December 2020
Correction to: S. Goddek et al. (eds.), Aquaponics Food Production Systems, https://doi.org/10.1007/978-3-030-15943-6_3
References
Alberts-Hubatsch H, Ende S, Schuhn A, von der Marwitz C, Wirtz A, Fuchs V, Henjes J, Slater M (2017) Integration of hybrid striped bass Morone saxatilis x M. chrysops, noble crayfish Astacus Astacus, watercress Nasturtium officinale and microalge Nannochloropsis limnetica in an experimental aquaponic system. Dubrovnik meeting abstract, EAS 2017 Oostende, Belgium
Al-Hafedh YS, Alam A, Beltagi MS (2008) Food production and water conservation in a recirculating aquaponic system in Saudi Arabia at different ratios of fish feed to plants. J World Aquacult Soc 39(4):510–520. https://doi.org/10.1111/j.1749-7345.2008.00181.x
Appelbaum S, Kotzen B (2016) Further investigations of aquaponics using brackish water resources of the Negev desert. Ecocycles Sci J Eur Ecocycles Soc 2(2):26–35. https://doi.org/10.19040/ecocycles.v2i2s.53
Bais HP, Park SW, Weir TL, Callaway RM, Vivanco JM (2004) How plants communicate using the underground information superhighway. Trends Plant Sci 9(1):26–32
Barnhart C, Hayes L, Ringle D (2015) Food safety hazards associated with smooth textured leafy greens produced in aquaponic, hydroponic, and soil-based systems with and without roots in retail. University of Minnesota Aquaponics. The University of Minnesota Aquaponics. 17 p
Baßmann B, Brenner M, Palm HW (2017) Stress and welfare of African catfish (Clarias gariepinus Burchell, 1822) in a coupled aquaponic system. Water 9(7):504. https://doi.org/10.3390/w9070504
Baßmann B, Harbach H, Weißbach S, Palm HW (2018) Effect of plant density in coupled aquaponics on the welfare status of African catfish (Clarias gariepinus). J World Aquacult Soc. (in press)
Bittsanszky A, Uzinger N, Gyulai G, Mathis A, Villarroel M, Kotzen B, Komives T (2016) Nutrient supply of plants in aquaponic systems. Ecocycles Sci J Eur Ecocycles Soc 2(2):17–20. https://doi.org/10.19040/ecocycles.v2i2.57
Blidariu F, Drasovean A, Grozea A (2013a) Evaluation of phosphorus level in green lettuce conventional grown under natural conditions and aquaponic system. Bull Univ Agric Sci Vet Med Cluj-Napoca Anim Sci Biotechnol:128–135
Blidariu F, Alexandru D, Adrian G, Isidora R, Dacian L (2013b) Evolution of nitrate level in green lettuce conventional grown under natural conditions and aquaponic system. Sci Pap Anim Sci Biotechnol 46(1):244–250
Chalmers GA (2004) Aquaponics and food safety. Alberta April, Lethbridge, 77 p
Comeau Y (2008) Microbial metabolism. In: Biological wastewater treatment: principles, modeling and design, Cap, 2. IWA/Cambridge University Press, London, pp 9–32
Dakora FD, Phillips DA (2002) Root exudates as mediators of mineral acquisition in low-nutrient environments. In: Food security in nutrient-stressed environments: exploiting plants’ genetic capabilities. Springer, Dordrecht, pp 201–213
Danaher JJ, Shultz RC, Rakocy JE, Bailey DS (2013) Alternative solids removal for warm water recirculating raft aquaponic systems. J World Aquacult Soc 44(3):374–383. https://doi.org/10.1111/jwas.12040
Davy AJ, Bishop GF, Costa CSB (2001) Salicornia L. (Salicornia pusilla J. Woods, S. ramosissima J. Woods, S. europaea L., S. obscura PW ball & Tutin, S. nitens PW ball & Tutin, S. fragilis PW ball & Tutin and S. dolichostachya moss). J Ecol 89(4):681–707
Delaide B, Goddek S, Gott J, Soyeurt H, Jijakli MH (2016) Lettuce (Lactuca sativa L. var. Sucrine) growth performance in complemented aquaponic solution outperforms hydroponics. Water 8(10):467
Delaide B, Delhaye G, Dermience M, Gott J, Soyeurt H, Jijakli MH (2017) Plant and fish production performance, nutrient mass balances, energy and water use of the PAFF box, a small-scale aquaponic system. Aquac Eng 78:130–139
Diem TNT, Konnerup D, Brix H (2017) Effects of recirculation rates on water quality and Oreochromis niloticus growth in aquaponic systems. Aquac Eng 78:95–104. https://doi.org/10.1016/j.aquaeng.2017.05.002
Edaroyati MP, Aishah HS, Al-Tawaha AM (2017) Requirements for inserting intercropping in aquaponics system for sustainability in agricultural production system. Agron Res 15(5):2048–2067. https://doi.org/10.15159/AR.17.070
Endut A, Jusoh A, Ali N, Wan Nik WNS, Hassan A (2009) Effect of flow rate on water quality parameters and plant growth of water spinach (Ipomoea aquatica) in an aquaponic recirculating system. Desalin Water Treat 5:19–28. https://doi.org/10.5004/dwt.2009.559
FAO (2017) In: Subasinghe R (ed) World aquaculture 2015: a brief overview, FAO fisheries and aquaculture circular no. 1140. FAO, Rome
Geelen C (2016) Dynamic model of an INAPRO demonstration aquaponic system. Thesis biobased chemistry and technology. Wageningen University Agrotechnology and Food Sciences. 50 p
Goddek S, Vermeulen T (2018) Comparison of Lactuca sativa growth performance in conventional and RAS-based hydroponic systems. Aquac Int:1–10. https://doi.org/10.1007/s10499-018-0293-8
Goddek S, Espinal C, Delaide B, Jijakli M, Schmautz Z, Wuertz S, Keesman K (2016) Navigating towards decoupled aquaponic systems: a system dynamics design approach. Water 8:303. https://doi.org/10.3390/w8070303
Graber A, Junge R (2009) Aquaponic systems: nutrient recycling from fish wastewater by vegetable production. Desalination 246(1–3):147–156
Graber A, Antenen N, Junge R (2014) The multifunctional aquaponic system at ZHAW used as research and training lab. In: Conference VIVUS: transmission of innovations, knowledge and practical experience into everyday practice. Strahinj: Biotehniški center, Naklo. http://www.bc-naklo.si/uploads/media/29-Graber-Antenen-Junge-Z.pdf
Gunning D (2016) Cultivating Salicornia europaea (Marsh Samphire), Irish Sea Fisheries Board, http://www.bim.ie/media/bim/content/news,and,events/BIM,Cultivating,,Salicornia,europaea,-,Marsh,Samphire.pdf
Heuvelink E (2018) Tomatoes. In: Heuvelink E (ed) Crop production science in horticulture. Wageningen University/CABI, Wallingford, 388 p
Hortidaily (2015) Dutch start third trial on tomatoes with 100% LED-lighting: 100,6 Kg/m2 at the Improvement Centre in Bleiswijk. What’s next? https://www.hortidaily.com/article/6022598/kg-m-at-the-improvement-centre-in-bleiswijk-what-s-next/
Hussain T, Verma AK, Tiwari VK, Prakash C, Rathore G, Shete AP, Nuwansi KKT (2014) Optimizing koi carp, Cyprinus carpio var. koi (Linnaeus, 1758), stocking density and nutrient recycling with spinach in an aquaponic system. J World Aquacult Soc 45(6):652–661. https://doi.org/10.1111/jwas.12159
Hussain T, Verma AK, Tiwari VK, Prakash C, Rathore G, Shete AP, Saharan N (2015) Effect of water flow rates on growth of Cyprinus carpio var. koi (Cyprinus carpio L., 1758) and spinach plant in aquaponic system. Aquac Int 23(1):369–384. https://doi.org/10.1007/s10499-014-9821-3
Jeppesen E, Jensen JP, SØndergaard M, Lauridsen T, Landkildehus F (2000) Trophic structure, species richness and biodiversity in Danish lakes: changes along a phosphorus gradient. Freshw Biol 45(2):201–218
Keskinen T, Marjomäki TJ (2003) Growth of pikeperch in relation to lake characteristics: total phosphorus, water colour, lake area and depth. J Fish Biol 63(5):1274–1282
Khandaker M, Kotzen B (2018) The potential for combining living wall and vertical farming systems with aquaponics with special emphasis on substrates. Aquac Res 49(4):1454–1468
Knaus U, Palm HW (2017a) Effects of fish biology on ebb and flow aquaponical cultured herbs in northern Germany (Mecklenburg Western Pomerania). Aquaculture 466:51–63. https://doi.org/10.1016/j.aquaculture.2016.09.025
Knaus U, Palm HW (2017b) Effects of the fish species choice on vegetables in aquaponics under spring-summer conditions in northern Germany (Mecklenburg Western Pomerania). Aquaculture 473:62–73. https://doi.org/10.1016/j.aquaculture.2017.01.020
Knaus U, Segade Á, Robaina L (2015) Training School 1 – Aquaponic trials: improving water quality and plant production through fish management and diet. 25–29 May 2015, Universidad de Las Palmas de Gran Canaria, Spain. Cost Action FA1305, The EU Aquaponics Hub – Realising Sustainable Integrated Fish and Vegetable Production for the EU
Knaus U, Appelbaum S, Palm HW (2018a) Significant factors affecting the economic sustainability of closed backyard aquaponics systems. Part IV: autumn herbs and polyponics. AACL Bioflux 11(6):1760–1775
Knaus U, Appelbaum S, Castro C, Sireeni J, Palm HW (2018b) Growth performance of basil in a small-scale aquaponic system with the production of tilapia (Oreochromis niloticus) and African catfish (Clarias gariepinus). (In preparation)
König B, Junge R, Bittsanszky A, Villarroel M, Komives T (2016) On the sustainability of aquaponics. Ecocycles Sci J Eur Ecocycles Soc 2(1):26–32. https://doi.org/10.19040/ecocycles.v2i1.50
Kotzen B, Appelbaum S (2010) An investigation of aquaponics using brackish water resources in the Negev Desert. J Appl Aquac 22(4):297–320. https://doi.org/10.1080/10454438.2010.527571
Lehmonen R, Sireeni J (2017) Comparison of plant growth and quality in hydroponic and aquaponic systems. Bachelor’s thesis, University of Jyväskylä, Jyväskylä, Finland. 27 p
Lennard WA, Leonard BV (2004) A comparison of reciprocating flow versus constant flow in an integrated, gravel bed, aquaponic test system. Aquac Int 12(6):539–553. https://doi.org/10.1007/s10499-004-8528-2
Lennard WA, Leonard BV (2006) A comparison of three different hydroponic sub-systems (gravel bed, floating and nutrient film technique) in an aquaponic test system. Aquac Int 14(6):539–550. https://doi.org/10.1007/s10499-006-9053-2
Lewis WM, Yopp JH, Schramm HL Jr, Brandenburg AM (1978) Use of hydroponics to maintain quality of recirculated water in a fish culture system. Trans Am Fish Soc 107(1):92–99. https://doi.org/10.1577/1548-8659(1978)107<92:UOHTMQ>2.0.CO;2
Lobillo JR, Fernández-Cabanás VM, Carmona E, Candón FJL (2014) Manejo básico y resultados preliminares de crecimiento y supervivencia de tencas (Tinca tinca L.) y lechugas (Lactuca sativa L.) en un prototipo acuapónico. ITEA 110(2):142–159. https://doi.org/10.12706/itea.2014.009
Lorena S, Cristea V, Oprea L (2008) Nutrients dynamic in an aquaponic recirculating system for sturgeon and lettuce (Lactuca sativa) production. Lucrări Științifice-Zootehnie și Biotehnologii, Universitatea de Științe Agricole și Medicină Veterinară a Banatului Timișoara 41(2):137–143
Lorenzen L (2017) Vergleich des Wachstums von chinesischem Blätterkohl (Brassica rapa chinensis) in drei verschiedenen Hydroponiksubsystemen unter aquaponischen Bedingungen. Agrar- und Umweltwissenschaftliche Fakultät, Lehrstuhl Aquakultur und Sea-Ranching, Universität Rostock. Masterarbeit. 63 p. [in German]
Love DC, Fry JP, Li X, Hill ES, Genello L, Semmens K, Thompson RE (2015) Commercial aquaponics production and profitability: findings from an international survey. Aquaculture 435:67–74. https://doi.org/10.1016/j.aquaculture.2014.09.023
Loyacano HA, Grosvenor RB (1973) Effects of Chinese waterchestnut in floating rafts on production of channel catfish in plastic pools. Proc Annu Conf Southeast Assoc Game Fish Comm 27:471–473
Mariscal-Lagarda MM, Páez-Osuna F, Esquer-Méndez JL, Guerrero-Monroy I, del Vivar AR, Félix-Gastelum R (2012) Integrated culture of white shrimp (Litopenaeus vannamei) and tomato (Lycopersicon esculentum mill) with low salinity groundwater: management and production. Aquaculture 366:76–84. https://doi.org/10.1016/j.aquaculture.2012.09.003
McMurtry MR, Nelson PV, Sanders DC, Hodges L (1990) Sand culture of vegetables using recirculated aquacultural effluents. Appl Agric Res 5(4):280–284
McMurtry MR, Sanders DC, Patterson RP, Nash A (1993) Yield of tomato irrigated with recirculating aquacultural water. J Prod Agric 6(3):428–432. https://doi.org/10.2134/jpa1993.0428
McMurtry MR, Sanders DC, Cure JD, Hodson RG, Haning BC, Amand PCS (1997) Efficiency of water use of an integrated fish/vegetable co-culture system. J World Aquacult Soc 28(4):420–428. https://doi.org/10.1111/j.1749-7345.1997.tb00290.x
Munguia-Fragozo P, Alatorre-Jacome O, Rico-Garcia E, Torres-Pacheco I, Cruz-Hernandez A, Ocampo-Velazquez RV, Garcia-Trejo JF, Guevara-Gonzalez RG (2015) Perspective for aquaponic systems: “omic” technologies for microbial community analysis. BioMed Res Int 2015: 480386. Hindawi Publishing Corporation. BioMed Research International. 2015, 10 pages. https://doi.org/10.1155/2015/480386
Naegel LC (1977) Combined production of fish and plants in recirculating water. Aquaculture 10(1):17–24. https://doi.org/10.1016/0044-8486(77)90029-1
Nelson RL, Pade JS (2007) Aquaponic equipment the clarifier. Aquaponics J 4(47):30–31
Nozzi V, Strofaldi S, Piquer IF, Di Crescenzo D, Olivotto I, Carnevali O (2016) Amyloodinum ocellatum in Dicentrarchus labrax: study of infection in salt water and freshwater aquaponics. Fish Shellfish Immunol 57:179–185. https://doi.org/10.1016/j.fsi.2016.07.036
Nuwansi KKT, Verma AK, Prakash C, Tiwari VK, Chandrakant MH, Shete AP, Prabhath GPWA (2016) Effect of water flow rate on polyculture of koi carp (Cyprinus carpio var. koi) and goldfish (Carassius auratus) with water spinach (Ipomoea aquatica) in recirculating aquaponic system. Aquac Int 24(1):385–393. https://doi.org/10.1007/s10499-015-9932-5
Palm HW, Seidemann R, Wehofsky S, Knaus U (2014a) Significant factors influencing the economic sustainability of closed aquaponic systems. part I: system design, chemo-physical parameters and general aspects. AACL Bioflux 7(1):20–32
Palm HW, Bissa K, Knaus U (2014b) Significant factors affecting the economic sustainability of closed aquaponic systems. part II: fish and plant growth. AACL Bioflux. 7(3):162–175
Palm HW, Nievel M, Knaus U (2015) Significant factors affecting the economic sustainability of closed aquaponic systems. part III: plant units. AACL Bioflux 8(1):89–106
Palm HW, Strauch S, Knaus U, Wasenitz B (2016) Das FischGlasHaus – eine Innovationsinitiative zur energie- und nährstoffeffizienten Produktion unterschiedlicher Fisch- und Pflanzenarten in Mecklenburg-Vorpommern (“Aquaponik in MV”). Fischerei & Fischmarkt in Mecklenburg-Vorpommern 1/2016–16:38–47 [in German]
Palm HW, Wasenitz B, Knaus U, Bischoff A, Strauch SM (2017) Two years of aquaponics research in the FishGlassHouse – lessons learned. Dubrovnik meeting abstract, Aquaculture Europe 2017, EAS Oostende Belgium
Palm HW, Knaus U, Appelbaum S, Goddek S, Strauch SM, Vermeulen T, Jijakli MH, Kotzen B (2018) Towards commercial aquaponics: a review of systems, designs, scales and nomenclature. Aquac Int 26(3):813–842
Palm HW, Knaus U, Wasenitz B, Bischoff-Lang AA, Strauch SM (2019) Proportional up scaling of African catfish (Clarias gariepinus Burchell, 1822) commercial recirculating aquaculture systems disproportionally affects nutrient dynamics. Aquac Int 26(3):813–842. https://doi.org/10.1016/j.aquaculture.2018.03.021
Pantanella E (2012) Nutrition and quality of aquaponic systems. Ph.D. thesis. Università degli Studi della Tuscia. Viterbo, Italy. 124 p
Presas Basalo F (2017) Does water potassium concentration in aquaponics affect the performance of African catfish Clarias gariepinus (Burchell, 1822)? Master’s thesis, Department of Animal Sciences Aquaculture and Fisheries Group, Wageningen University and University of Rostock Department of Aquaculture and Sea-Ranching. 75 p
Pribbernow M (2016) Vergleich des Wachstums von Basilikum (Ocimum basilicum L.) in drei verschiedenen Hydroponik-Subsystemen unter aquaponischer Produktion. Agrar- und Umweltwissenschaftliche Fakultät, Lehrstuhl Aquakultur und Sea-Ranching, Universität Rostock. Masterarbeit. 98 p. [in German]
Rakocy JE (1989) Hydroponic lettuce production in a recirculating fish culture system. In: Island perspectives. Vol. 3. agricultural experiment station, University of the Virgin Islands. pp 5–10
Rakocy JE (2012) Chapter 14: aquaponics – integrating fish and plant culture. In: Tidwell JH (ed) Aquaculture production systems. Wiley-Blackwell, Oxford, pp 344–386
Rakocy JE, Shultz RC, Bailey DS (2000) Commercial aquaponics for the Caribbean: proceedings of the Gulf and Caribbean Fisheries Institute [Proc. Gulf Caribb. Fish. Inst.]. no. 51, pp 353–364
Rakocy J, Shultz RC, Bailey DS, Thoman ES (2003) Aquaponic production of tilapia and basil: comparing a batch and staggered cropping system. In: South Pacific soilless culture conference-SPSCC 648. pp 63–69
Rakocy JE, Bailey DS, Shultz RC, Thoman ES (2004) Update on tilapia and vegetable production in the UVI aquaponic system. In: New dimensions on farmed Tilapia: proceedings of the sixth international symposium on Tilapia in Aquaculture, held September. pp 12–16
Rakocy JE, Masser MP, Losordo TM (2006) Recirculating aquaculture tank production systems: aquaponics-integrating fish and plant culture. SRAC Publication 454:1–16
Rakocy JE, Bailey DS, Shultz RC, Danaher JJ (2011) A commercial-scale aquaponic system developed at the University of the Virgin Islands. In: Proceedings of the 9th international symposium on Tilapia in Aquaculture. pp 336–343
Ronzón-Ortega M, Hernández-Vergara MP, Pérez-Rostro CI (2012) Hydroponic and aquaponic production of sweet basil (Ocimum basilicum) and giant river prawn (Macrobrachium rosenbergii). Trop Subtrop Agroecosyst 15(2):63–71
Sace CF, Fitzsimmons KM (2013) Vegetable production in a recirculating aquaponic system using Nile tilapia (Oreochromis niloticus) with and without freshwater prawn (Macrobrachium rosenbergii). Acad J Agric Res 1(12):236–250
Saha S, Monroe A, Day MR (2016) Growth, yield, plant quality and nutrition of basil (Ocimum basilicum L.) under soilless agricultural systems. Ann Agric Sci 61(2):181–186
Savidov N (2005) Evaluation of aquaponics technology in Alberta, Canada. Aquaponics Journal 37:20–25
Schmautz Z, Graber A, Jaenicke S, Goesmann A, Junge R, Smits TH (2017) Microbial diversity in different compartments of an aquaponics system. Arch Microbiol 199(4):613–620
Seawright DE, Stickney RR, Walker RB (1998) Nutrient dynamics in integrated aquaculture–hydroponics systems. Aquaculture 160(3–4):215–237. https://doi.org/10.1016/S0044-8486(97)00168-3
Shete AP, Verma AK, Chadha NK, Prakash C, Peter RM, Ahmad I, Nuwansi KKT (2016) Optimization of hydraulic loading rate in aquaponic system with common carp (Cyprinus carpio) and mint (Mentha arvensis). Aquac Eng 72:53–57. https://doi.org/10.1016/j.aquaeng.2016.04.004
Simeonidou M, Paschos I, Gouva E, Kolygas M, Perdikaris C (2012) Performance of a small-scale modular aquaponic system. AACL Bioflux. 5(4):182–188
Skar SLG, Liltved H, Drengstig A, Homme JM, Kledal PR, Paulsen H, Björnsdottir R, Oddson S, Savidov N (2015) Aquaponics NOMA (Nordic Marin) – new innovations for sustainable aquaculture in the Nordic countries. Nordic Innovation Publication 2015:06, 108 p
Somerville C, Cohen M, Pantanella E, Stankus A, Lovatelli A (2014) Small-scale aquaponic food production. Integrated fish and plant farming, FAO fisheries and aquaculture technical paper no. 589. FAO, Rome. (262 pp)
Strauch SM, Wenzel LC, Bischoff A, Dellwig O, Klein J, Schüch A, Wasenitz B, Palm HW (2018) Commercial African catfish (Clarias gariepinus) recirculating aquaculture systems: assessment of element and energy pathways with special focus on the phosphorus cycle. Sustainability 2018(10):1805. https://doi.org/10.3390/su10061805
Strauch SM, Bischoff AA, Bahr J, Baßmann B, Oster M, Wasenitz B, Palm HW (2019, submitted) Effects of ortho-phosphate on growth performance, welfare and product quality of juvenile African catfish (Clarias gariepinus). Fishes 4:3. https://doi.org/10.3390/fishes4010003
Sutton RJ, Lewis WM (1982) Further observations on a fish production system that incorporates hydroponically grown plants. Progress Fish Cult 44(1):55–59. https://doi.org/10.1577/1548-8659(1982)44[55,FOOAFP]2.0.CO;2
Tarafdar JC, Claassen N (1988) Organic phosphorus compounds as a phosphorus source for higher plants through the activity of phosphatases produced by plant roots and microorganisms. Biol Fertil Soils 5(4):308–312
Tarafdar JC, Yadav RS, Meena SC (2001) Comparative efficiency of acid phosphatase originated from plant and fungal sources. J Plant Nutr Soil Sci 164(3):279–282
Thorarinsdottir RI (2014) Implementing commercial aquaponics in Europe – first results from the Ecoinnovation project EcoPonics. Aquaculture Europe 14, October 14–17 2014, San Sebastian, Spain
Thorarinsdottir RI, Kledal PR, Skar SLG, Sustaeta F, Ragnarsdottir KV, Mankasingh U, Pantanella E, van de Ven R, Shultz RC (2015) Aquaponics guidelines. 64 p
Tran H (2015) Aquaponics-coupled and decoupled systems and the water quality needs of each. World Aquaculture 2015 – Meeting Abstract Jeju, Korea, May 27, 2015. World Aquaculture Society PO Box 397 Sorrento, LA 70778–0397 (USA)
Tyson RV, Simonne EH, Treadwell DD, White JM, Simonne A (2008) Reconciling pH for ammonia biofiltration and cucumber yield in a recirculating aquaponic system with perlite biofilters. Hortscience 43(3):719–724
USGS. The USGS Water Science School. https://water.usgs.gov/edu/gallery/global-water-volume.html
Van der Heijden PGM, Roest CWJ, Farrag F, ElWageih H, Sadek S, Hartgers EM, Nysingh SL (2014) Integrated agri-aquaculture with brackish waters in Egypt: mission report (March 9–March 17, 2014) (No. 2526). Alterra Wageningen UR
Villarroel M, Rodriguez Alvariño JM, Duran Altisent JM (2011) Aquaponics: integrating fish feeding rates and ion waste production for strawberry hydroponics. Span J Agric Res 9(2):537–545
Waller U, Buhmann AK, Ernst A, Hanke V, Kulakowski A, Wecker B, Orellana J, Papenbrock J (2015) Integrated multi-trophic aquaculture in a zero-exchange recirculation aquaculture system for marine fish and hydroponic halophyte production. Aquac Int 23(6):1473–1489. https://doi.org/10.1007/s10499-015-9898-3
Watten BJ, Busch RL (1984) Tropical production of tilapia (Sarotherodon aurea) and tomatoes (Lycopersicon esculentum) in a small-scale recirculating water system. Aquaculture 41(3):271–283. https://doi.org/10.1016/0044-8486(84)90290-4
Wermter L (2016) Comparison of three different hydroponic sub-systems of Cucumis sativus L. grown in an aquaponic system. Department of Agronomy. Faculty of Agricultural and Environmental Sciences (AUF), Professorship of Crop production and Aquaculture and Sea-Ranching. University of Rostock, Germany. Masterthesis. 40 p
Wilson G (2005) Greenhouse aquaponics proves superior to inorganic hydroponics. Aquaponics J 39(4):14–17
Yavuzcan Yildiz H, Robaina L, Pirhonen J, Mente E, Domínguez D, Parisi G (2017) Fish welfare in aquaponic systems: its relation to water quality with an emphasis on feed and faeces-a review. Water 9(1):13
Zimmermann J (2017) Vergleich des Wachstums von Marokkanischer Minze (Mentha spicata) in drei verschiedenen Hydroponik Subsystemen unter aquaponischer Produktion. Agrar- und Umweltwissenschaftliche Fakultät, Lehrstuhl Aquakultur und Sea-Ranching, Universität Rostock. Masterarbeit. 71 p. [in German]
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2019 The Author(s)
About this chapter
Cite this chapter
Palm, H.W., Knaus, U., Appelbaum, S., Strauch, S.M., Kotzen, B. (2019). Coupled Aquaponics Systems. In: Goddek, S., Joyce, A., Kotzen, B., Burnell, G.M. (eds) Aquaponics Food Production Systems. Springer, Cham. https://doi.org/10.1007/978-3-030-15943-6_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-15943-6_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-15942-9
Online ISBN: 978-3-030-15943-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)