Abstract
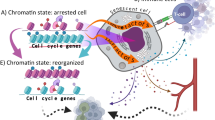
Cancers are frequently addicted to initiating oncogenes that elicit aberrant cellular proliferation, self-renewal, and apoptosis. Restoration of oncogenes to normal physiologic regulation can elicit dramatic reversal of the neoplastic phenotype, including reduced proliferation and increased apoptosis of tumor cells (Science 297(5578):63–64, 2002). In some cases, oncogene inactivation is associated with compete elimination of a tumor. However, in other cases, oncogene inactivation induces a conversion of tumor cells to a dormant state that is associated with cellular differentiation and/or loss of the ability to self-replicate. Importantly, this dormant state is reversible, with tumor cells regaining the ability to self-renew upon oncogene reactivation. Thus, understanding the mechanism of oncogene inactivation-induced dormancy may be crucial for predicting therapeutic outcome of targeted therapy. One important mechanistic insight into tumor dormancy is that oncogene addiction might involve regulation of a decision between self-renewal and cellular senescence. Recent evidence suggests that this decision is regulated by multiple mechanisms that include tumor cell-intrinsic, cell-autonomous mechanisms and host-dependent, tumor cell-non-autonomous programs (Mol Cell 4(2):199–207, 1999; Science 297(5578):102–104, 2002; Nature 431(7012):1112–1117, 2004; Proc Natl Acad Sci U S A 104(32):13028–13033, 2007). In particular, the tumor microenvironment, which is known to be critical during tumor initiation (Cancer Cell 7(5):411–423, 2005; J Clin Invest 121(6):2436–2446, 2011), prevention (Nature 410(6832):1107–1111, 2001), and progression (Cytokine Growth Factor Rev 21(1):3–10, 2010), also appears to dictate when oncogene inactivation elicits the permanent loss of self-renewal through induction of cellular senescence (Nat Rev Clin Oncol 8(3):151–160, 2011; Science 313(5795):1960–1964, 2006; N Engl J Med 351(21):2159–21569, 2004). Thus, oncogene addiction may be best modeled as a consequence of the interplay amongst cell-autonomous and host-dependent programs that define when a therapy will result in tumor dormancy.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Weinstein IB (2002) Cancer. Addiction to oncogenes—the Achilles heal of cancer. Science 297(5578):63–64
Felsher DW, Bishop JM (1999) Reversible tumorigenesis by MYC in hematopoietic lineages. Mol Cell 4(2):199–207
Jain M et al (2002) Sustained loss of a neoplastic phenotype by brief inactivation of MYC. Science 297(5578):102–104
Shachaf CM et al (2004) MYC inactivation uncovers pluripotent differentiation and tumour dormancy in hepatocellular cancer. Nature 431(7012):1112–1117
Wu CH et al (2007) Cellular senescence is an important mechanism of tumor regression upon c-Myc inactivation. Proc Natl Acad Sci U S A 104(32):13028–13033
de Visser KE, Korets LV, Coussens LM (2005) De novo carcinogenesis promoted by chronic inflammation is B lymphocyte dependent. Cancer Cell 7(5):411–423
Dougan M et al (2011) A dual role for the immune response in a mouse model of inflammation-associated lung cancer. J Clin Invest 121(6):2436–2446
Shankaran V et al (2001) IFNgamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature 410(6832):1107–1111
Ruffell B et al (2010) Lymphocytes in cancer development: polarization towards pro-tumor immunity. Cytokine Growth Factor Rev 21(1):3–10
Zitvogel L, Kepp O, Kroemer G (2011) Immune parameters affecting the efficacy of chemotherapeutic regimens. Nat Rev Clin Oncol 8(3):151–160
Galon J et al (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313(5795):1960–1964
Dave SS et al (2004) Prediction of survival in follicular lymphoma based on molecular features of tumor-infiltrating immune cells. N Engl J Med 351(21):2159–2169
Felsher DW (2003) Cancer revoked: oncogenes as therapeutic targets. Nat Rev Cancer 3(5):375–380
Weinstein IB, Joe A (2008) Oncogene addiction. Cancer Res 68(9):3077–3080; discussion 3080
Sharma SV, Settleman J (2007) Oncogene addiction: setting the stage for molecularly targeted cancer therapy. Genes Dev 21(24):3214–3231
Huettner CS et al (2000) Reversibility of acute B-cell leukaemia induced by BCR-ABL1. Nat Genet 24(1):57–60
Chin L et al (1999) Essential role for oncogenic Ras in tumour maintenance. Nature 400(6743):468–472
Hoeflich KP et al (2006) Oncogenic BRAF is required for tumor growth and maintenance in melanoma models. Cancer Res 66(2):999–1006
Boxer RB et al (2004) Lack of sustained regression of c-MYC-induced mammary adenocarcinomas following brief or prolonged MYC inactivation. Cancer Cell 6(6):577–586
Giuriato S et al (2006) Sustained regression of tumors upon MYC inactivation requires p53 or thrombospondin-1 to reverse the angiogenic switch. Proc Natl Acad Sci USA 103(44): 16266–16271
Shchors K et al (2006) The Myc-dependent angiogenic switch in tumors is mediated by interleukin 1beta. Genes Dev 20(18):2527–2538
Tran PT et al (2008) Combined inactivation of MYC and K-Ras oncogenes reverses tumorigenesis in lung adenocarcinomas and lymphomas. PLoS One 3(5):e2125
Hait WN, Hambley TW (2009) Targeted cancer therapeutics. Cancer Res 69(4):1263–1267; discussion 1267
Sawyers C (2004) Targeted cancer therapy. Nature 432(7015):294–297
Druker BJ et al (2001) Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 344(14):1031–1037
Hudis CA (2007) Trastuzumab—mechanism of action and use in clinical practice. N Engl J Med 357(1):39–51
Chapman PB et al (2011) Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med 364(26):2507–2516
Cataldo VD et al (2011) Treatment of non-small-cell lung cancer with erlotinib or gefitinib. N Engl J Med 364(10):947–955
Felsher DW (2004) Reversibility of oncogene-induced cancer. Curr Opin Genet Dev 14(1):37–42
Sharma SV, Settleman J (2006) Oncogenic shock: turning an activated kinase against the tumor cell. Cell Cycle 5(24):2878–2880
Kaelin WG Jr (2005) The concept of synthetic lethality in the context of anticancer therapy. Nat Rev Cancer 5(9):689–698
Albini A, Sporn MB (2007) The tumour microenvironment as a target for chemoprevention. Nat Rev Cancer 7(2):139–147
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674
Coussens LM et al (2000) MMP-9 supplied by bone marrow-derived cells contributes to skin carcinogenesis. Cell 103(3):481–490
Bissell MJ, Radisky D (2001) Putting tumours in context. Nat Rev Cancer 1(1):46–54
Choi PS et al (2011) Lymphomas that recur after MYC suppression continue to exhibit oncogene addiction. Proc Natl Acad Sci U S A 108(42):17432–17437
Gorre ME et al (2001) Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 293(5531):876–880
Pao W et al (2005) KRAS mutations and primary resistance of lung adenocarcinomas to gefitinib or erlotinib. PLoS Med 2(1):e17
Aguirre-Ghiso JA (2007) Models, mechanisms and clinical evidence for cancer dormancy. Nat Rev Cancer 7(11):834–846
Felsher DW (2010) MYC inactivation elicits oncogene addiction through both tumor cell-intrinsic and host-dependent mechanisms. Genes Cancer 1(6):597–604
Nardella C et al (2011) Pro-senescence therapy for cancer treatment. Nat Rev Cancer 11(7):503–511
Felsher DW (2008) Reversing cancer from inside and out: oncogene addiction, cellular senescence, and the angiogenic switch. Lymphat Res Biol 6(3–4):149–154
Felsher DW (2008) Tumor dormancy and oncogene addiction. APMIS 116(7–8):629–637
Bishop JM (1991) Molecular themes in oncogenesis. Cell 64(2):235–248
Reya T et al (2001) Stem cells, cancer, and cancer stem cells. Nature 414(6859):105–111
Dick JE (2008) Stem cell concepts renew cancer research. Blood 112(13):4793–4807
Wang JC, Dick JE (2005) Cancer stem cells: lessons from leukemia. Trends Cell Biol 15(9):494–501
Sell S (2004) Stem cell origin of cancer and differentiation therapy. Crit Rev Oncol Hematol 51(1):1–28
Nguyen LV et al (2012) Cancer stem cells: an evolving concept. Nat Rev Cancer 12(2):133–143
Das B et al (2009) The idea and evidence for the tumor stemness switch. In: Rajasekhar V, Vemuri M (eds) Regulatory networks in stem cells. Humana Press, New York, pp 473–487
Zheng H et al (2008) Pten and p53 converge on c-Myc to control differentiation, self-renewal, and transformation of normal and neoplastic stem cells in glioblastoma. Cold Spring Harb Symp Quant Biol 73:427–437
Wang J et al (2008) c-Myc is required for maintenance of glioma cancer stem cells. PLoS One 3(11):e3769
Marquardt JU et al (2011) Human hepatic cancer stem cells are characterized by common stemness traits and diverse oncogenic pathways. Hepatology 54(3):1031–1042
Hayflick L, Moorhead PS (1961) The serial cultivation of human diploid cell strains. Exp Cell Res 25:585–621
Hayflick L (1965) The limited in vitro lifetime of human diploid cell strains. Exp Cell Res 37:614–636
Lundblad V, Szostak JW (1989) A mutant with a defect in telomere elongation leads to senescence in yeast. Cell 57(4):633–643
Yu GL et al (1990) In vivo alteration of telomere sequences and senescence caused by mutated tetrahymena telomerase RNAs. Nature 344(6262):126–132
Chen Q, Ames BN (1994) Senescence-like growth arrest induced by hydrogen peroxide in human diploid fibroblast F65 cells. Proc Natl Acad Sci U S A 91(10):4130–4134
Di Leonardo A et al (1994) DNA damage triggers a prolonged p53-dependent G1 arrest and long-term induction of Cip1 in normal human fibroblasts. Genes Dev 8(21):2540–2551
Schmitt CA et al (2002) A senescence program controlled by p53 and p16INK4a contributes to the outcome of cancer therapy. Cell 109(3):335–346
Chang BD et al (1999) A senescence-like phenotype distinguishes tumor cells that undergo terminal proliferation arrest after exposure to anticancer agents. Cancer Res 59(15):3761–3767
Michishita E et al (1999) 5-Bromodeoxyuridine induces senescence-like phenomena in mammalian cells regardless of cell type or species. J Biochem 126(6):1052–1059
O’Brien W, Stenman G, Sager R (1986) Suppression of tumor growth by senescence in virally transformed human fibroblasts. Proc Natl Acad Sci U S A 83(22):8659–8663
Serrano M et al (1997) Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88(5):593–602
Zhu J et al (1998) Senescence of human fibroblasts induced by oncogenic Raf. Genes Dev 12(19):2997–3007
Braig M et al (2005) Oncogene-induced senescence as an initial barrier in lymphoma development. Nature 436(7051):660–665
Dimri GP et al (1995) A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci U S A 92(20):9363–9367
Narita M et al (2003) Rb-mediated heterochromatin formation and silencing of E2F target genes during cellular senescence. Cell 113(6):703–716
Collado M et al (2005) Tumour biology: senescence in premalignant tumours. Nature 436(7051):642
Chan HM et al (2005) The p400 E1A-associated protein is a novel component of the p53 –> p21 senescence pathway. Genes Dev 19(2):196–201
Chen Z et al (2005) Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 436(7051):725–730
van Riggelen J et al (2010) The interaction between Myc and Miz1 is required to antagonize TGFbeta-dependent autocrine signaling during lymphoma formation and maintenance. Genes Dev 24(12):1281–1294
Reimann M et al (2010) Tumor stroma-derived TGF-beta limits Myc-driven lymphomagenesis via Suv39h1-dependent senescence. Cancer Cell 17(3):262–272
Zhuang D et al (2008) C-MYC overexpression is required for continuous suppression of oncogene-induced senescence in melanoma cells. Oncogene 27(52):6623–6634
Lin AW et al (1998) Premature senescence involving p53 and p16 is activated in response to constitutive MEK/MAPK mitogenic signaling. Genes Dev 12(19):3008–3019
Rakhra K et al (2010) CD4(+) T cells contribute to the remodeling of the microenvironment required for sustained tumor regression upon oncogene inactivation. Cancer Cell 18(5):485–498
Xue W et al (2007) Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 445(7128):656–660
Balkwill F, Mantovani A (2001) Inflammation and cancer: back to Virchow? Lancet 357(9255):539–545
Dunn GP et al (2002) Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol 3(11):991–998
Schreiber RD, Old LJ, Smyth MJ (2011) Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 331(6024):1565–1570
de Visser KE, Eichten A, Coussens LM (2006) Paradoxical roles of the immune system during cancer development. Nat Rev Cancer 6(1):24–37
Andreu P et al (2010) FcRgamma activation regulates inflammation-associated squamous carcinogenesis. Cancer Cell 17(2):121–134
Girardi M et al (2004) Characterizing the protective component of the alphabeta T cell response to transplantable squamous cell carcinoma. J Invest Dermatol 122(3):699–706
Lin EY et al (2001) Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy. J Exp Med 193(6):727–740
Hung K et al (1998) The central role of CD4(+) T cells in the antitumor immune response. J Exp Med 188(12):2357–2368
Martin-Manso G et al (2008) Thrombospondin 1 promotes tumor macrophage recruitment and enhances tumor cell cytotoxicity of differentiated U937 cells. Cancer Res 68(17):7090–7099
Zhou P et al (2010) Mature B cells are critical to T-cell-mediated tumor immunity induced by an agonist anti-GITR monoclonal antibody. J Immunother 33(8):789–797
Wu J, Lanier LL (2003) Natural killer cells and cancer. Adv Cancer Res 90:127–156
DeNardo DG et al (2009) CD4(+) T cells regulate pulmonary metastasis of mammary carcinomas by enhancing protumor properties of macrophages. Cancer Cell 16(2):91–102
Condeelis J, Pollard JW (2006) Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 124(2):263–266
Coussens LM, Werb Z (2002) Inflammation and cancer. Nature 420(6917):860–867
Greten FR et al (2004) IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell 118(3):285–296
Ekbom A et al (1990) Ulcerative colitis and colorectal cancer. A population-based study. N Engl J Med 323(18):1228–1233
Wakabayashi O et al (2003) CD4+ T cells in cancer stroma, not CD8+ T cells in cancer cell nests, are associated with favorable prognosis in human non-small cell lung cancers. Cancer Sci 94(11):1003–1009
Zhang JP et al (2009) Increased intratumoral IL-17-producing cells correlate with poor survival in hepatocellular carcinoma patients. J Hepatol 50(5):980–989
Mantovani A et al (2008) Cancer-related inflammation. Nature 454(7203):436–444
Zhang H et al (2003) Concordant down-regulation of proto-oncogene PML and major histocompatibility antigen HLA class I expression in high-grade prostate cancer. Cancer Immun 3:2
Zheng P et al (1998) Proto-oncogene PML controls genes devoted to MHC class I antigen presentation. Nature 396(6709):373–376
Sumimoto H et al (2006) The BRAF-MAPK signaling pathway is essential for cancer-immune evasion in human melanoma cells. J Exp Med 203(7):1651–1656
Boni A et al (2010) Selective BRAFV600E inhibition enhances T-cell recognition of melanoma without affecting lymphocyte function. Cancer Res 70(13):5213–5219
Borrello MG et al (2005) Induction of a proinflammatory program in normal human thyrocytes by the RET/PTC1 oncogene. Proc Natl Acad Sci U S A 102(41):14825–14830
Ancrile B, Lim KH, Counter CM (2007) Oncogenic Ras-induced secretion of IL6 is required for tumorigenesis. Genes Dev 21(14):1714–1719
Sparmann A, Bar-Sagi D (2004) Ras-induced interleukin-8 expression plays a critical role in tumor growth and angiogenesis. Cancer Cell 6(5):447–458
Sodir NM et al (2011) Endogenous Myc maintains the tumor microenvironment. Genes Dev 25(9):907–916
Boshoff C, Weiss R (2002) AIDS-related malignancies. Nat Rev Cancer 2(5):373–382
Ray-Coquard I et al (2009) Lymphopenia as a prognostic factor for overall survival in advanced carcinomas, sarcomas, and lymphomas. Cancer Res 69(13):5383–5391
Soucek L et al (2007) Mast cells are required for angiogenesis and macroscopic expansion of Myc-induced pancreatic islet tumors. Nat Med 13(10):1211–1218
Shiao SL, Coussens LM (2010) The tumor-immune microenvironment and response to radiation therapy. J Mammary Gland Biol Neoplasia 15(4):411–421
Obeid M et al (2007) Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med 13(1):54–61
Restifo NP (2010) Can antitumor immunity help to explain “oncogene addiction”? Cancer Cell 18(5):403–405
Acosta JC et al (2008) Chemokine signaling via the CXCR2 receptor reinforces senescence. Cell 133(6):1006–1018
Beatty G, Paterson Y (2001) IFN-gamma-dependent inhibition of tumor angiogenesis by tumor-infiltrating CD4+ T cells requires tumor responsiveness to IFN-gamma. J Immunol 166(4):2276–2282
Kuilman T et al (2008) Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 133(6):1019–1031
Muller-Hermelink N et al (2008) TNFR1 signaling and IFN-gamma signaling determine whether T cells induce tumor dormancy or promote multistage carcinogenesis. Cancer Cell 13(6):507–518
Li SS et al (2002) T lymphocyte expression of thrombospondin-1 and adhesion to extracellular matrix components. Eur J Immunol 32(4):1069–1079
Li SS et al (2006) Endogenous thrombospondin-1 is a cell-surface ligand for regulation of integrin-dependent T-lymphocyte adhesion. Blood 108(9):3112–3120
Young GD, Murphy-Ullrich JE (2004) The tryptophan-rich motifs of the thrombospondin type 1 repeats bind VLAL motifs in the latent transforming growth factor-beta complex. J Biol Chem 279(46):47633–47642
Becker C et al (2004) TGF-beta suppresses tumor progression in colon cancer by inhibition of IL-6 trans-signaling. Immunity 21(4):491–501
Tang B et al (2007) Transforming growth factor-beta can suppress tumorigenesis through effects on the putative cancer stem or early progenitor cell and committed progeny in a breast cancer xenograft model. Cancer Res 67(18):8643–8652
Muranski P, Restifo NP (2009) Adoptive immunotherapy of cancer using CD4(+) T cells. Curr Opin Immunol 21(2):200–208
Gonzalez-Angulo AM, Hortobagyi GN, Ellis LM (2011) Targeted therapies: peaking beneath the surface of recent bevacizumab trials. Nat Rev Clin Oncol 8(6):319–320
Wrzesinski C et al (2010) Increased intensity lymphodepletion enhances tumor treatment efficacy of adoptively transferred tumor-specific T cells. J Immunother 33(1):1–7
Tran PT et al (2011) Survival and death signals can predict tumor response to therapy after oncogene inactivation. Sci Transl Med 3(103):103ra99
Califano A (2011) Striking a balance between feasible and realistic biological models. Sci Transl Med 3(103):103ps39
Willmann JK et al (2008) Molecular imaging in drug development. Nat Rev Drug Discov 7(7):591–607
Pysz MA, Gambhir SS, Willmann JK (2010) Molecular imaging: current status and emerging strategies. Clin Radiol 65(7):500–516
Massoud TF, Gambhir SS (2003) Molecular imaging in living subjects: seeing fundamental biological processes in a new light. Genes Dev 17(5):545–580
Nguyen QD, Aboagye EO (2010) Imaging the life and death of tumors in living subjects: preclinical PET imaging of proliferation and apoptosis. Integr Biol 2(10):483–495
Michalski MH, Chen X (2011) Molecular imaging in cancer treatment. Eur J Nucl Med Mol Imaging 38(2):358–377
Acknowledgements
The authors would like to acknowledge current members of the Felsher laboratory for critical discussion and previous members who have contributed to characterizing various models of oncogene addiction.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Bellovin, D.I., Das, B., Felsher, D.W. (2013). Tumor Dormancy, Oncogene Addiction, Cellular Senescence, and Self-Renewal Programs. In: Enderling, H., Almog, N., Hlatky, L. (eds) Systems Biology of Tumor Dormancy. Advances in Experimental Medicine and Biology, vol 734. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-1445-2_6
Download citation
DOI: https://doi.org/10.1007/978-1-4614-1445-2_6
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-1444-5
Online ISBN: 978-1-4614-1445-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)