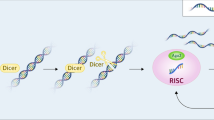
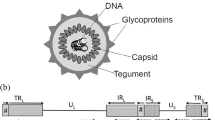
Abstract
In the age of extensive global traffic systems, the close neighborhood of man and livestock in some regions of the world, as well as inadequate prevention measures and medical care in poorer countries, greatly facilitates the emergence and dissemination of new virus strains. The appearance of avian influenza viruses that can infect humans, the spread of the severe acute respiratory syndrome (SARS) virus, and the unprecedented raging of human immunodeficiency virus (HIV) illustrate the threat of a global virus pandemic. In addition, viruses like hepatitis B and C claim more than one million lives every year for want of efficient therapy. Thus, new approaches to prevent virus propagation are urgently needed. Antisense strategies are considered a very attractive means of inhibiting viral replication, as oligonucleotides can be designed to interact with any viral RNA, provided its sequence is known. The ensuing targeted destruction of viral RNA should interfere with viral replication without entailing negative effects on ongoing cellular processes. In this review, we will give some examples of the employment of antisense oligonucleotides, ribozymes, and RNA interference strategies for antiviral purposes. Currently, in spite of encouraging results in preclinical studies, only a few antisense oligonucleotides and ribozymes have turned out to be efficient antiviral compounds in clinical trials. The advent of RNA interference now seems to be refueling hopes for decisive progress in the field of therapeutic employment of antisense strategies.
Chapter PDF
Similar content being viewed by others
References
Agrawal S, Kandimalla ER (2004) Antisense and siRNA as agonists of toll-like receptors. Nat Biotechnol 22:1533–1537
Arias F, Dector MA, Segovia L, Lopez T, Camacho M, Isa P, Espinosa R, Lopez S (2004) RNA silencing of rotavirus gene expression. Virus Res 102:43–51
Arteaga HJ, Hinkula J, van Dijk-Härd I, Dilber MS, Wahren B, Christensson B, Mohamed AJ, Smith CIE (2003) Choosing CCR5 or Rev siRNA in HIV-1. Nat Biotechnol 21:230–231
Arzumanov A, Walsh AP, Rajwanshi VK, Kumar R, Wengel J, Gait MJ (2001) Inhibition of HIV-1 Tat-dependent trans activation by steric block chimeric 2′-O-methyl/LNA oligoribonucleotides. Biochemistry 40:14645–14654
Bai J, Banda N, Lee NS, Rossi J, Akkina R (2002) RNA-based anti-HIV gene therapeutic constructs in SCID-hu mouse model. Mol Ther 6:770–782
Banerjea A, Li M-J, Bauer G, Remling L, Lee N-S, Rossi J, Akkina R (2003) Inhibition of HIV-1 by lentiviral vector-transduced siRNAs in T lymphocytes differentiated in SCID-hu mice and CD34+ progenitor cell-derived macrophages. Mol Ther 8:62–71
Barik S (2004) Control of nonsegmented negative-stand RNA virus replication by siRNA. Virus Res 102:27–35
Beigelman L, McSwiggen JA, Draper KG, Gonzalez C, Jensen K, Karpeisky AM, Modak AS, Matulic-Adamic J, DiRenzo AB, Haeberli P, Sweedler D, Tracz D, Grimm S, Wincott FE, Thackaray VG, Usman N (1995) Chemical modification of hammerhead ribozymes. JBiol Chem 270:25702–25708
Bitko V, Barik S (2001) Phenotypic silencing of cytoplasmic genes using sequence-specific double-stranded short interfering RNA and its application in the reverse genetics of wild type negative-strand RNA viruses. BMC Microbiol 1:34
Bitko V, Musiyenko A, Shulyayeva O, Barik S (2005) Inhibition of respiratory viruses by nasally administered siRNA. Nat Med 11:50–55
Boden D, Pusch O, Lee F, Tucker L, Ramratnam B (2003) Human immunodeficiency virus type 1 escapes from RNA interference. J Virol 77:11531–11535
Bridge AJ, Pebemard S, Ducraux A, Nicoulaz L, Iggo R (2003) Induction of an interferon response by RNAi vectors in mammalian cells. Nat Genet 34:263–264
Brummelkamp TR, Bernards R, Agami R (2002) A system for stable expression of short interfering RNAs in mammalian cells. Science 296:550–553
Coburn GA, Cullen BR (2002) Potent and specific inhibition of human immunodeficiency virus type 1 replication by RNA interference. J Virol 76:9225–9231
Crooke ST (2004) Progress in antisense technology. Annu Rev Med 55:61–95
Darfeuille F, Hansen JB, Orum H, Di Primo C, Toulme J-J (2004) LNA/DNA chimeric oligomers mimic RNA aptamers targeted to the TAR RNA element of HIV-1. Nucleic Acids Res 32:3101–3107
Das AT, Brummelkamp TR, Westerhout EM, Vink M, Madiredjo M, Bernards R, Berkhout B (2004) Human immunodeficiency virus type 1 escapes from RNA interference-mediated inhibition. J Virol 78:2601–2605
De Beuckelaer A, Fürste J-P, Gruszecka M, Wittmann-Liebold B, Erdmann VA (1999) Selection of RNA aptamers that bind to a peptide of the canyon region of human rhinovirus 14. In: Wagner E et al. (eds) Proceedings of the International Congress on Endocytobiology, Symbiosis and Biomedicine, Freiburg im Breisgau, Germany, 5–9 April 1998, Endocytobiology, VII. “From Symbiosis to Eukaryotism”. Freiburg, pp 565–577
De Clercq E (2004) Antivirals and antiviral strategies. Nat Rev Microbiol 2:704–720
Doench JG, Sharp PA (2004) Specificity of microRNA target selection in translational repression. Genes Dev 18:504–511
Dorsett Y, Tuschl T (2004) siRNAs: applications in functional genomics and potential as therapeutics. Nat Rev Drug Discov 3:318–329
Doudna JA, Cech TR (2002) The chemical repertoire of natural ribozymes. Nature 418:222–228
Eckstein F (2000) Phosphorothioate oligonucleotides: what is their origin and what is unique about them? Antisense Nucleic Acid Drug Dev 10:117–121
Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T (2001) Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 411:494–498
Elmén J, Zhang H-Y, Zuber B, Ljungberg K, Wahren B, Wahlestedt C, Liang Z (2004) Locked nucleic acids containing antisense oligonucleotides enhance inhibition of HIV-1 genome dimerization and inhibit virus replication. FEBS Lett 578:285–290
Enserink M (2004) Looking the pandemic in the eye. Science 306:392–394
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC (1998) Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391:806–811
Ge Q, McManus MT, Nguyen T, Shen C-H, Sharp PA, Eisen HN, Chen J (2003) RNA interference of influenza virus production by directly targeting mRNA for degradation and indirectly inhibiting all viral transcription. Proc Natl Acad Sci USA 100:2718–2723
Ge Q, Filip L, Bai A, Nguyen T, Eisen HN, Chen J (2004) Inhibition of influenza virus productionin virus-infected mice by RNA interference. Proc Natl Acad Sci USA 101:8676–8681
Gitlin L, Karelsky S, Andino R (2002) Short interfering RNA confers intracellular antiviral immunity in human cells. Nature 418:430–434
Goila R, Banerjea A (1998) Sequence specific cleavage of the HIV-1 coreceptor CCR5 gene by a hammer-head ribozyme and a DNA-enzyme: inhibition of the coreceptor function by a DNA-enzyme. FEBS Lett 435:233–238
Grunweller A, Wyszko E, Bieber B, Jahnel R, Erdmann VA, Kurreck J (2003) Comparison of different antisense strategies in mammalian cells using locked nucleic acids, 2′O-methyl RNA, phosphorothioates and siRNA. Nucleic Acids Res 31:3185–3193
Haasnoot PCJ, Cupac D, Berkhout B (2003) Inhibition of virus replication by RNA interference. J Biomed Sci 10:607–616
Haley B, Zamore PD (2004) Kinetic analysis of the RNAi enzyme complex. Nat Struct Mol Biol 11:599–606
Hannon GJ, Rossi JJ (2004) Unlocking the potential of the human genome with RNA interference. Nature 431:371–377
Hu W-Y, Myers CP, Kilzer JM, Pfaff SL, Bushman FD (2002) Inhibition of retroviral pathogenesis by RNA interference. Curr Biol 12:1301–1311
Jackson AL, Bartz SR, Schelter J, Kobayashi SV, Burchard J, Mao M, Li B, Cavet G, Linsey PS (2003) Expression profiling reveals off-target gene regulation by RNAi. Nat Biotechnol 21:635–637
Jacque J-M, Triques K, Stevenson M (2002) Modulation of HIV-1 replication by RNA interference. Nature 418:435–438
Jepsen JS, Wengel J (2004) LNA-antisense rivals siRNA for gene silencing. Curr Opin Drug Discov Devel 7:188–194
Jing N, Xu X (2001) Rational drug design of DNA oligonucleotides as HIV inhibitors. Curr Drug Targets 1:79–90
Khvorova A, Lescoute A, Westhof E, Jayasena SD (2003a) Sequence elements outside the hammerhead ribozyme catalytic core enable intracellular activity. Nat Struct Biol 10:708–712
Khvorova A, Reynolds A, Jayasena SD (2003b) Functional siRNAs and miRNAs exhibit strand bias. Cell 115:209–216
Klein C, Bock CT, Wedemeyer H, Wustefeld T, Locarnini S, Dienes HP, Kubicka S, Manns MP, Trautwein C (2003) Inhibition of hepatitis B virus replication in vivo by nucleoside analogues and siRNA. Gastroenterology 125:9–18
Kretschmer-Kazemi Far R, Sczakiel G (2003) The activity of siRNA in mammalian cells is related to structural target accessibility: a comparison with antisense oligonucleotides. Nucleic Acids Res 31:4417–4424
Kühl U, Pauschinger M, Schwimmbeck PL, Seeberg B, Lober C, Noutsias M, Poller W, Schultheiss H-P (2003) Interferon-beta treatment eliminates cardiotropic viruses and improves left ventricular function in patients with myocardial persistence of viral genomes and left ventricular dysfunction. Circulation 107:2793–2798
Kurreck J (2003) Antisense technologies: improvement through novel chemical modifications. Eur J Biochem 270:1628–1644
Kurreck J, Wyszko E, Gillen C, Erdmann VA (2002) Design of antisense oligonucleotides stabilized by locked nucleic acids. Nucleic Acids Res 30:1911–1918
Levin AA (1999) A review of issues in the pharmacokinetics and toxicology of phosphorothioate antisense oligonucleotides. Biochim Biophys Acta 1489:69–84
Macejak D, Jensen KL, Jamison S, Domenico K, Roberts EC, Chaudhary N, von Carlowitz I, Bellon L, Tong MJ, Conrad A, Pavco PA, Blatt LM (2000) Inhibition of Hepatitis C Virus (HCV)-RNA-dependent translation and replication of a chimeric HCV Poliovirus using synthetic stabilized ribozymes. Hepatology 31:769–776
Martinand-Mari, C, Lebleu B, Robbins I (2003) Oligonucleotide-based strategies to inhibit human hepatitis C virus. Oligonucleotides 13:539–548
McCaffrey AP, Meuse L, Pham TT, Conklin DS, Hannon GJ, Kay MA (2002) RNA interference in adult mice. Nature 418:38–39
McCaffrey AP, Nakai H, Pandey K, Huang Z, Salazar FH, Xu H, Wieland SF, Marion PL, Kay MA (2003) Inhibition of hepatitis B virus inmice by RNA interference. Nat Biotechnol 21:639–644
McKnight KL, Heinz BA (2003) RNA as a target for developing antivirals. Antivir Chem Chemother 14:61–73
Meister G, Tuschl T (2004) Mechanisms of gene silencing by double-stranded RNA. Nature 431:343–349
Michienzi A, Castonotto D, Lee N, Li S, Zaia J, Rossi JJ (2003) RNA-mediated inhibition of HIV in a gene therapy setting. Ann N Y Acad Sci 1002:63–71
Mittal V (2004) Improving the efficiency of RNA interference in mammals. Nat Rev Genet 5:355–365
Ngok FK, Mitsuyasu RT, Macpherson JL, Boyd MP, Symonds GP, Amado RG (2004) Clinical gene therapy research utilizing ribozymes. Methods Mol Biol 252:581–598
Novina CD, Murray MF, Dykxhoorn DM, Beresford PJ, Riess J, Lee S-K, Collman RG, Lieberman J, Shankar P, Sharp PA (2002) siRNA-directed inhibition of HIV-1 infection. Nat Med 8:681–686
Ojwang JO, Hampel A, Looney DJ, Wong-Staal F, Rappaport J (1992) Inhibition of the human immunodeficiency virus type 1 expression by a hairpin ribozyme. Proc Natl Acad Sci USA 89:10802–10806
Pan W-H, Xin P, Morrey JD, Clawson GA (2004) A self-processing ribozyme cassette: utility against human papillomavirus 11 E6/E7 mRNA and hepatitis B virus. Mol Ther 9:596–606
Peracchi A (2004) Prospects for antiviral ribozymes and deoxyribozymes. Rev Med Virol 14:47–64
Phipps KM, Martinez A, Lu J, Heinz BA, Zhao G (2004) Small interfering RNA molecules as potential anti-human rhinovirus agents: in vitro potency, specificity and mechanism. Antiviral Res 61:49–55
Qin X-F, An DS, Chen ISY, Baltimore D (2003) Inhibiting HIV-1 infection in human T cells by lentiviral-mediated delivery of small interfering RNA against CCR5. Proc Natl Acad Sci USA 100:183–188
Radhakrishnan SK, Layden TJ, Gartel AL (2004) RNA interference as a new strategy against viral hepatitis. Virology 323:173–181
Randall G, Rice CM (2004) Interfering with hepatitis C virus RNA replication. Virus Res 102:19–25
Randall G, Grakoui A, Rice CM (2003) Clearance of replicating hepatitis C virus replicon RNAs in cell culture by small interfering RNAs. Proc Natl Acad Sci USA 100:235–240
Reynolds A, Leake D, Boese Q, Scaringe S, Marshall WS, Khvorova A (2004) Rational siRNA design for RNA interference. Nat Biotechnol 22:326–330
Ryther RCC, Flynt AS, Phillips III JA, Patton JG (2005) siRNA therapeutics: big potential from small RNAs. Gene Ther 12:5–11
Saleh M-C, Van Rij RP, Andino R (2004) RNA silencing in viral infections: insights from poliovirus. Virus Res 102:11–17
Santoro SW, Joyce GF (1997) A general purpose RNA-cleaving DNA enzyme. Proc Natl Acad Sci USA 94:4262–4266
Scacheri PC, Rozenblatt-Rosen O, Caplen NJ, Wolfsberg TG, Umayam L, Lee JC, Hughes CM, Shanmugam KS, Bhattacharjee A, Meyerson M, Collins FS (2004) Short interfering RNAs can induce unexpected and divergent changes in the levels of untargeted proteins in mammalian cells. Proc Natl Acad Sci USA 101:1892–1897
Schubert S, Kurreck J (2004) Ribozyme-and deoxyribozyme-strategies for medical applications. Curr Drug Targets 5:667–681
Schubert S, Gül DC, Grunert H-P, Zeichhardt H, Erdmann VA, Kurreck J (2003) RNA cleaving “10–23” DNAzymes with enhanced stability and activity. Nucleic Acids Res 31:5982–5992
Schubert S, Fürste JP, Werk D, Grunert H-P, Zeichhardt H, Erdmann VA, Kurreck J (2004) Gaining target access for deoxyribozymes. J Mol Biol 339:355–363
Schubert S, Grunert H-P, Zeichhardt H, Werk D, Erdmann VA, Kurreck J (2005a) Maintaining Inhibition: siRNA double expression vectors against coxsackieviral RNAs. J Mol Biol 346:457–465
Schubert S, Grünweller A, Erdmann VA, Kurreck J (2005b) Local RNA target structure influences siRNA efficacy: systematic analysis of intentionally designed binding regions. J Mol Biol 348:883–893
Schwarz DS, Hutvágner G, Du T, Xu Z, Aronin N, Zamore PD (2003) Asymmetry in the assembly of the RNAi enzyme complex. Cell 115:199–208
Seksek O, Bolard J (2004) Delivery agents for oligonucleotides. Methods Mol Biol 252:545–568
Semizarov D, Frost L, Sarthy A, Kroeger P, Halbert DN, Fesik SW (2003) Specificity of short interfering RNA determined through gene expression signatures. Proc Natl Acad Sci USA 100:6347–6352
Silva J, Chang K, Hannon GJ, Rivas FV (2004) RNA-interference-based functional genomics in mammalian cells: reverse genetics coming of age. Oncogene 23:8401–8409
Sioud M (2004) Therapeutic siRNAs. Trends Pharmacol Sci 25:22–28
Sledz CA, Holko M, deVeer MJ, Silverman RH, Williams BR (2003) Activation of the interferon system by short-interfering RNAs. Nat Cell Biol 5:834–839
Sohail M, Southern EM (2000) Selecting optimal antisense reagents. Adv Drug Deliv Rev 44:23–34
Soutschek J, Akinc A, Bramlage B, Charisse K, Constien R, Donoghue M, Elbashir S, Geick A, Hadwiger P, Harborth J, John M, Kesavan V, Lavine G, Pandey RK, Racie T, Rajeev KG, Röhl I, Toudjarska I, Wang G, Wuschko S, Bumcrot D, Koteliansky V, Limmer S, Manoharan M, Vornlocher H-P (2004) Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature 432:173–178
Sullenger BA, Gilboa E (2002) Emerging clinical applications of RNA. Nature 418:252–258
Takahashi H, Hamazaki H, Habu Y, Hayashi M, Abe T, Miyano-Kurosaki N, Takaku H (2004) A new modified DNA enzyme that targets influenza virus A mRNA inhibits viral infection in cultured cells. FEBS Lett 560:69–74
Thomas CE, Ehrhardt A, Kay MA (2003) Progress and problems with the use of viral vectors for gene therapy. Nat Rev Genet 4:346–358
Tompkins SM, Lo C-Y, Tumpey TM, Epstein SL (2004) Protection against lethal influenza virus challenge by RNA interference in vivo. Proc Natl Acad Sci USA 101:8682–8686
Uprichard SL, Boyd B, Althage A, Chisari FV (2005) Clearance of hepatitis B virus from the liver of transgenic mice by short hairpin RNAs. Proc Natl Acad Sci USA 102:773–778
Usman N, Blatt LM (2000) Nuclease-resistant synthetic ribozymes: developing a new class of therapeutics. J Clin Invest 106:1197–1202
Wilson JA, Jayasena S, Khvorova A, Sabatinos S, Rodrigue-Gervais IG, Arya S, Sarangi F, Harris-Brandts M, Beaulieu S, Richardson CD (2003) RNA interference blocks gene expression and RNA synthesis from hepatitis C replicons propagated in human liver cells. Proc Natl Acad Sci USA 100:2783–2788
Yuan J, Cheung PKM, Zhang H, Chau D, Yanagawa B, Cheung C, Luo H, Wang Y, Suarez A, McManus BM, Yang D(2004) A phosphorothioate antisense oligodeoxynucleotide specifically inhibits coxsackievirus B3 replication in cardiomyocytes and mouse hearts. Lab Invest 84:703–714
Zamecnik PC, Stephenson ML (1978) Inhibition of Rous sarcoma virus replication and cell transformation by a specific oligodeoxynucleotide. Proc Natl Acad Sci USA 75:280–284
Zhang J, Yamada O, Sakamoto T, Yoshida H, Iwai T, Matsushita Y, Shimamura H, Araki H, Shimotohno K (2004) Down-regulation of viral replication by adenoviral-mediated expression of siRNA against cellular cofactors for hepatitis C virus. Virology 320:135–143
Zhang W, Yang H, Kong X, Mohapatra S, San Juan-Vergara H, Hellermann G, Behera S, Singam R, Lockey RF, Mohapatra SS (2005) Inhibition of respiratory syncytial virus infection with intranasal siRNA nanoparticles targeting the viral NS1 gene. Nat Med 11:56–62
Zhang X, Xu Y, Ling H, Hattori T (1999) Inhibition of infection of incoming HIV-1 virus by RNA cleaving DNA enzyme. FEBS Lett 458:151–156
Zhou C, Bahner IC, Larson GP, Zaia JA, Rossi JJ, Kohn EB (1994) Inhibition of HIV-1 inhuman T-lymphocytes by retrovirally transduced anti-tat and rev hammerhead ribozymes. Gene 149:33–39
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2006 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Schubert, S., Kurreck, J. (2006). Oligonucleotide-Based Antiviral Strategies. In: Erdmann, V., Barciszewski, J., Brosius, J. (eds) RNA Towards Medicine. Handbook of Experimental Pharmacology, vol 173. Springer, Berlin, Heidelberg. https://doi.org/10.1007/3-540-27262-3_13
Download citation
DOI: https://doi.org/10.1007/3-540-27262-3_13
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-27261-8
Online ISBN: 978-3-540-27262-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)