Abstract
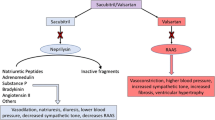
It has been known since the 1990s that long-term morbidity and mortality is improved in patients with heart failure with reduced ejection fraction (HFrEF) by treatments that target the renin–angiotensin–aldosterone system (RAAS). It has also long been thought that enhancement of the activity of natriuretic peptides (NPs) could potentially benefit patients with HFrEF, but multiple attempts to realize this benefit had failed over the years – until 2014, when a large, phase III, randomized, controlled clinical trial (PARADIGM-HF) was completed comparing sacubitril/valsartan with enalapril, a well-established treatment for HFrEF. Sacubitril/valsartan (formerly known as LCZ696) is a first-in-class angiotensin receptor neprilysin inhibitor (ARNI) that simultaneously suppresses RAAS activation through blockade of angiotensin II type 1 receptors and enhances vasoactive peptides including NPs through inhibition of neprilysin, the enzyme responsible for their degradation. In PARADIGM-HF, patients with HFrEF treated with sacubitril/valsartan had 20% less risk for cardiovascular death or hospitalization for heart failure (the primary endpoint), 20% less risk for cardiovascular death, 21% less risk for first hospitalization for heart failure, and 16% less risk for death from any cause, compared with enalapril (all p < 0.001). Concerning tolerability, the sacubitril/valsartan group had higher proportions of patients with hypotension and nonserious angioedema but lower proportions with renal impairment, hyperkalemia, and cough, compared with the enalapril group. The use of sacubitril/valsartan has been endorsed by the latest heart failure treatment guidelines in Europe and the USA. This chapter reviews the discoveries, scientific reasoning, and clinical evidence that led to the development of sacubitril/valsartan, the first novel therapy in a new drug class to improve survival in HFrEF in the last 15 years.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- ACE:
-
Angiotensin-converting enzyme
- ACEI:
-
Angiotensin-converting enzyme inhibitor
- AE:
-
Adverse event
- ANP:
-
Atrial natriuretic peptide
- ARB:
-
Angiotensin receptor blocker
- ARNI:
-
Angiotensin receptor neprilysin inhibitor
- AT1 :
-
Angiotensin II type 1
- AUC:
-
Area under the plasma concentration curve
- bid:
-
Twice daily
- BNP:
-
B-type natriuretic peptide
- BP:
-
Blood pressure
- cGMP:
-
Cyclic guanosine monophosphate
- CI:
-
Confidence interval
- C max :
-
Maximum plasma concentration
- CNP:
-
C-type natriuretic peptide
- CSF:
-
Cerebrospinal fluid
- CV:
-
Cardiovascular
- ED:
-
Emergency department
- EF:
-
Ejection fraction
- eGFR:
-
Estimated glomerular filtration rate
- EMPHASIS-HF:
-
Eplerenone in Mild Patients Hospitalization and Survival Study in Heart Failure
- HbA1c:
-
Glycated hemoglobin
- HF:
-
Heart failure
- HFpEF:
-
Heart failure with preserved ejection fraction
- HFrEF:
-
Heart failure with reduced ejection fraction
- HR:
-
Hazard ratio
- hsTnT:
-
High sensitivity troponin T
- KCCQ:
-
Kansas City Cardiomyopathy Questionnaire
- LVEF:
-
Left ventricular ejection fraction
- MAGGIC:
-
Meta-analysis Global Group in Chronic Heart Failure
- MedDRA:
-
Medical Dictionary for Regulatory Activities
- MI:
-
Myocardial infarction
- NEP:
-
Neprilysin
- NEPI:
-
Neprilysin inhibitor
- NNT:
-
Number needed to treat
- NP:
-
Natriuretic peptide
- NPR:
-
Natriuretic peptide receptor
- NT-proBNP:
-
N-terminal pro B-type natriuretic peptide
- NYHA:
-
New York Heart Association
- OATP:
-
Organic anion transporter protein
- PARADIGM-HF:
-
Prospective comparison of ARNI with ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure
- PARAMOUNT:
-
Prospective comparison of ARNI with ARB on Management Of heart failUre with preserved ejectioN fraction
- PP:
-
Pulse pressure
- PRA:
-
Plasma renin activity
- PRC:
-
Plasma renin concentration
- RAAS:
-
Renin–angiotensin–aldosterone system
- SNS:
-
Sympathetic nervous system
- SOLVD:
-
Studies of Left Ventricular Dysfunction
- T max :
-
Time to peak concentration
References
Ando S, Rahman MA, Butler GC, Senn BL, Floras JS (1995) Comparison of candoxatril and atrial natriuretic factor in healthy men. Effects on hemodynamics, sympathetic activity, heart rate variability, and endothelin. Hypertension 26(6 Pt 2):1160–1166
Akahori M, Ayalasomayajula S, Langenickel T, Pal P, Zhou W, Sunkara G (2016) Pharmacokinetics after single ascending dose, food effect, and safety of sacubitril/valsartan (LCZ696), an angiotensin receptor and neprilysin inhibitor, in healthy Japanese subjects. Eur J Drug Metab Pharmacokinet [Epub ahead of print]. doi:10.1007/s13318-016-0354-1
Ayalasomayajula S, Jordaan P, Pal P et al (2015) Assessment of drug interaction potential between LCZ696, an angiotensin receptor neprilysin inhibitor, and digoxin or warfarin. Clin Pharmacol Biopharm 4:147
Ayalasomayajula S, Han Y, Langenickel T et al (2016a) In vitro and clinical evaluation of OATP-mediated drug interaction potential of sacubitril/valsartan (LCZ696). J Clin Pharm Ther 41(4):424–431
Ayalasomayajula S, Langenickel T, Chandra P et al (2016b) Effect of food on the oral bioavailability of the angiotensin receptor neprilysin inhibitor sacubitril/valsartan (LCZ696) in healthy subjects. Int J Clin Pharmacol Ther 54(12):1012–1018
Ayalasomayajula SP, Langenickel TH, Jordaan P et al (2016c) Effect of renal function on the pharmacokinetics of LCZ696 (sacubitril/valsartan), an angiotensin receptor neprilysin inhibitor. Eur J Clin Pharmacol 72(9):1065–1073
Ayalasomayajula S, Pan W, Han Y et al (2016d) Assessment of drug-drug interaction potential between atorvastatin and LCZ696, A novel angiotensin receptor neprilysin inhibitor, in healthy Chinese male subjects. Eur J Drug Metab Pharmacokinet [Epub ahead of print]
Baguet JP, Barone-Rochette G, Neuder Y (2009) Candesartan cilexetil in the treatment of chronic heart failure. Vasc Health Risk Manage 5(1):257–264
Bayes-Genis A, Morant-Talamante N, Lupon J (2016) Neprilysin and natriuretic peptide regulation in heart failure. Curr Heart Failure Rep 13(4):151–157
Bevan EG, Connell JM, Doyle J, Carmichael HA, Davies DL, Lorimer AR, McInnes GT (1992) Candoxatril, a neutral endopeptidase inhibitor: efficacy and tolerability in essential hypertension. J Hypertens 10(7):607–613
Bohm M, Refsgaard J, Ramires F et al (2015) Effect of the angiotensin receptor neprilysin inhibitor LCZ696 compared with enalapril according to systolic blood pressure in PARADIGM-HF [abstract no. P1794]. Eur J Heart Failure 17(Suppl 1):393
Burnett JC Jr (1999) Vasopeptidase inhibition: a new concept in blood pressure management. J Hypertens Suppl 17(1):S37–S43
Butler J, Braunwald E, Gheorghiade M (2014) Recognizing worsening chronic heart failure as an entity and an end point in clinical trials. JAMA 312(8):789–790
Cannon J, Shen L, Jhund P et al (2016) Dementia-related adverse events in PARADIGM-HF and other trials in heart failure with reduced ejection fraction. Eur J Heart Failure [Epub ahead of print]. doi:10.1002/ejhf.687.
Carr AA, Prisant LM (1996) Losartan: first of a new class of angiotensin antagonists for the management of hypertension. J Clin Pharmacol 36(1):3–12
Claggett B, Packer M, McMurray JJV et al (2015) Estimating the long-term treatment benefits of sacubitril-valsartan. N Engl J Med 373(23):2289–2290
Cleland JG, Swedberg K (1998) Lack of efficacy of neutral endopeptidase inhibitor ecadotril in heart failure. The international ecadotril multi-centre dose-ranging study investigators. Lancet 351(9116):1657–1658
Cohn JN (1996) The management of chronic heart failure. N Engl J Med 335(7):490–498
Cohn JN, Tognoni G (2001) A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med 345(23):1667–1675. doi:10.1056/NEJMoa010713
Cohn JN, Johnson G, Ziesche S et al (1991) A comparison of enalapril with hydralazine-isosorbide dinitrate in the treatment of chronic congestive heart failure. N Engl J Med 325(5):303–310
Colucci WS, Elkayam U, Horton DP et al (2000) Intravenous nesiritide, a natriuretic peptide, in the treatment of decompensated congestive heart failure. N Engl J Med 343(4):246–253
CONSENSUS (1987) Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). The CONSENSUS trial study group. N Engl J Med 316(23):1429–1435. doi:10.1056/NEJM198706043162301
Cubeddu LX (2009) Iatrogenic QT abnormalities and fatal arrhythmias: mechanisms and clinical significance. Curr Cardiol Rev 5(3):166–176
Damman K, Andersen K, Belohlavek J et al. (2015) Angiotensin receptor neprilysin inhibition and renal function in heart failure: results from PARADIGM-HF [abstract no. 3301]. Eur Heart J 36(Suppl 1):545
Daniels LB, Maisel AS (2007) Natriuretic peptides. J Am Coll Cardiol 50(25):2357–2368
de Bold AJ, Raymond JJ, Bencosme SA (1978) Atrial specific granules of the rat heart: light microscopic staining and histochemical reactions. J Histochem Cytochem 26(12):1094–1102
de Bold AJ, Borenstein HB, Veress AT, Sonnenberg H (1981) A rapid and potent natriuretic response to intravenous injection of atrial myocardial extract in rats. Life Sci 28(1):89–94
Desai AS, McMurray JJV, Packer M et al (2015) Effect of the angiotensin-receptor-neprilysin inhibitor LCZ696 compared with enalapril on mode of death in heart failure patients. Eur Heart J 36(30):1990–1997
Desai A, Claggett B, Packer M et al (2016a) Influence of sacubitril/valsartan (LCZ696) on 30-day readmission after heart failure hospitalization. J Am Coll Cardiol 68(3):241–248
Desai AS, Solomon S, Claggett B et al (2016b) Factors associated with noncompletion during the run-in period before randomization and influence on the estimated benefit of LCZ696 in the PARADIGM-HF trial. Circ Heart Failure 9(6)
Favrat B, Burnier M, Nussberger J, Lecomte JM, Brouard R, Waeber B, Brunner HR (1995) Neutral endopeptidase versus angiotensin converting enzyme inhibition in essential hypertension. J Hypertens 13(7):797–804
Flarakos J, Du Y, Bedman T et al (2016) Disposition and metabolism of [C] Sacubitril/Valsartan (formerly LCZ696) an angiotensin receptor neprilysin inhibitor, in healthy subjects. Xenobiotica 46(11):1–15
Gafford JT, Skidgel RA, Erdos EG, Hersh LB (1983) Human kidney “enkephalinase”, a neutral metalloendopeptidase that cleaves active peptides. Biochemistry 22(13):3265–3271
Gan L, Langenickel T, Petruck J et al (2016) Effects of age and sex on the pharmacokinetics of LCZ696, an angiotensin receptor neprilysin inhibitor. J Clin Pharmacol 56(1):78–86
Gori M, Senni M, Claggett B et al (2015) Effect of LCZ696 on urinary albumin excretion and relation to outcomes in patients with heart failure [abstract no. 3302]. Eur Heart J 36(Suppl 1):545
Gros C, Souque A, Schwartz JC, Duchier J, Cournot A, Baumer P, Lecomte JM (1989) Protection of atrial natriuretic factor against degradation: diuretic and natriuretic responses after in vivo inhibition of enkephalinase (EC 3.4.24.11) by acetorphan. Proc Natl Acad Sci U S A 86(19):7580–7584
Gu J, Noe A, Chandra P et al (2010) Pharmacokinetics and pharmacodynamics of LCZ696, a novel dual-acting angiotensin receptor-neprilysin inhibitor (ARNi). J Clin Pharmacol 50(4):401–414
Han Y, Ayalasomayajula S, Pan W et al (2016) Pharmacokinetics, safety and tolerability of sacubitril/valsartan (LCZ696) After single-dose administration in healthy Chinese subjects. Eur J Drug Metab Pharmacokinet [Epub ahead of print]. doi:10.1007/s13318-016-0328-3
Hawkridge AM, Heublein DM, Bergen HR III, Cataliotti A, Burnett JC Jr, Muddiman DC (2005) Quantitative mass spectral evidence for the absence of circulating brain natriuretic peptide (BNP-32) in severe human heart failure. Proc Natl Acad Sci U S A 102(48):17442–17447
Heeschen C, Hamm CW, Mitrovic V, Lantelme NH, White HD (2004) N-terminal pro-B-type natriuretic peptide levels for dynamic risk stratification of patients with acute coronary syndromes. Circulation 110(20):3206–3212
Holmes SJ, Espiner EA, Richards AM, Yandle TG, Frampton C (1993) Renal, endocrine, and hemodynamic effects of human brain natriuretic peptide in normal man. J Clin Endocrinol Metab 76(1):91–96
Jhund PS, Claggett B, Packer M et al (2014) Independence of the blood pressure lowering effect and efficacy of the angiotensin receptor neprilysin inhibitor, LCZ696, in patients with heart failure with preserved ejection fraction: an analysis of the PARAMOUNT trial. Eur J Heart Failure 16(6):671–677
Jhund P, Claggett B, Solomon S et al (2015a) Elevated high sensitivity troponin is associated with poorer outcomes in patients with heart failure and reduced by LCZ696 [abstract no. P214]. Eur Heart J 36(Suppl 1):22
Jhund PS, Fu M, Bayram E et al (2015b) Efficacy and safety of LCZ696 (sacubitril-valsartan) according to age: insights from PARADIGM-HF. Eur Heart J 36(38):2576–2584
Kahn JC, Patey M, Dubois-Rande JL et al (1990) Effect of sinorphan on plasma atrial natriuretic factor in congestive heart failure. Lancet 335(8681):118–119
Kang PM, Landau AJ, Eberhardt RT, Frishman WH (1994) Angiotensin II receptor antagonists: a new approach to blockade of the renin-angiotensin system. Am Heart J 127(5):1388–1401
Katz AM (2008) The “modern” view of heart failure: how did we get here? Circ Heart Failure 1(1):63–71
Kobalava Z, Kotovskaya Y, Averkov O et al (2016) Pharmacodynamic and pharmacokinetic profiles of sacubitril/valsartan (LCZ696) in patients with heart failure and reduced ejection fraction. Cardiovasc Ther 34(4):191–198
Kostis JB, Packer M, Black HR, Schmieder R, Henry D, Levy E (2004) Omapatrilat and enalapril in patients with hypertension: the omapatrilat cardiovascular treatment vs. enalapril (OCTAVE) trial. Am J Hypertens 17(2):103–111
Kristensen S, Preiss D, Jhund P et al (2016a) Risk related to pre–diabetes mellitus and diabetes mellitus in heart failure with reduced ejection fraction: insights from prospective comparison of ARNI With ACEI to determine impact on global mortality and morbidity in heart failure trial. Circ Heart Failure 9(1), e002560
Kristensen SL, Martinez F, Jhund PS et al (2016b) Geographic variations in the PARADIGM-HF heart failure trial. Eur Heart J 37(41):3167–3174
Ksander GM, Ghai RD, deJesus R et al (1995) Dicarboxylic acid dipeptide neutral endopeptidase inhibitors. J Med Chem 38(10):1689–1700
Kulmatycki KLT, Ng D, Pal P, Zhou W, Rajman I, Rebello S, Sunkara G, Chandra P (2014) Pharmacokinetics of single-dose LCZ696 in subjects with mild and moderate hepatic impairment. Clin Pharmacol Drug Dev 3(Suppl 1):21
Langenickel TH, Dole WP (2012) Angiotensin receptor-neprilysin inhibition with LCZ696: a novel approach for the treatment of heart failure. Drug Discovery Today Ther Strategies 9(4):e131–e139
Langenickel TH, Jordaan P, Petruck J et al (2016a) Single therapeutic and supratherapeutic doses of sacubitril/valsartan (LCZ696) do not affect cardiac repolarization. Eur J Clin Pharmacol 72(8):917–924
Langenickel TH, Tsubouchi C, Ayalasomayajula S et al (2016b) The effect of LCZ696 (sacubitril/valsartan) on amyloid-beta concentrations in cerebrospinal fluid in healthy subjects. Br J Clin Pharmacol 81(5):878–890
Lisy O, Jougasaki M, Schirger JA, Chen HH, Barclay PT, Burnett JC Jr (1998) Neutral endopeptidase inhibition potentiates the natriuretic actions of adrenomedullin. Am J Physiol 275(3 Pt 2):F410–F414
Liu Y, Studzinski C, Beckett T, Murphy MP, Klein RL, Hersh LB (2010) Circulating neprilysin clears brain amyloid. Mol Cell Neurosci 45(2):101–107
Makani H, Messerli FH, Romero J, Wever-Pinzon O, Korniyenko A, Berrios RS, Bangalore S (2012) Meta-analysis of randomized trials of angioedema as an adverse event of renin-angiotensin system inhibitors. Am J Cardiol 110(3):383–391
Massien C, Azizi M, Guyene TT, Vesterqvist O, Mangold B, Menard J (1999) Pharmacodynamic effects of dual neutral endopeptidase-angiotensin-converting enzyme inhibition versus angiotensin-converting enzyme inhibition in humans. Clin Pharmacol Ther 65(4):448–459
Matsue Y, Kagiyama N, Yoshida K et al (2015) Carperitide Is associated with increased in-hospital mortality in acute heart failure: a propensity score-matched analysis. J Card Failure 21(11):859–864
McKinnie SM Fischer C, Tran KM, Wang W, Mosquera F, Oudit GY, Vederas JC (2016) The metalloprotease neprilysin degrades and inactivates apelin peptides. Chembiochem 17(16):1495–1498
McMurray JJ, Ostergren J, Swedberg K et al (2003) Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function taking angiotensin-converting-enzyme inhibitors: the CHARM-added trial. Lancet 362(9386):767–771
McMurray JJ, Adamopoulos S, Anker SD et al (2012) ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The task force for the diagnosis and treatment of acute and chronic heart failure 2012 of the European society of cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur Heart J 33(14):1787–1847. doi:10.1093/eurheartj/ehs104
McMurray JJ, Packer M, Desai AS et al (2013) Dual angiotensin receptor and neprilysin inhibition as an alternative to angiotensin-converting enzyme inhibition in patients with chronic systolic heart failure: rationale for and design of the Prospective comparison of ARNI with ACEI to Determine Impact on Global Mortality and morbidity in Heart Failure trial (PARADIGM-HF). Eur J Heart Failure 15(9):1062–1073
McMurray JJ, Packer M, Desai AS et al (2014a) Baseline characteristics and treatment of patients in prospective comparison of ARNI with ACEI to determine impact on global mortality and morbidity in heart failure trial (PARADIGM-HF). Eur J Heart Failure 16(7):817–825
McMurray JJ, Packer M, Desai AS et al (2014b) Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med 371(11):993–1004
McMurray J, Packer M, Desai A et al (2015) A putative placebo analysis of the effects of LCZ696 on clinical outcomes in heart failure. Eur Heart J 36(7):434–439
Miller WL, Phelps MA, Wood CM, Schellenberger U, Van Le A, Perichon R, Jaffe AS (2011) Comparison of mass spectrometry and clinical assay measurements of circulating fragments of B-type natriuretic peptide in patients with chronic heart failure. Circ Heart Failure 4(3):355–360
Mistry NB, Westheim AS, Kjeldsen SE (2006) The angiotensin receptor antagonist valsartan: a review of the literature with a focus on clinical trials. Expert Opin Pharmacother 7(5):575–581
Mogensen U, Jhund P, Kober L et al (2016a) Outcomes in patients with prior myocardial infarction in PARADIGM-HF. Eur Heart J 37(Abstract Supplement):193
Mogensen U, Swedberg K, Kober L et al (2016b) Outcomes in patients with and without atrial fibrillation in PARADIGM-HF and effect of sacubitril/valsartan according to atrial fibrillation status. Eur Heart J 37(Abstract Supplement):193
Mozaffarian D, Benjamin EJ, Go AS et al (2015) Heart disease and stroke statistics–2015 update: a report from the American Heart Association. Circulation 131(4):e29–e322
Mukoyama M, Nakao K, Hosoda K et al (1991) Brain natriuretic peptide as a novel cardiac hormone in humans. Evidence for an exquisite dual natriuretic peptide system, atrial natriuretic peptide and brain natriuretic peptide. J Clin Invest 87(4):1402–1412
Muller P, Cohen T, de Gasparo M, Sioufi A, Racine-Poon A, Howald H (1994) Angiotensin II receptor blockade with single doses of valsartan in healthy, normotensive subjects. Eur J Clin Pharmacol 47(3):231–245
Nakao K, Sugawara A, Morii N et al (1986) The pharmacokinetics of alpha-human atrial natriuretic polypeptide in healthy subjects. Eur J Clin Pharmacol 31(1):101–103
Niederkofler EE, Kiernan UA, O’Rear J et al (2008) Detection of endogenous B-type natriuretic peptide at very low concentrations in patients with heart failure. Circ Heart Failure 1(4):258–264
Niemi M (2007) Role of OATP transporters in the disposition of drugs. Pharmacogenomics 8(7):787–802
Northridge DB, Jardine AG, Alabaster CT et al (1989) Effects of UK 69 578: a novel atriopeptidase inhibitor. Lancet 2(8663):591–593
Northridge DB, Jardine A, Henderson E, Dilly SG, Dargie HJ (1992) Increased circulating atrial natriuretic factor concentrations in patients with chronic heart failure after inhibition of neutral endopeptidase: effects on diastolic function. Br Heart J 68(4):387–391
O’Connor CM, Starling RC, Hernandez AF et al (2011) Effect of nesiritide in patients with acute decompensated heart failure. N Engl J Med 365(1):32–43
Ogawa T, de Bold AJ (2014) The heart as an endocrine organ. Endocr Connect 3(2):R31–R44
Okumura N, Jhund PS, Gong J et al (2016a) Effects of sacubitril/valsartan in the PARADIGM-HF trial (prospective comparison of ARNI with ACEI to determine impact on global mortality and morbidity in heart failure) according to background therapy. Circ Heart Failure 9(9):e003212
Okumura N, Jhund PS, Gong J et al (2016b) Importance of clinical worsening of heart failure treated in the outpatient setting: evidence from the prospective comparison of ARNI with ACEI to determine impact on global mortality and morbidity in heart failure trial (PARADIGM-HF). Circulation 133(23):2254–2262
Packer M, Califf RM, Konstam MA, Krum H, McMurray JJ, Rouleau JL, Swedberg K (2002) Comparison of omapatrilat and enalapril in patients with chronic heart failure: the omapatrilat versus enalapril randomized trial of utility in reducing events (OVERTURE). Circulation 106(8):920–926
Packer M, Holcomb R, Abraham WT, Anker S, Dickstein K, Filippatos G et al (2016) Rationale for and design of the TRUE-AHF trial: the effects of ularitide on the short-term clinical course and long-term mortality of patients with acute heart failure. Eur J Heart Fail [Epub ahead of print]. doi:10.1002/ejhf.698
Packer M, McMurray JJV, Desai AS (2015) Angiotensin receptor neprilysin inhibition compared with enalapril on the risk of clinical progression in surviving patients with heart failure. Circulation 131(1):54–61
Pfeffer MA, McMurray JJ, Velazquez EJ et al (2003) Valsartan, captopril, or both in myocardial infarction complicated by heart failure, left ventricular dysfunction, or both. N Engl J Med 349(20):1893–1906
PI (2015) Entresto Prescribing Information. Highlights of prescribing information. US FDA http://www.pharma.us.novartis.com/product/pi/pdf/entresto.pdf. Accessed 12 Jan 2016
Ponikowski P, Voors A, Anker S et al (2016) 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Failure 18(8):891–975
Potter LR (2011) Natriuretic peptide metabolism, clearance and degradation. FEBS J 278(11):1808–1817
Publication Committee, for the VMAC Investigators (2002) Intravenous nesiritide vs nitroglycerin for treatment of decompensated congestive heart failure: a randomized controlled trial. JAMA 287(12):1531–1540
Rademaker MT, Charles CJ, Espiner EA, Nicholls MG, Richards AM, Kosoglou T (1998) Combined neutral endopeptidase and angiotensin-converting enzyme inhibition in heart failure: role of natriuretic peptides and angiotensin II. J Cardiovasc Pharmacol 31(1):116–125
Richards AM, Wittert GA, Crozier IG, Espiner EA, Yandle TG, Ikram H, Frampton C (1993) Chronic inhibition of endopeptidase 24.11 in essential hypertension: evidence for enhanced atrial natriuretic peptide and angiotensin II. J Hypertens 11(4):407–416
Roques BP, Fournie-Zaluski MC, Soroca E, Lecomte JM, Malfroy B, Llorens C, Schwartz JC (1980) The enkephalinase inhibitor thiorphan shows antinociceptive activity in mice. Nature 288(5788):286–288
Rouleau JL, Pfeffer MA, Stewart DJ et al (2000) Comparison of vasopeptidase inhibitor, omapatrilat, and lisinopril on exercise tolerance and morbidity in patients with heart failure: IMPRESS randomised trial. Lancet 356(9230):615–620
Ruilope LM, Dukat A, Bohm M, Lacourciere Y, Gong J, Lefkowitz MP (2010) Blood-pressure reduction with LCZ696, a novel dual-acting inhibitor of the angiotensin II receptor and neprilysin: a randomised, double-blind, placebo-controlled, active comparator study. Lancet 375(9722):1255–1266
Sanchez-Borges M, Gonzalez-Aveledo LA (2010) Angiotensin-converting enzyme inhibitors and angioedema. Allergy Asthma Immunol Res 2(3):195–198
Sebastian JL, McKinney WP, Kaufman J, Young MJ (1991) Angiotensin-converting enzyme inhibitors and cough. Prevalence in an outpatient medical clinic population. Chest 99(1):36–39
Senni M, McMurray JJV, Wachter R et al (2016) Initiating sacubitril/valsartan (LCZ696) in heart failure: results of TITRATION, a double-blind, randomized comparison of two uptitration regimens. Eur J Heart Failure 18(9):1193–1202
Seymour AA, Swerdel JN, Abboa-Offei B (1991) Antihypertensive activity during inhibition of neutral endopeptidase and angiotensin converting enzyme. J Cardiovasc Pharmacol 17(3):456–465
Seymour AA, Asaad MM, Lanoce VM, Fennell SA, Cheung HS, Rogers WL (1993) Inhibition of neutral endopeptidase 3.4.24.11 in conscious dogs with pacing induced heart failure. Cardiovasc Res 27(6):1015–1023
Seymour AA, Abboa-Offei BE, Smith PL, Mathers PD, Asaad MM, Rogers WL (1995) Potentiation of natriuretic peptides by neutral endopeptidase inhibitors. Clin Exp Pharmacol Physiol 22(1):63–69
Shi J, Wang X, Nguyen J, Wu AH, Bleske BE, Zhu HJ (2016) Sacubitril is selectively activated by carboxylesterase 1 (CES1) in the liver and the activation is affected by CES1 genetic variation. Drug Metab Dispos 44(4):554–559
Simpson J, Jhund PS, Silva Cardoso J et al (2015) Comparing LCZ696 with enalapril according to baseline risk using the MAGGIC and EMPHASIS-HF risk scores: an analysis of mortality and morbidity in PARADIGM-HF. J Am Coll Cardiol 66(19):2059–2071
Solomon SD, Zile M, Pieske B et al (2012) The angiotensin receptor neprilysin inhibitor LCZ696 in heart failure with preserved ejection fraction: a phase 2 double-blind randomised controlled trial. Lancet 380(9851):1387–1395
Solomon SD, Claggett B, Desai AS et al (2016a) Influence of ejection fraction on outcomes and efficacy of sacubitril/valsartan (LCZ696) in heart failure with reduced ejection fraction: the prospective comparison of ARNI with ACEI to determine impact on global mortality and morbidity in heart failure (PARADIGM-HF) trial. Circ Heart Failure 9(3), e002744
Solomon SC, Packer M, Desai A, Zile MR, Swedberg K, Rouleau J, Shi V, Lefkowitz M, McMurray JJ (2016b) Efficacy of sacubitril/valsartan relative to a prior decompensation The PARADIGM-HF trial. JACC Heart Failure 4:816. doi:10.1016/j.jchf.2016.05.002
SOLVD (1991) Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med 325(5):293–302. doi:10.1056/NEJM199108013250501
Stephenson SL, Kenny AJ (1987) The hydrolysis of alpha-human atrial natriuretic peptide by pig kidney microvillar membranes is initiated by endopeptidase-24.11. Biochem J 243(1):183–187
Sudoh T, Kangawa K, Minamino N, Matsuo H (1988) A new natriuretic peptide in porcine brain. Nature 332(6159):78–81
Sudoh T, Minamino N, Kangawa K, Matsuo H (1990) C-type natriuretic peptide (CNP): a new member of natriuretic peptide family identified in porcine brain. Biochem Biophys Res Commun 168(2):863–870
Trippodo NC, Fox M, Monticello TM, Panchal BC, Asaad MM (1999) Vasopeptidase inhibition with omapatrilat improves cardiac geometry and survival in cardiomyopathic hamsters more than does ACE inhibition with captopril. J Cardiovasc Pharmacol 34(6):782–790
Unger T, Gohlke P (1994) Converting enzyme inhibitors in cardiovascular therapy: current status and future potential. Cardiovasc Res 28(2):146–158
Vardeny O, Claggett B, Packer M et al (2016) Efficacy of sacubitril/valsartan vs. enalapril at lower than target doses in heart failure with reduced ejection fraction: the PARADIGM-HF trial. Eur J Heart Failure 18:1228. doi:10.1002/ejhf.580
Waeber BNJ, Brunner HR (1990) Angiotensin-converting enzyme inhibitors in hypertension. In: Laragh JH, Brenner BM (eds) Hypertension: pathophysiology. Diagnosis and management. Raven Press, New York, pp 2209–2232
Weber MA (2001) Vasopeptidase inhibitors. Lancet 358(9292):1525–1532. doi:10.1016/S0140-6736(01)06584-9
Williams J, Bristow M, Fowler M et al (1995) Guidelines for the evaluation and management of heart failure. Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on Evaluation and Management of Heart Failure). Circulation 92(9):2764–2784
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey Jr DE, Colvin MM et al (2016) 2016 ACC/AHA/HFSA focused update on new pharmacological therapy for heart failure: an update of the 2013 ACCF/AHA guideline for the Management of Heart Failure: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J Card Failure 134(13):e282–e293
Zile MR, Claggett BL, Prescott M, McMurray JJV, Packer M, Rouleau JL (2016) Prognostic implications of changes in N-Terminal pro-B-Type natriuretic peptide in patients with heart failure. J Am Coll Cardiol 68(22):2425–2436
Acknowledgments/Disclosures
Y. Khder, M. P. Lefkowitz, and V. Shi are employees of Novartis, the manufacturer of sacubitril/valsartan. The authors thank Roohi Chopra, Sreedevi Boggarapu, and Ronald B. Langdon (all of Novartis) for the writing and editorial support.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Khder, Y., Shi, V., McMurray, J.J.V., Lefkowitz, M.P. (2016). Sacubitril/Valsartan (LCZ696) in Heart Failure. In: Bauersachs, J., Butler, J., Sandner, P. (eds) Heart Failure. Handbook of Experimental Pharmacology, vol 243. Springer, Cham. https://doi.org/10.1007/164_2016_77
Download citation
DOI: https://doi.org/10.1007/164_2016_77
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-59658-7
Online ISBN: 978-3-319-59659-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)