Abstract
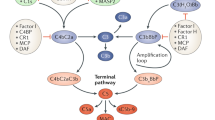
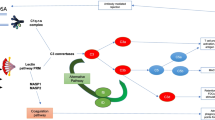
Experimental and clinical observations suggest that the xenograft rejection is based on two mechanisms. In animal species without natural antibodies, a mechanism similar to that in the allogeneic situation is responsible for the rejection. On the other hand, in species combinations where preformed natural antibodies (PNAB) directed against xenogeneic antigens are present, a hyper-acute rejection is typical. The principle features of this hyperacute xenogeneic renal rejection (HXAR) are a rapid increase of arterial resistance, cessation of the organ’s microcirculation within minutes, extravasation of blood and hematuria and cessation of urine production. Up to now, no reliable method to suppress this HXAR has been developed. Therefore, the major interest still lies either in the elimination of preformed antibodies or in blocking the major response of humoral mediators of which platelet-activating factor (PAF, PAF-acether) is supposed to be one. Recently, it was shown that PAF-acether does participate in transplant rejection. The first association with hyperacute rejection was suggested by Camussi et al. (1). In this reaction, PAF is regarded as a potent mediator of inflammatory reactions because of its broad range of biological activities. Commonly, the i.v. injection of PAF into different animal species is followed by hypotension, pulmonary hypertension, bronchoconstriction and increase of vascular permeability as systemic effects. The cellular sources of PAF are polymorphonuclear leukocytes (PMN), mast cells, platelets and endothelial cells. PAF is released from these cells when stimulated but also induces its own activation. Neutrophils and eosinophils participate in endothelial cell injury, e.g. by secreting proteases which destroy the basal membrane and activate the complement cascade. The close relationship between PAF synthesis and endothelial cell-dependent neutrophil adherence as well as the fact that PAF directly stimulates PMN activation and adhesion suggest that PAF may directly participate in transplant rejection (2, 3). The first evidence for PAF involvement in renal immune injury comes from the observation that PAF is released during hyperacute allograft rejection in kidneys. The participation of platelets and leukocytes in xenograft rejection provides the basis for the rationale of our trial to use PAF-antagonists in organ xenotransplantation.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Camussi, G., Niesen, N., Tetta, C., Saunders, R.N. and Milgrom, F.: Release of platelet-activating factor from rabbit heart perfused in vitro by sera with transplantation alloantibodies. Transplantation 44, 113–118 (1987).
Ito, S., Camussi, G., Tetta, C., Milgrom, F. and Andres, G.: Hyperacute renal allograft rejection in the rabbit. Lab. Invest 51, 148–161 (1984).
Braquet, P., Touqui, L., Shen, T.Y. and Vergraftig, B.B.: Perspectives in platelet-activating factor research. Pharmacol. Rev. 39, 97–145 (1987).
Weir, D.M. (ed.): Handbook of Experimental Immunology, pp. 720–744. Blackwell Scientific, Oxford, Edinburgh (1967).
Hugli, T.E. Vallota, H.E. and Müller-Eberhard, H.J.: Purification and partial characterization of human and porcine C3a anaphylatoxin. J. Biol. Chem. 250, 1472–1478 (1975).
Gorski, J.P.: Quantitation of human complement fragment C4a in physiological fluids by competitive inhibition radioimmune assay. J. Immun. Methods 47, 61–73 (1981).
Conzen, P., Habazettl, H. Gutmann, R., Hobhahn, J., Goetz, A., Peter, K. and Brendel, W.: Thromboxane mediation of pulmonary hemodynamic responses after neutralization of heparin by protamine in pigs. Anesth. Analg. 68, 25–31 (1989).
Weber, K.H. and Heuer, H.O.: Hetrazepines as antagonists of platelet-activating factor. Med. Res. Rev. 9, 181–218 (1989).
Miyagawa, S., Hirose, H., Shirakura, R., Nakata, S., Naka, Y. et al.: The mechanism of discordant xenograft rejection. Transplant. Proc. 21, 520–521 (1989).
Reemtsma, K.: Xenografts. Transplant. Proc. 21 517–518 (1989).
Auchincloss, H.: Xenogeneic transplantation. Transplantation 46, 1–20 (1988).
Braquet, P.: BN 52021 and related compounds; a series of highly specific PAF-acether receptor antagonists. Prostaglandins 30,687–690 (1985).
Shen, T., Hwang, S., Chang, M., Doebber, T., Lam, M., Wu, M. and Wang, X.: The isolation and characterization of kadsurenone from haifentent (piper futokadsura) as an orally active specific receptor antagonist of platelet-activating factor. Int. J. Tissue Reac. 5, 339–345 (1985).
Terashita, Z., Tsushima, S., Yoshioka, Y., Nomura, H., Inada, Y. and Nishikana, K.: CV-3988, a specific antagonist of platelet-activating factor (PAF). Life Sci. 32, 1975–1977 (1983).
Araki, H. and Lefer, A.M.: Role of prostacyclin in the preservation of ischemic myocardial tissue in the perfused cat heart. Circulation Res. 47, 757–763 (1980).
Adachi, H., Rosengard, B.R. and Hutchins, G.M.: Effects of cyclosporine, aspirin, and cobra venom factor on discordant cardiac xenograft survival in rats. Transplant. Proc. 19, 1145 (1987).
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1991 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Saumweber, D.M., Bergmann, R., Hammer, C., Brendel, W. (1991). The relationship of eicosanoids and complement components to hyperacute xenogeneic rejection and its modification by the PAF-antagonist WEB 2086BS. In: Abouna, G.M., Kumar, M.S.A., White, A.G. (eds) Organ Transplantation 1990. Developments in Surgery, vol 11. Springer, Dordrecht. https://doi.org/10.1007/978-94-011-3386-9_62
Download citation
DOI: https://doi.org/10.1007/978-94-011-3386-9_62
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-010-5497-3
Online ISBN: 978-94-011-3386-9
eBook Packages: Springer Book Archive