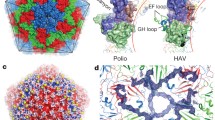
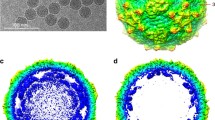
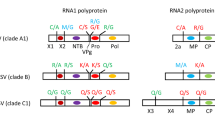
Abstract
Picornaviruses are a family of viruses which belong to the large group of positive-sense, single-stranded RNA viruses (RUECKERT 1996). It was realized 30 years ago that the product of the translation of the RNA genome of these viruses is proteolytically processed to yield the mature viral proteins (SUMMERS) and (MAIZEL 1968; KORANT 1972). Subsequently, it could be shown for two different picornaviruses that the processing enzyme is a specific, virally encoded proteinase (PELHAM 1978; GORBALENYA et al. 1979; KORANT et al. 1979; PALMENBERG et al. 1979). Once the amino-acid sequences of the viral proteinases became available, predictions were made concerning the structure of the picornaviral 3C proteinases (GROBALENYA et al. 1986; BAZAN and FLETTERICK 1988; GORBALENYA et al. 1989). These predictions were remarkable. Based on an analysis of several conserved sequence motifs within the amino-acid sequence of the 3C proteinases, it was suggested that these proteinases are structurally related to the chymotrypsin-like proteinases but with a cysteine residue as the active-site nucleophile. Crystal structures of 3C proteinases from two picornaviruses confirmed this prediction (ALLAIRE et al. 1994; MATTHEWS et al. 1994). At present, crystal structures of 3C proteinases from viruses, belonging to three different genera of the picornaviruses, have been published (MATTHEWS et al. 1994; BERGMANN et al. 1997; MOSIMANN et al. 1997).
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Allaire M, Chernaia MM, Malcolm BA, James MNG (1994) Picornaviral 3C cysteine proteinases have a fold similar to chymotrypsin-like serine proteinases. Nature 369:72–76
Andino R, Rieckhof GE, Trono DE, Baltimore D (1990) Substitutions in the protease (3CPro) can suppress a mutation in the 5’ noncoding region. J Virol 64:607–612
Andino R, Rickhof GE, Achacoso PL, Baltimore D (1993) Poliovirus RNA synthesis utilizes an RNP complex formed around the 5′-end of viral RNA. EMBO J 12:3587–3598
Andreoletti L, Hober D, Hober-Vandenberghe C, Belaich S, Vantyghem MC, Lefebvre J, Wattre P (1997) Detection of Coxsackie B virus RNA sequences in whole blood samples from adult patients at the onset of type I diabets mellitus. J Med Virol 52:121–127
Bazan JF, Fletterick RJ (1988) Viral cysteine proteinases are homologous to the trypsin-like family of serine proteinases: structural and functional implications. Proc Natl Acad Sci U S A 85:7872–7876
Belsham GJ (1993) Distinctive features of foot-and-mouth disease virus, a member of the picornavirus family; aspects of virus protein synthesis, protein processing and structure. Prog Biophys Mol Biol 60:241–260
Bergmann EM (1998) Hepatitis A virus picornain 3C. In: ai]Barrett AD, Rawlings NJ, Woesner F (eds) Handbook of proteolytic enzymes. Academic Press, London
Bergmann EM, James MNG (1999) Proteolytic enzymes of the viruses of the family Picornaviridae. In: Dunn B (ed) Proteinases of infectious agents. Academic Press, San Diego
Bergmann EM, Mosimann SC, Chernaia MM, Malcolm BA, James MNG (1997) The refined crystal structure of the 3C gene product from hepatitis A virus: specific proteinase activity and RNA recognition. J Virol 71:2436–2448
Bienz K, Egger D, Rasser Y, Bossart W (1983) Intracellular distribution of poliovirus proteins and the induction of virus-specific cytoplasmic structures. Virology 131:39–48
Bienz K, Egger D, Troxler M, Pasamontes L (1990) Structural organization of poliovirus RNA replication is mediated by viral proteins of the P2 genomic region. J Virol 64:1156–1163
Bode W, Huber R (1992) Natural protein proteinase inhibitors and their interactions with proteinases. Eur J Biochem 204:433–451
Borman AM, Kean KM (1997) Intact eukaryotic initiation factor 4G is required for hepatitis A virus internal initiation of translation. Virology 237:129–136
Brill BM, Kati WM, Montgomery D, Karwowski JP, Humphrey PE, Jackson M, Clement JJ, Kadam S, Chen RH, McAlpine JB (1997) Novel triterpene sulfates from fusarium compactum using a rhinovirus 3C protease inhibitor screen. J Antibiot (Tokyo) 49:541–546
Brocklehurst K, Watts AB, Patel M, Verma C, Thomas EW (1998) Cysteine proteinases. In: ai]Sinnott ML (ed) Comprehensive biological catalysis. Academic Press, London
Carthy CM, Yang D, Anderson DR, Wilson JE, McManus BM (1997) Myocarditis as systemic disease: new perspectives on pathogenesis. Clin Exp Pharmacol Physiol 24:997–1003
Center for Disease Control (1998) Progress towards global eradication of poliomyelitis 1997. MMWR Morb Mort Wkly Rep 47:414–419
Chang L-Y, Huang Y-C, Lin,TY (1998) Fulminant neurogenic pulmonary oedema with hand, foot and mouth diease. Lancet 352:367–368
Chotia C (1984) Principles that determine the structure of proteins. Annu Rev Biochem 53:537–572
Clarke IN, Lambden PR (1997) Viral zoonoses and food of animal origin: caliciviruses and human disease. Arch Virol Suppl 13:141–152
Cochi SL, Hull HF, Sutter RW, Wilfert CM, Katz SL (eds) (1997) Global poliomyelitis eradication initiative: status report. J Infect Dis 175[Suppl 1]
Cordingley MG, Callahan PL, Sardana VV, Garsky VM, Colonno RJ, (1990) Substrate requirements of human rhinovirus 3C protease for peptide cleavage in vitro. J Biol Chem 265:9062–9065
Couch RB (1996) Rhinoviruses. In: Fields BN, Knipe DM, Howley PM, Channock RM, Melnick JL, Monath TP, Roizmann BE, Straus SE (eds) Fields’ virology. Lippincott-Raven, Philadelphia
Donnelly MLL, Gani D, Flint M, Monaghan S, Ryan MD (1997) The cleavage activity of aphtho and cardiovirus 2A proteins. J Gen Virol 78:13–21
Dougherty WG, Semler BL (1993) Expression of virus-encoded proteinases: functional and structural similarities with cellular enzymes. Microbiol Rev 57:781–822
Dragovich PS, Webber SE, Babine, RE, Fuhrman SA, Patick AK, Matthews DA, Lee CA, Reich SH, Prins TJ, Marakovits JT, Littlefield ES, Zhou R, Tikhe J, Ford CE, Wallace MB, Meador JW III, Ferre RA, Brown EL, Binford SI, Harr JE, DeLisle DM, Worland ST (1998a) Structure-based design, synthesis and biological evalu-ation of irreversible human rhinovirus 3C protease inhibitors. 1. Michael acceptor structure-activity studies. J Med Chem 41:2806–2818
Dragovich PS, Webber SE, Babine, RE, Fuhrman SA, Patick AK, Matthews DA, Reich SH, Marakovits JT, Prins TJ, Zhou R, Tikhe J, Littlefield ES, Bleckman TM, Wallace MB, Little TL, Ford CE, Meador JW III, Ferre RA, Brown EL, Binford SI, DeLisle DM, Worland ST (1998b) Structure-based design, synthesis and biological evalu-ation of irreversible human rhinovirus 3C protease inhibitors. 2. Peptide structure-activity studies. J Med Chem 41:2819–2834
Gamarnik AV, Andino R (1997) Two functional complexes formed by KH domain con-taining proteins with the 5’ noncoding region of poliovirus RNA. RNA 3:882–892
Gonzalez P, Sanches A, Rivera P, Jimenez C, Hernandez F (1997) Rotavirus and Coro-navirus outbreak: etiology of annual diarrhea in Costa Rican children. Rev Biol Trop 45:898–991
Gorbalenya AE, Snijder EJ (1996) Viral cysteine proteinases. Perspect Drug Discov-ery Design 6:64–86
Gorbalenya AE, Svitkin YV, Kazachkov YA, Agol VI (1979) Encephalomyocarditis virus-specific polypeptide p22 is involved in the processing of the viral precursor polypeptide. FEBS Lett 108:1–5
Gorbalenya AE, Blinov VM, Donchenko AP (1986) Poliovirus-encoded proteinase 3C: a possible evolutionary link between cellular serine and cysteine proteinase fam-ilies. FEBS Lett 194:253–257
Gorbalenya AE, Donchenko AP, Blinov VM, Koonin EV (1989) Cysteine proteinases of positive strand RNA viruses and chymotrypsin-like serine proteinases: a dis-tinct protein superfamily with a common structural fold. FEBS Lett 243:103–114
Green KY (1997) The role of human caliciviruses in epidemic gastroenteritis. Arch Virol Suppl 13:153–165
Haghighat A, Svitkin Y, Novoa I, Küchler E, Skern T, Sonnenberg N (1996) The elF4G-eIF4E complex is the target for direct cleavage by the rhinovirus 2A proteinase. J Virol 70:8444–8450
Hämmerle T, Molla A, Wimmer E (1992) Mutational analysis of the proposed FG loop of poliovirus proteinase 3C identified amino acids that are necessary for 3CD cleavage and might be determinants of a function distinct from proteolytic activ-ity. J Virol 66:6028–6034
Hanecak R, Semler BL, Ariga H, Anderson CW, Wimmer E (1984) Expression of a cloned gene segment of poliovirus in E. coli: evidence for autocatalytic production of the viral proteinase. Cell 37:1063–1073
Harmon SA, Updike W, Xi-Ju J, Summers DF, Ehrenfeld E (1992) Polyprotein pro-cessing in cis and in trans by hepatitis A virus 3C protease cloned and expressed in E. coli. J Virol 66:5242–5247
Harris KS, Xiang W, Alexander LS, Lane WS, Paul AV, Wimmer E (1994) Interactions of poliovirus polypeptide 3CDPro with the 5’ and 3’ termini of the poliovirus genome. J Biol Chem 269:27004–27014
Hoetelmans RMW, Meenhorst PL, Mulder JW, Burger DM, Koks CHW, Beijnen JH (1997) Clinical pharmacology of HIV protease inhibitors: focus on saquinavir, ini-dinavir and ritonavir. Pharm World Sci 19:159–175
Hollinger FB, Ticehurst JR (1996) Hepatitis A virus. In: Fields BN, Knipe DM, Howley PM, Channock RM, Melnick JL, Monath TP, Roizmann BE, Straus SE (eds) Fields’ virology. Lippincott-Raven, Philadelphia
Holmes KV, Lai MC (1996) Coronaviridae: the viruses and their replication. In: Fields BN, Knipe DM, Howley PM, Channock RM, Melnick JL, Monath TP, Roizmann BE, Straus SE (eds) Fields’ virology. Lippincott-Raven, Philadelphia
James MNG (1993) Convergence of active-centre geometries among the proteolytic enzymes. In: Bond JS, Barrett AJ (eds) Proteolysis and protein turnover. Portland, London
Jewell DA, Swietnicki AW, Dunn, BM, Malcolm BA (1992) Hepatitis A virus 3C pro-teinase substrate specificity. Biochemistry 31:7862–7869
Jia X-Y, Summers DF, Ehrenfeld E (1993) Primary cleavage of the HAV capsid protein precursor in the middle of the proposed 2A coding region. Virology 193:515–519
Jungheim LN, Cohen JD, Johnson RB, Villareal EC, Wakulchik M, Loncharich RJ, Wang QM (1997) Inhibition of human rhinovirus 3C protease by homophthalim-ides. Bioorg Med Chem Lett 7:1589–1594
Kadam S, Poddig J, Humphrey P, Karwowski J, Jackson M, Tennent S, Fung L, Hochlowski J, Rasmussen R, McAlpine J (1994) Citrinin hydrate and radicinin: human rhinovirus 3C protease inhibitors discovered in a target-directed microbial screen. J Antibiot (Tokyo) 47:836–839
Kaldor SW, Hammond M, Dressman BA, Labus JM, Chadwell FW, Kline AD, Heinz BA (1995) Glutamine-derived aldehydes for the inhibition of human rhinovirus 3C proteinase. Bioorg Med Chem Lett 5:2021–2026
Kapikian AZ, Estes MK, Chanock RM (1996) Norwalk group of viruses. In: ai]Fields BN, Knipe DM, Howley PM, Channock RM, Melnick JL, Monath TP, Roizmann BE, Straus SE (eds) Fields’ virology, Lippincott-Raven, Philadelphia
Kong JS, Venkatraman S, Furness K, Nimkar S, Shepherd TA, Wang QM, Aubé J, Hanzlik R (1998) Synthesis and evaluation of peptidyl Michael acceptors that inactivate human rhinovirus 3C protease and inhibit virus replication. J Med Chem 41:2579–2587
Korant BD (1972) Cleavage of viral precursor proteins in vivo and in vitro. J Virol 10:751–759
Korant BD, Rizzo CJ (1997) The HIV protease and therapies for AIDS. Adv Exp Med Biol 421:279–284
Korant BD, Chow N, Lively M, Powers J (1979) Virus-specified protease in poliovirusinfected HeLa cells. Proc Natl Acad Sci USA 76:2992–2995
Kräusslich H-G, Wimmer E (1988) Viral proteinases. Annu Rev Biochem 57:701–754
Kusov YY, Gauss-Müller V (1997) In vitro RNA binding of the hepatitis A virus proteinase 3C (HAV 3CPro) to secondary structure elements within the 5′ terminus of the HAV genome. RNA 3:291–302
Landsteiner K, Popper E (1909) Übertraqung der Poliomyelitis acuta auf Affen. Z Immunitätsforschung Orig 2:377–390
Leong LEC, Walker PA, Porter AG (1993) Human rhinovirus 14 protease 3C (3CPro) binds specifically to the 5′-noncoding region of the viral RNA. J Biol Chem 268:25735–25739
Liu DX, Xu HY, Brown TD (1997) Proteolytic processing of the coronavirus infectious bronchitis virus 1a polyprotein: identification of a 10 kilodalton polypeptide and determination of its cleavage sites. J Virol 71:1814–1820
Loeffler F, Frosch P (1964) Report of the Commission for Research on Foot-and-Mouth Disease. In: ai]Hahon N (ed) Selected papers on virology. Prentice-Hall, Englewood Cliffs
Long LA, Orr DC, Cameron JM, Dunn BM, Kay J (1989) A consensus sequence for substrate hydrolysis by rhinovirus 3C proteinase. FEBS Lett 258:75–78
Lu XT, Sims AL, Denison MR (1998) Mouse hepatitis virus 3C-like proteinase cleaves a 22 kilodalton protein from the ORF 1a polyprotein in virus-infected cells and in vitro. J Virol 72:2265–2279
Makela MJ, Puhakka T, Runskanen O, Leinonen M, Saikku AP, Kimpimaki M, Blomquist S, Hyppia T, Arstilla P (1998) Viruses and bacteria in the etiology of the common cold. J Clin Microbiol 36:539–542
Malcolm BA, Lowe C, Shechosky S, McKay RT, Yang CC, Shah VJ, Simon RJ, Vederas JC, Santi DV (1995) Peptide aldehyde inhibitors of hepatitis A virus 3C proteinase. Biochemistry 34:8172–8179
Martfn-Alonso JM, Casais R, Boga JA, Parra F (1996) Processing of rabbit hemorrhagic disease virus polyprotein. J Virol 70:1261–1265
Martin A, Escriou N, Chao SF, Girard M, Lemon SM, Wychowski C (1995) Identification and site-directed mutagenesis of the primary (2A/2B) cleavage site of the hepatitis A virus polyprotein: functional impact on the infectivity of HAV RNA transcripts. Virology 213:213–222
Matthews DA, Smith WW, Ferre RA, Condon B, Budahazi G, Sisson W, Villafranca JE, Janson CA, McElroy HE, Gribskov CL, Worland S (1994) Structure of human rhi-novirus 3C protease reveals a trypsin-like polypeptide fold, RNA-binding site and means for cleaving precursor polyprotein. Cell 77:761–771
McCall JO, Kadam S, Katz L (1994) A high capacity microbial screen for inhibitors of human rhinovirus protease 3C. Biotechnology 12:1012–1016
McKendrick JE, Frormann S, Luo C, Semchuck P, Vederas JC, Malcolm BA (1998) Rapid mass spectrometric determination of preferred irreversible proteinase inhibitors in combinatorial libraries. Int J Mass Spectrom 176:113–124
Melnick JL (1996) Enteroviruses: Polioviruses Coxsackie viruses, echoviruses, and newer enteroviruses. In: Fields BN, Knipe DM, Howley PM, Channock RM, Melnick JL, Monath TP, Roizmann BE, Straus SE (eds) Fields’ virology. Lippincott-Raven, Philadelphia
Miller SD, Vanderlugt CL, Smith-Begolka W, Pao W, Yauch RL, Neville KL, Katz-Levy Y, Carrizosa A, Kim BS (1997) Persistent infection with Theiler’s virus leads to CNS autoimmunity via epitope spreading. Nat Med 3:1133–1136
Morris TS, Frormann S, Shechosky S, Lowe C, Lall MS, Gauss-Müller V, Purcell RH, Emerson SU, Vederas JC, Malcolm BA (1997) In vitro and ex vivo inhibition of hepatitis A virus 3C proteinase by a peptidyl monofluoromethyl ketone. Bioorg Med Chem Lett 5:797–807
Mosimann SC, Chernaia MM, Sia S, Plotch S, James MNG (1997) Refined X-ray crystallographic structure of the poliovirus 3C gene product. J Mol Biol 273:1032–1047
Ng LFP, Liu DX (1998) Identification of a 24-kDa polypeptide processed from the coronavirus infectious bronchitis virus 1a polyprotein by the 3C-like proteinase and determination of its cleavage sites. Virology 243:388–395
Nicklin MJ, Harris KS Pallai PV, Wimmer E (1988) Poliovirus proteinase 3C: largescale expression purification and specific cleavage activity on natural and synthetic substrates in vitro. J Virol 62:4586–4593
Niklasson B, Hornfeldt B, Lundman B (1998) Could myocarditis, insulin-dependent diabetes mellitus and Guillain-Barre syndrome be caused by one or more infectious agents carried by rodents? Emerg Infect Dis 4:187–193
Orr DC, Long AC, Kay J, Dunn BM, Cameron JM (1989) Hydrolysis of a series of synthetic peptide substrates by the human rhinovirus 14 3C proteinase cloned and expressed in E. coll.J Gen Virol 70:2931–2992
Pallai PV, Burkhardt F, Shoog M, Schreiner K, Bax P, Cohen KA, Hansen G, Palladino DE, Harris KS, Nicklin MJ, Wimmer E (1989) Cleavage of synthetic peptides by purified poliovirus 3C proteinase. J Biol Chem 264:9738–9741
Palmenberg AC (1990) Proteolytic processing of picornaviral polyprotein. Annu Rev Microbiol 44:602–623
Palmenberg AC, Rueckert RR (1982) Evidence for intramolecular self-cleavage of picornaviral replicase precursors. J Virol 41:244–249
Palmenberg AC, Pallansch MA, Rueckert RR (1979) Protease required for processing picornaviral coat protein residues resides in the viral replicase gene. J Virol 32:770–778
Palmenberg AC, Parks GD, Hall DJ, Ingraham RH, Seng TW, Pallai PV (1992) Proteolytic processing of the cardioviral P2 region: primary 2A/2B cleavage in clone derived precursors. Virology 190:754–762
Parsley TB, Towner JS, Blyn LB, Ehrenfeld E, Semler BL (1997) Poly (rC) binding protein 2 forms a ternary complex with the 5′-terminal sequences of poliovirus RNA and the viral 3CD proteinase. RNA 3:1124–1134
Pebody RG, Leino T, Ruuutu P, Kinnunen L, Davidkin I, Nohynek H, Leinikki P (1998) Foodborne outbreaks of hepatitis A in a low endemic country: an emerging problem? Epidemiol Infect 120:55–59
Pelham HRB (1978) Translation of encephalomyocarditis virus RNA in vitro yields an active proteolytic processing enzyme. Eur J Chem 85:457–462
Perona JJ, Craik CS (1995) Structural basis of substrate specificity in the serine proteinases. Protein Sci 4:337–360
Porter AG (1993) Picornavirus nonstructural proteins: emerging roles in virus replication and inhibition of host cell functions. J Virol 67:6917–6921
Qasim MA, Ranjbar MR, Wynn S, Anderson S, Laskowski M Jr (1995) Ionizable P1 residues in serine proteinase inhibitors undergo large pK shifts on complex formation. J Biol Chem 270:27419–27422
Rasnick D (1996) Small synthetic inhibitors of cysteine proteinases. Perspect Drug Discov Design 6:47–63
Read RJ, James MNG (1986) Introduction to the protein inhibitors: X-ray crystallography. In: ai]Barrett AJ, Salvesen G (eds) Proteinase inhibitors. Elsevier Science, Amsterdam
Roehl HH, Parsley TB, Ho TV, Semler BL (1997) Processing of a cellular polypeptide by 3CD proteinase is required for poliovirus ribonucleoprotein comlex formation. J Virol 71:578–585
Roivainen M, Knip M, Hyöty H, Kulmala P, Hiltunen M, Vähäsalo P, Hovi T, Äkerblom HK (1998) Several different enterovirus serotypes can be associated with prediabetic autoimmune episodes and onset of overt IDDM. Childhood Diabetes in Finland (DiMe) Study Group J Med Virol 56:74–78
Rueckert RR (1996) Picornaviridae: the viruses and their replication. In: ai]Fields BN, Knipe DM, Howley PM, Channock RM, Melnick JL, Monath TP, Roizmann BE, Straus SE (eds) Fields’ virology. Lippincott-Raven, Philadelphia
Ryan MD, Flint M (1997) Virus-encoded proteinases of the picornavirus super-group. J Gen Virol 78:699–723
Schechter I, Berger A (1967) On the size of the active site in proteases. I. Papain. Biochem Biophys Res Commun 27:157–162
Schiller JJ, Kanjanahaluethai A, Baker SC (1998) Processing of the coronavirus MHV-JHM polymerase polyprotein: identification of precursors and proteolytic products spanning 400kilodaltons of ORF 1a. Virology 242:288–302
Schultheiss T, Kusov YY, Gauss-Müller V (1994) Proteinase 3C of hepatitis A virus (HAV) cleaves the HAV polyprotein P2-P3 at all sites including VP1/2A and 2A/2B. Virology 198:275–281
Schultheiss T, Emerson SU, Purcell RH, Gauss-Müller V (1995) Polyprotein processing in echovirus 22 — a first assessment. Biochem Biophys Res Commun 219: 1120–1127
Seybert A, Ziebuhr J, Siddell SG (1997) Expression and characterization of a recombinant murine coronavirus 3C-like proteinase. J Gen Virol 78:71–75
Sham JL, Rosenbrook W, Kari W, Betebenner DA, Wideburg NE, Saldivar A, Plattner JJ, Norbeck DW (1995) Potent inhibitors of the human rhinovirus 3C protease containing a backbone modified glutamine. J Chem Soc Perkin Trans 1:1081–1082
Shepherd TA, Cox GA, McKinney A, Tang J, Wakulchik M, Zimmermann RE, Villareal EC (1996) Small peptidic aldehyde inhibitors of human rhinovirus 3C protease. Bioorg Med Chem Lett 6:2893–2896
Singh SB, Cordingley MG, Ball RG, Smith JL, Dombrowski AW, Goetz MA (1991) Structure and stereochemistry of thysanone: A novel human rhinovirus 3C protease inhibitor from Thysanophora penicilloides. Tetrahedron Lett 32:5279–5282
Sjogren MH (1998) Preventing acute liver disease in patients with chronic liver disease. Hepatology 27:887–888
Skern T (1998) Picornain 3C. In: Barrett AD, Rawlings NJ, Woesner F (eds) Handbook of proteolytic enzymes. Academic Press, London
Skiles JW, McNeil D (1990) Spiro indolinone α-lactams inhibitors of poliovirus and rhinovirus 3C-proteinases. Tetrahedron Lett 31:7277–7280
Snijder EJ, Wassenar ALM, van Dinten LC, Spaan WJM, Gorbalenya AE (1996) The arterivirus Nsp4 protease is the prototype of a novel group of chymotrypsin-like enzymes, the 3C-like serine proteases. J Biol Chem 271:4864–4871
Steinmann L, Conlon P (1997) Viral damage and the breakdown of self-tolerance. Nat Med 3:1085–1087
Storer AC, Ménard R (1994) Catalytic mechanism in papain family of cysteine peptidases. Methods Enzymol 244:486–500
Summers DF, Maizel JV (1968) Evidence for large precursor proteins in poliovirus synthesis. Proc Natl Acad Sci USA 59:966–971
Sutters RW, Cochi SL (1997) Ethical dilemmas in worldwide polio eradication programs. Am J Public Health 87:913–916
Taylor CE, Taylor ME, Cutts F (1997) Ethical dilemmas in current planning for polio eradication. Am J Public Health 87:922–925
Teterina NL, Bienz K, Egger D, Gorbalenya AE, Ehrenfeld E (1997a) Induction of intracellular membrane rearrangements by HAV proteins 2C and 2BC. Virology 237:66–77
Teterina NL, Gorbalenya AE, Egger D, Bienz K, Ehrenfeld E (1997b) Poliovirus 2C protein determinants of membrane binding and rearrangements in mammalian cells. J Virol 71:8962–8972
Thiel TK (1998) Hepatitis A vaccination. Am Fam Physician 57:1500–1501
Tibbies KW, Brierley I, Cavanagh D, Brown TDK (1996) Characterization in vitro of an autocatalytic processing activity associated with the predicted 3C-like proteinase domain of the Coronavirus avian infectious bronchitis virus. J Virol 70:1923–1930
Vento S, Garofano T, Renzini C, Cainelli F, Casali F, Ghironzi G, Ferraro T, Conaia E (1998) Fulminant hepatitis associated with hepatitis A virus superinfection in patients with chronic hepatitis C. N Engl J Med 338:286–290
Walker PA, Leong LEC, Porter AG (1995) Sequence and structural determinants of the interaction between the 5′-noncoding region of picornavirus RNA and rhinovirus protease 3C. J Biol Chem 270:14510–14516
Webber SE, Tikhe J, Worland ST, Fuhrmann SA, Hendrickson TF, Matthews DA, Love RA, Patick AK, Meador JW, Ferre PA, Brown EL, Delisle DM, Ford CE, Binford SL (1996) Design synthesis and evaluation of nonpeptide inhibitors of human rhinovirus 3C proteinase. J Med Chem 39:5072–5882
Webber SE, Okano K, Little TL, Reich SH, Xin Y, Fuhrman SA, Matthews DA, Love RA, Hendrickson TF, Patick AK, Meador JW III, Ferre RA, Brown EL, Ford CE, Binford SL, Worland ST (1998) Tripeptide aldehyde inhibitors of human rhinovirus 3C protease: design, synthesis, biological evaluation and cocrystal structure solution of P1 glutamine isosteric replacements. J Med Chem 41:2786–2805
Weidner JR, Dunn BM (1991) Development of synthetic peptide substrates for the poliovirus 3C proteinase. Arch Biochem Biophys 286:402–408
Whiting AK, Peticolas WL (1994) Details of the acyl-enzyme intermediate and the oxyanion hole in serine protease catalysis. Biochemistry 33:552–561
Wimmer E (1982) Genome linked proteins of viruses. Cell 28:199–201
Wimmer E, Hellen CUT, Cao X (1993) Genetics of poliovirus. Annu Rev Genet 27:353–436
Wirblich C, Sibilia M, Boniotti MB, Rossi C, Thiel H-J, Meyers G (1995) 3C-like protease of rabbit hemorrhagic disease virus: identification of cleavage sites in the ORF1 polyprotein and analysis of cleavage specificity. J Virol 69:7159–7169
Xiang WS, Harris KS, Alexander L, Wimmer E (1995) Interaction between the 5′-terminal cloverleaf and 3AB/3CDPro of poliovirus is essential for RNA application. J Virol 69:3658–3667
Xiang W, Cuconati A, Hope D, Kirkegaard K, Wimmer E (1998) Complete protein linkage map of poliovirus P3 proteins: interaction of polymerase 3Dpol with Vpg and with genetic variants of 3AB. J Virol 72:6732–6741
Yalamanchili D, Weidman K, Dasgupta A (1997) Cleavage of transcriptional activator Oct-1 by poliovirus encoded protease 3Cpro. Virology 239:176–185
Ypma-Wong MF, Dewalt PG, Johnson VH, Lamb JG, Semler BL (1988) Protein 3CD is the major poliovirus proteinase responsible for cleavage of the PI capsid precursor. Virology 166:265–270
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2000 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Bergmann, E.M., James, M.N.G. (2000). The 3C Proteinases of Picornaviruses and Other Positive-Sense, Single-Stranded RNA Viruses. In: von der Helm, K., Korant, B.D., Cheronis, J.C. (eds) Proteases as Targets for Therapy. Handbook of Experimental Pharmacology, vol 140. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-57092-6_7
Download citation
DOI: https://doi.org/10.1007/978-3-642-57092-6_7
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-63023-1
Online ISBN: 978-3-642-57092-6
eBook Packages: Springer Book Archive