Abstract
Maintaining alertness is critical to the successful performance of a variety of tasks, ranging from target detection to language comprehension. Previous studies have identified neural correlates of fluctuations in alertness, namely the cingulo-opercular network (CON) and the default mode network (DMN). The current study examined the extent to which pupil dilation, an autonomic physiological response, tracks variation in the CON and DMN in a functional neuroimaging and pupillometry resting-state session. The results revealed that increasing pupil dilation was associated with increasing CON activity and decreasing DMN activity, and that regions of these networks were functionally anti-correlated. Furthermore, individuals with better self-reported attention control abilities (i.e., more mindful of the present moment) exhibited a reduction in the trade-off between these systems. These results suggest that pupillometry provides a valuable proxy of activity in the neural systems that underlie alertness, which may be impacted by individual differences in mindfulness.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Successfully performing a long or tedious task depends on one’s ability to maintain alertness. Also referred to as arousal, sustained attention, or vigilance (but contrasted with focused attention) [1], alertness is associated with broad sensitivity to changes in the environment or in one’s internal state. Fluctuations in alertness are regulated by the widespread cortical release of norepinephrine via the locus coeruleus (LC) brainstem nucleus [2]. In particular, baseline LC activity is characterized by a relatively slow, tonic pattern of neural firing, which is linked to the intrinsic control of general arousal, that is, tonic alertness [3].
Cortical neural and physiological proxies of LC function have been investigated in order to circumvent the methodological challenges of recording LC brainstem activity in humans [4]. In particular, the LC has connections with the cingulo-opercular network (CON), which comprises the anterior cingulate cortex (ACC), bilateral anterior insulae/frontal opercula (AI/fO), and thalamus. The CON is engaged in challenging conditions [5], but more broadly, as individuals maintain alertness either at rest or during a task [6–8].
For example, in a recent functional magnetic resonance imaging (fMRI) study, participants performed a vigilance task in which they identified brief targets presented at unpredictable intervals [6]. Greater activity in the CON prior to stimulus presentation predicted better target detection. Interestingly, a vigilance benefit was also associated with reduced suppression of the default mode network (DMN). The DMN, whose core regions include the precuneus, bilateral angular gyri (AG), and the medial prefrontal cortex (mPFC), is canonically suppressed during task performance but upregulated at rest [9] or during mind wandering or self-reflection [10, 11]. Though DMN and CON activity are commonly anti-correlated across time [12, 13], research has shown that this relation varies across individuals [14] and following attention-based training programs (i.e., mindfulness meditation) [15, 16]. However, less is known about the extent to which CON-DMN connectivity is modulated by individual differences in the tendency to be aware of one’s environment versus self-reflect, that is, their dispositional mindfulness [17].
Though there is mounting evidence that the balance between CON and DMN activity indexes tonic alertness, fMRI biomarkers are not feasible to collect for a variety of environments or individuals. Physiological measures of alertness may be more practical, such as pupil dilation, which is an autonomic, physiological response that varies with phasic and tonic changes in LC activity [18, 19]. Additionally, increasing pupil size has been associated with greater alertness in rats [20] and with noradrenergic drugs that increase arousal in humans [21].
Additional research is needed, however, to elucidate the extent to which pupil dilation can be used as a proxy of CON and DMN activity, or if it instead indexes a limited portion of the tonic alertness continuum (e.g., from attentive to inattentive). For example, one recent study has demonstrated that average pupil dilation across an fMRI resting-state scan, in which participants are given no explicit task, was correlated with activity in the LC and with regions of the ACC, right insula, and visual cortex [19]. These types of scans are sensitive to spontaneous fluctuations in neural activity across time (for example, as individuals naturally cycle through various states of alertness). This study suggests, then, that pupil dilation intrinsically varies with activity in attention-related regions. However, only positive correlations between dilation and neural activity were reported (increasing dilation with increasing activity) and only within a subset of CON regions. Given that the ACC and DMN are typically anti-correlated, we additionally predict that decreasing pupil dilation should be associated with greater DMN activity.
The current resting-state study aims to replicate and extend previous work by testing the extent to which (1) increasing pupil dilation is related to greater activity in regions of the CON, (2) decreasing dilation is related to greater activity in regions of the DMN, and (3) these areas function as coherent networks that are anti-correlated with one another. Finally, following research suggesting that individual differences in awareness of one’s own attentional states modulates the relationship between CON and DMN activity [15, 16], we examined the extent to which (4) self-reported dispositional mindfulness altered functional connectivity between these networks.
2 Method
2.1 Participants
Twenty-three young adult participants were included in this study (an additional 15 were tested but had unusable pupillometry data, defined below). Participants were 18 to 30 years old (M = 20.8, SD = 3.2; 14 female), monolingual native American-English speakers, and right-handed (Edinburgh Handedness Inventory [22], M = 84.6, SD = 11.6, on a scale from −100 = maximally left handed to +100 = maximally right handed). All reported normal or corrected-to-normal visual acuity and hearing, no history of neurological or psychological disorders or events, no history of drug abuse, and no contraindications for MRI scanning, such as metal implants. They also indicated that they had no more than minimal meditation experience (e.g., breathing exercises during beginner-level yoga, but no mindfulness meditation exposure) and were not currently involved in cognition- or meditation-based training. Participants gave informed consent in compliance with the University of Maryland Institutional Review Board and the Declaration of Helsinki and were compensated for their participation.
2.2 Materials
Participants completed questionnaires regarding demographics, handedness, and mindfulness online using Qualtrics software (Provo, UT). The Five Facet Mindfulness Questionnaire (FFMQ) is a measure of individual differences in mindful disposition in daily life activities [23]. Participants self-report on a five-point Likert scale how true each of 39 statements are for them (e.g., “When I’m walking, I deliberately notice the sensations of my body moving”). A total mindfulness score was obtained by summing the responses (with appropriate items reverse scored): values could range from 39 (minimum) to 195 (maximum dispositional mindfulness).
2.3 Procedure
After providing informed consent, participants completed demographic and handedness questionnaires. In the MRI environment, structural and field-mapping scans were collected prior to the resting-state functional scans.
During the subsequent functional scans, physiological measurements were collected to control for potential individual differences that could confound our imaging results (i.e., respiration, heart rate). Respiration and heart rate were collected using a BIOPAC system that sampled at 1000 Hz (BIOPAC Systems, Inc., Goleta, CA, USA). Pupil dilation was measured with an MR-compatible EyeLink 1000 Hz eye tracker (SR-Research, Ontario, Canada).
The resting-state scan lasted nine minutes and was collected prior to any experimental task to avoid potential carry-over effects that could influence network activity during the resting state [24]. For the duration of the scan, participants were instructed to fixate on five hash marks in the center of the screen (i.e., #####), which subtended four degrees of visual angle horizontally and one degree vertically. Such eyes-open, central fixation protocols have been shown to yield more robust and reliable patterns of resting-state connectivity, including for regions of the DMN and CON [25].
Following the scan session, participants filled out the FFMQ questionnaire, were debriefed, and compensated for their participation.
2.4 Analysis
Pupillometry.
Pupil size values that the eye-tracker software detected as poor quality, occurring during a blink, a saccade, or a fixation that fell along the extreme edges of the screen (more than two-thirds of the screen width or height away from center), were removed and linearly interpolated. Participants for whom more than 50 % of the data required interpolation were removed from analyses [26]. Of the remaining 23 participants, on average 23.1 % (SD = 16.8 %) of trial time points were interpolated. Data were smoothed with a five-point moving average, down-sampled into 2200 ms bins to match the acquisition rate of the functional images (following previous research [19]), and z-score transformed. Finally, linear regression was used to estimate the extent to which the horizontal and vertical coordinates that described the gaze position predicted pupil size for each trial. The residualized variance, after accounting for the impact of gaze position on pupil, was entered into neuroimaging analyses. The average non-normalized pupil size for each individual was not correlated with the FFMQ scores, r(21) = .16, p = .47.
Neuroimaging.
Preprocessing. Structural images were preprocessed using SPM8 (http://www.fil.ion.ucl.ac.uk/spm). The images were coregistered to Montreal Neurological Institute (MNI) space, skull-stripped (Brain Extraction Toolkit; [27]), segmented (using the New Segment tool), and the gray-matter segmented images were then normalized to a gray-matter standard-space MNI template.
Functional images were preprocessed using SPM8. Steps included: slice time correction, realignment and unwarping, coregistration to the MNI-aligned structural image, normalization to MNI space (using the warping parameters derived from structural preprocessing), and smoothing (8 mm kernel). The Linear Model of the Global Signal method [28] was then used to detrend the global mean signal fluctuations from the preprocessed images. Image volumes with significantly deviant signal (more than 2.5 standard deviations) relative to the global mean and with a significantly deviant number of voxels from their voxel-wise mean across the run were identified and modeled as two nuisance regressors in the first-level analysis [29]. Two motion nuisance regressors, representing 3D translational and rotational motion with respect to the onset of the scan, were calculated from the six realignment parameters generated in SPM via the Pythagorean Theorem [30, 31].
Correction for physiological noise was performed via RETROICOR [32] using Fourier expansions of different order for the estimated phases of the cardiac pulsation (2nd order) and respiration (2nd order). Corresponding confound regressors were created with the MATLAB PhysIO Toolbox [33] available as part of the TAPAS software collection (www.translationalneuromodeling.org/tapas). This yielded eight nuisance regressors for inclusion in first-level imaging statistics.
Statistics. A first-level analysis was performed to estimate the activity associated with the mean pupil dilation across the resting-state scan. The general linear model (GLM) contained a parameter that described the average pupil dilation (following preprocessing) during each TR and the censorship, motion, and physiological nuisance regressors described above. First-level GLMs were convolved with the SPM8 canonical HRF and high-pass filtered at 128 s. Contrasts were derived in the first-level analysis to examine the relative activation associated with increasing pupil dilation. Second-level GLMs were performed to examine the effects across participants.
The peak CON-related cluster resulting from the pupillometry analysis was used to define a seed region in a functional connectivity analysis. Activity across the timeseries was extracted for each participant from both the thresholded region of interest (ROI) and from a MNI gray matter mask (at least 20 % probability gray matter), which was included to control for overall fluctuations in neural activity. The two activity vectors were included as regressors in addition to the nuisance regressors in a first-level whole-brain analysis. Second-level statistics were performed to examine patterns of connectivity across individuals and their variation with individual differences in reported mindfulness (FFMQ score).
Thresholds for significance in imaging analyses were set at peak p < .001 uncorrected and cluster extent p < .01 uncorrected [34] unless otherwise noted. MNI coordinates of all of the significant clusters in each figure are listed in tables.
3 Results
3.1 Relationship Between Pupil Dilation and Resting-State Activity
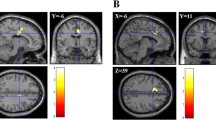
We first tested the extent to which pupil dilation is sensitive to changes in activity in the CON and DMN across time (Fig. 1, Table 1). Consistent with previous research [19], increasing pupil dilation during a resting-state scan was associated with greater activity in regions of the CON, though in this study we observed more extensive pattern of activity (ACC extending into supplementary motor area, bilateral AI, thalamus), auditory cortex (bilateral superior temporal gyrus), and visual cortex (lingual gyrus, extending into the calcarine sulcus; Fig. 1, red).
Extending previous work, we also observed that decreasing pupil dilation was related to greater activity in the right temporo-parietal junction extending into the AG and in the right anterior temporal pole (Fig. 1, blue), which are both part of the dorsal medial subsystem of the DMN that has been associated with reflecting on the mental states of oneself or others (i.e., mentalizing) [35].
3.2 Functional Connectivity and Individual Differences in Mindfulness
To evaluate the extent to which the pupil-related regions from the previous analysis formed coherent neural networks, a functional connectivity analysis was performed. An ROI was defined as the thresholded peak CON-related cluster (i.e., the mid/anterior cingulate cortex cluster in Fig. 1/Table 1 with peak coordinate at [7, 14, 44]). The timeseries of this dorsal ACC ROI and, as a control, a gray matter mask were extracted for each participant across the resting-state scan. These regressors were entered into a first-level GLM for each participant to reveal the neural regions with a correlated or anti-correlated pattern of activation across time.
Figure 2A (red, Table 2) shows that the ACC was engaged in synchrony with the CON and with sensory cortices; functionally correlated regions included the ACC, bilateral AI/fO extending into middle frontal gyri, the thalamus, and regions of visual and auditory cortex. As predicted, activity in the ACC was anti-correlated with that of regions of the DMN (blue), including the bilateral angular gyri, inferior temporal cortices, mPFC, precuneus, hippocampi, and cerebellum.
(A) ACC activity is positively functionally correlated with activity in other CON regions, visual and auditory cortex (red) and anti-correlated with DMN regions (blue). More mindful individuals tended to exhibit a weaker anti-correlation between areas of the DMN and ACC (pink). (B) Extracted effect estimates (a.u.) clarify the relation between greater mindfulness (higher FFMQ score) and reduced anti-correlation of the ACC ROI and right AG shown in (A) (Color figure online).
FFMQ scores (M = 129.3, SD = 12.3) were entered as a regressor in a second-level GLM to investigate the extent to which the anti-correlation between ACC and DMN activity was modulated by individual differences in dispositional mindfulness. With increasing mindfulness, a weaker anti-correlation between the ACC and regions of the DMN, specifically in the right AG and the precuneus (Fig. 2A pink, Table 2), was observed. To clarify the direction of this result, the effect in the right AG cluster for each individual was plotted against their FFMQ score. Figure 2B depicts that while less mindful individuals exhibited a pattern of functional connectivity commonly reported in the literature (a strong, negative relation), mindful individuals tended to show little to no anti-correlation between the ACC and the right AG.
4 Discussion
The current study demonstrated a tight link between a physiological and a neural measure of tonic alertness. Supporting previous work [19], greater pupil dilation was associated with increasing neural activity within regions of the CON during the course of a resting-state session, in which participants simply fixated a screen for nine minutes. The CON has been described as a salience network, which integrates conflict monitoring, autonomic responses, and reward processing [36]. This system appears to facilitate the detection of salient stimuli that lead to rewarding behaviors, that is, an LC-driven pattern of scanning attention. The pupil-associated activity in this study may have been more extensive than previously reported for methodological reasons such as its enrollment of a larger, younger sample (i.e., 60 % more participants, mean age nine years younger, max age 18 years younger). Though research is needed to test the impact of such factors, aging has been shown to alter alertness and functional connectivity between the ACC and DMN even in middle-aged adults [37, 38].
Following studies showing that CON activity enhances sensory processing [39], we also observed that regions of primary and secondary auditory and visual cortices were associated with increasing pupil dilation in addition to the CON. However, the present study employed a resting-state design in which no stimuli were presented nor explicit task given, other than to fixate crosshairs in the center of a screen. The fixation symbols as well as scanner noise were consistent throughout the session. However with increasing alertness, participants may have become more aware of these environmental stimuli, which could explain the observed upregulation of sensory cortices. Though resting-state studies are informative about the systems underlying basic cognitive functions [40], future research should continue to explore the extent to which pupil dilation indexes alertness during explicit tasks (e.g., [19]). Indeed, the adaptive gain theory of LC function indicates that optimal task performance is associated with moderate levels of tonic alertness [2]. Very high levels of arousal, such as occur in periods of stress, may not be fully sampled in a resting-state study.
When not focused on the external environment, participants also likely engaged in mind wandering. The DMN is typically suppressed during active task processing, but relatively engaged during mind wandering and self-reflection [10, 11]. Extending previous research [19], we observed that decreasing dilation was associated with increasing engagement of the dorsal medial subsystem of the DMN. In particular, these regions are thought to interact with core regions of the DMN to support mentalizing [35]. Mentalizing involves ruminating on one’s own or other people’s mental states [41] and is thought to be a major component of mind wandering [42, 43]. Future research may more firmly establish the relationship between pupil dilation and activity in this DMN subsystem by probing the frequency and contents of mind wandering across a study.
To verify that the observed pupil-related regions were engaged as part of the CON and DMN networks, a functional connectivity analysis was performed using the pupil-related ACC region as a seed (along with a gray-matter control seed). In line with previous resting-state studies [12, 13], ACC activity was synchronously engaged with activity across the entire CON and anti-correlated with regions of the DMN. Thus, pupil dilation appears to track a range of alertness via its sensitivity to the trade-off between CON and DMN activity. By linking physiological and neural measures, our results suggest that pupillometry may provide an online measure of changes in alertness that is easier to collect and less costly than neuroimaging metrics.
Finally, we observed that the extent to which individuals had mindful dispositions altered the connectivity between the CON and DMN. Mindfulness refers to the ability to be aware in the present moment, non-judgmentally accepting one’s thoughts and experiences [17]. In this study, individuals who scored higher on the FFMQ exhibited a weaker negative to null correlation. Though the CON and DMN are typically anti-correlated [12, 13], some research suggests that reading [44], autobiographical planning [45], vigilance tasks [6], or even awareness of mind wandering [10] may benefit from the coordinated activation of frontal attention and DMN networks. These studies hypothesized that the systems work together to regulate inner-directed thoughts or maintain internalized task goals in the face of shifting environmental stimuli. Extrapolating our results, one may expect a positive correlation with greater levels of dispositional mindfulness than sampled in the current study (Fig. 2B), such as may occur with mindfulness meditation training [46]. Indeed, a recent study showed increasing positive connectivity between frontal-attention and DMN regions following mindfulness meditation compared to a relaxation-only control [16]. Additionally, CON-DMN co-activations may be more readily observed in the context of explicit tasks, which impose greater demands on attentional processes. Studies currently underway will be analyzed to test these hypotheses.
The current study has implications for both basic and applied research. The ability to maintain tonic alertness is critical for performing many job-related activities accurately, quickly, and with minimal effort. For example, for government language professionals, detecting relevant information that occurs infrequently within text or spoken materials often requires substantial vigilance: individuals must be engaged in their primary task goal (reading/listening) while minimizing mind wandering. The ability to track fluctuations in alertness with pupillometry could lead to the development of real-time predictors of performance [47], thus improving our ability to select, train, and evaluate our workforce.
5 Conclusions
The current study demonstrated that variation in baseline pupil dilation tracks changes in the neural systems that underlie tonic alertness. In a simultaneous pupillometry and functional neuroimaging resting-state study, increasing dilation was associated with the engagement of the CON, while decreasing dilation was associated with greater DMN activity. A functional connectivity analysis revealed that these networks were anti-correlated, which was modulated by individual differences in self-reported mindful behavior in daily life. This work supports the feasibility of collecting online measures of tonic alertness and highlights that individual differences may modulate the neural systems that underlie it.
References
Oken, B.S., Salinsky, M.C., Elsas, S.M.: Vigilance, alertness, or sustained attention: physiological basis and measurement. Clin. Neurophysiol. 117, 1885–1901 (2006)
Aston-Jones, G., Cohen, J.D.: An integrative theory of locus coeruleus-norepinephrine function: adaptive gain and optimal performance. Annu. Rev. Neurosci. 28, 403–450 (2005)
Posner, M.I.: Measuring alertness. Ann. N. Y. Acad. Sci. 1129, 193–199 (2008)
Eckert, M.A., Keren, N.I., Aston-Jones, G.: Looking Forward with the Locus Coeruleus (Commentary on Modafinil Shifts Human Locus Coeruleus to Low-Tonic, High-Phasic Activity During Functional MRI by Astafiev et al.). www.sciencemag.org/content/328/5976/309.1.short/reply#sci_el_13365
Vaden, K.I., Kuchinsky, S.E., Cute, S.L., Ahlstrom, J.B., Dubno, J.R., Eckert, M.A.: The cingulo-opercular network provides word-recognition benefit. J. Neurosci. 33, 18979–18986 (2013)
Coste, C.P., Kleinschmidt, A.: Cingulo-opercular network activity maintains alertness. Neuroimage 128, 264–272 (2016)
Sadaghiani, S., D’Esposito, M.: Functional characterization of the cingulo-opercular network in the maintenance of tonic alertness. Cereb. Cortex 25, 2763–2773 (2015)
Kuchinsky, S.E., Vaden, K.I., Ahlstrom, J.B., Cute, S.L., Humes, L.E., Dubno, J.R., Eckert, M.A.: Task-related vigilance during word recognition in noise for older adults with hearing loss. Exp. Aging Res. 42, 50–66 (2016)
Raichle, M.E., MacLeod, A.M., Snyder, A.Z., Powers, W.J., Gusnard, D.A., Shulman, G.L.: A default mode of brain function. Proc. Natl. Acad. Sci. 98, 676–682 (2001)
Christoff, K., Gordon, A.M., Smallwood, J., Smith, R., Schooler, J.W.: Experience sampling during fMRI reveals default network and executive system contributions to mind wandering. Proc. Natl. Acad. Sci. 106, 8719–8724 (2009)
Mittner, M., Boekel, W., Tucker, A.M., Turner, B.M., Heathcote, A., Forstmann, B.U.: When the brain takes a break: a model-based analysis of mind wandering. J. Neurosci. 34, 16286–16295 (2014)
Fox, M.D., Snyder, A.Z., Vincent, J.L., Corbetta, M., Van Essen, D.C., Raichle, M.E.: The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc. Natl. Acad. Sci. 102, 9673–9678 (2005)
Damoiseaux, J.S., Rombouts, S.A.R.B., Barkhof, F., Scheltens, P., Stam, C.J., Smith, S.M., Beckmann, C.F.: Consistent resting-state networks. Proc. Natl. Acad. Sci. 103, 13848–13853 (2006)
Chang, C., Glover, G.H.: Time-frequency dynamics of resting-state brain connectivity measured with fMRI. Neuroimage 50, 81–98 (2010)
Kilpatrick, L.A., Suyenobu, B.Y., Smith, S.R., Bueller, J.A., Goodman, T., Creswell, J.D., Tillisch, K., Mayer, E.A., Naliboff, B.D.: Impact of mindfulness-based stress reduction training on intrinsic brain connectivity. Neuroimage 56, 290–298 (2011)
Creswell, J.D., Taren, A.A., Lindsay, E.K., Greco, C.M., Gianaros, P.J., Fairgrieve, A., Marsland, A.L., Brown, K.W., Way, B.M., Rosen, R.K., Ferris, J.L.: Alterations in resting state functional connectivity link mindfulness meditation with reduced interleukin-6: a randomized controlled trial. Biol. Psychiatry. (in press)
Bishop, S.R., Lau, M., Shapiro, S., Carlson, L., Anderson, N.D., Carmody, J., Segal, Z.V., Abbey, S., Speca, M., Velting, D., Devins, G.: Mindfulness: a proposed operational definition. Clin. Psychol. Sci. Pract. 11, 230–241 (2004)
Gilzenrat, M.S., Nieuwenhuis, S., Jepma, M., Cohen, J.D.: Pupil diameter tracks changes in control state predicted by the adaptive gain theory of locus coeruleus function. Cogn. Affect. Behav. Neurosci. 10, 252–269 (2010)
Murphy, P.R., O’Connell, R.G., O’Sullivan, M., Robertson, I.H., Balsters, J.H.: Pupil diameter covaries with bold activity in human locus coeruleus. Hum. Brain Mapp. 35, 4140–4154 (2014)
McGinley, M.J., David, S.V., McCormick, D.A.: Cortical membrane potential signature of optimal states for sensory signal detection. Neuron 87, 179–192 (2015)
Phillips, M.A., Bitsios, P., Szabadi, E., Bradshaw, C.M.: Comparison of the antidepressants reboxetine, fluvoxamine and amitriptyline upon spontaneous pupillary fluctuations in healthy human volunteers. Psychopharmacology 149, 72–76 (2000)
Oldfield, R.C.: The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9, 97–113 (1971)
Baer, R.A., Smith, G.T., Hopkins, J., Krietemeyer, J., Toney, L.: Using self-report assessment methods to explore facets of mindfulness. Assessment 13, 27–45 (2006)
Pyka, M., Beckmann, C.F., Schöning, S., Hauke, S., Heider, D., Kugel, H., Arolt, V., Konrad, C.: Impact of working memory load on fMRI resting state pattern in subsequent resting phases. PLoS ONE 4, e7198 (2009)
Patriat, R., Molloy, E.K., Meier, T.B., Kirk, G.R., Nair, V.A., Meyerand, M.E., Prabhakaran, V., Birn, R.M.: The effect of resting condition on resting-state fMRI reliability and consistency: a comparison between resting with eyes open, closed, and fixated. Neuroimage 78, 463–473 (2013)
Siegle, G.J., Steinhauer, S.R., Stenger, V.A., Konecky, R., Carter, C.S.: Use of concurrent pupil dilation assessment to inform interpretation and analysis of fMRI data. Neuroimage 20, 114–124 (2003)
Smith, S.M.: Fast robust automated brain extraction. Hum. Brain Mapp. 17, 143–155 (2002)
Macey, P.M., Macey, K.E., Kumar, R., Harper, R.M.: A method for removal of global effects from fMRI time series. Neuroimage 22, 360–366 (2004)
Vaden, K.I., Muftuler, L.T., Hickok, G.: Phonological repetition-suppression in bilateral superior temporal sulci. Neuroimage 49, 1018–1023 (2010)
Kuchinsky, S.E., Vaden, K.I., Keren, N.I., Harris, K.C., Ahlstrom, J.B., Dubno, J.R., Eckert, M.A.: Word intelligibility and age predict visual cortex activity during word listening. Cereb. Cortex 22, 1360–1371 (2012)
Wilke, M.: An alternative approach towards assessing and accounting for individual motion in fMRI timeseries. Neuroimage 59, 2062–2072 (2012)
Glover, G.H., Li, T.Q., Ress, D.: Image-based method for retrospective correction of physiological motion effects in fMRI: RETROICOR. Magn. Reson. Med. 44, 162–167 (2000)
Kasper, L., Marti, S., Vannesjö, S.J., Hutton, C., Dolan, R., Weiskopf, N., Stephan, K.E., Prüssmann, K.P.: Cardiac artefact correction for human brainstem fMRI at 7 Tesla. In: Proceedings of the Organization for Human Brain Mapping, p. 395. San Francisco, CA (2009)
Poline, J.B., Worsley, K.J., Evans, A.C., Friston, K.J.: Combining spatial extent and peak intensity to test for activations in functional imaging. Neuroimage 5, 83–96 (1997)
Andrews-Hanna, J.R., Reidler, J.S., Sepulcre, J., Poulin, R., Buckner, R.L.: Functional-anatomic fractionation of the brain’s default network. Neuron 65, 550–562 (2010)
Seeley, W.W., Menon, V., Schatzberg, A.F., Keller, J., Glover, G.H., Kenna, H., Reiss, A.L., Greicius, M.D.: Dissociable intrinsic connectivity networks for salience processing and executive control. J. Neurosci. 27, 2349–2356 (2007)
Carrier, J., Monk, T.H., Buysse, D.J., Kupfer, D.J.: Sleep and morningness-eveningness in the “middle” years of life (20–59 y). J. Sleep Res. 6, 230–237 (1997)
Hampson, M., Tokoglu, F., Shen, X., Scheinost, D., Papademetris, X., Constable, R.T.: Intrinsic brain connectivity related to age in young and middle aged adults. PLoS ONE 7, e44067 (2012)
Sadaghiani, S., Hesselmann, G., Kleinschmidt, A.: Distributed and antagonistic contributions of ongoing activity fluctuations to auditory stimulus detection. J. Neurosci. 29, 13410–13417 (2009)
Buckner, R.L., Vincent, J.L.: Unrest at rest: default activity and spontaneous network correlations. Neuroimage 37, 1091–1096 (2007)
Frith, C.D., Frith, U.: The neural basis of mentalizing. Neuron 50, 531–534 (2006)
Buckner, R.L., Andrews-Hanna, J.R., Schacter, D.L.: The brain’s default network: anatomy, function, and relevance to disease. Ann. N. Y. Acad. Sci. 1124, 1–38 (2008)
Andrews-Hanna, J.R.: The brain’s default network and its adaptive role in internal mentation. Neuroscientist 18, 251–270 (2012)
Smallwood, J., Gorgolewski, K.J., Golchert, J., Ruby, F.J.M., Engen, H., Baird, B., Vinski, M.T., Schooler, J.W., Margulies, D.S.: The default modes of reading: modulation of posterior cingulate and medial prefrontal cortex connectivity associated with comprehension and task focus while reading. Front. Hum. Neurosci. 7, 734 (2013)
Spreng, R.N., Stevens, W.D., Chamberlain, J.P., Gilmore, A.W., Schacter, D.L.: Default network activity, coupled with the frontoparietal control network, supports goal-directed cognition. Neuroimage 53, 303–317 (2010)
Baer, R.A., Carmody, J., Hunsinger, M.: Weekly change in mindfulness and perceived stress in a mindfulness-based stress reduction program. J. Clin. Psychol. 68, 755–765 (2012)
Marshall, S.P.: The index of cognitive activity: measuring cognitive workload. In: Human Factors and Power Plants, Proceedings of the 2002 IEEE 7th Conference on Human Factors Modeling, vol. 7, pp. 5–9. IEEE, Scottsdale, AZ (2002)
Acknowledgements
This material is based upon work supported, in whole or in part, with funding from the United States Government. Any opinions, findings and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the University of Maryland, College Park and/or any agency or entity of the United States Government. We thank Jared Novick and Nina Hsu for their thoughtful discussion of this work as well as the study participants.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this paper
Cite this paper
Kuchinsky, S.E., Pandža, N.B., Haarmann, H.J. (2016). Linking Indices of Tonic Alertness: Resting-State Pupil Dilation and Cingulo-Opercular Neural Activity. In: Schmorrow, D., Fidopiastis, C. (eds) Foundations of Augmented Cognition: Neuroergonomics and Operational Neuroscience. AC 2016. Lecture Notes in Computer Science(), vol 9743. Springer, Cham. https://doi.org/10.1007/978-3-319-39955-3_21
Download citation
DOI: https://doi.org/10.1007/978-3-319-39955-3_21
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-39954-6
Online ISBN: 978-3-319-39955-3
eBook Packages: Computer ScienceComputer Science (R0)