Abstract
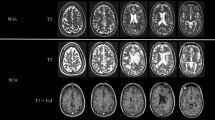
A 24-year-old patient (see Case 16) with relapsing-remitting multiple sclerosis (RRMS) returned 6 months after diagnosis with sub-acute diplopia. She described difficulty moving her eyes to the right and horizontal, binocular diplopia, maximal in leftward gaze. There were no other neurological symptoms. Examination revealed conjugate horizontal gaze palsy to the right and limited adduction of the right eye, constituting the ‘one-and-a-half syndrome.’ A repeat MRI scan revealed a new lesion in the right pons affecting the region of the abducens nucleus and the ipsilateral medial longitudinal fasciculus. There was no enhancement. The overall burden of lesions on MRI was otherwise stable, and the enhancing right frontal lesion previously seen was less prominent.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Gold R, Theodore Phillips J, Havrdova E, et al. P839 Delayed-release dimethyl fumarate and pregnancy: preclinical studies and pregnancy outcomes from clinical trials and postmarketing experience. Neurol Ther. 2015;1-12.
Wei QR, Runrong G, Xiangdong S. Studies on teratogenicity of dimethyl fumarate. J Hyg Res. 1990;19:28-31.
Karlsson G, Francis G, Koren G, et al. Pregnancy outcomes in the clinical development program of fingolimod in multiple sclerosis. Neurology. 2014;82:674-680.
Novartis Pharmaceuticals Corporation. Gilenya (fingolimod) prescribing information (2010). Revised August 2015. www.accessdata.fda.gov/drugsatfda_docs/label/2015/022527s019lbl.pdf. Accessed April 6, 2016.
Kieseier BC, Benamor M, Truffinet P, Henson LJ. Pregnancy outcomes for female patients and partners of male patients in the teriflunomide clinical development program. Presented at: ACTRIMS-ECTRIMS; September 12, 2014; Boston, Massachusetts, USA. Poster P846.
Genzyme Corporation. Aubagio (teriflunomide) prescribing information. Revised October 2014a. http://products.sanofi.us/aubagio/aubagio.pdf. Accessed April 6, 2016.
Cristiano L, Friend S, Bozic C, Bloomgren G. Evaluation of pregnancy outcomes from the TYSABRI (Natalizumab) pregnancy exposure registry. Neurology. 2013;80(Meeting Abstracts 1):P02.127.
Biogen Idec Inc. Tysabri (natalizumab) prescribing information. Revised May 2015. www.tysabri.com/prescribingInfo. Accessed April 6, 2016.
Genzyme Corporation. Lemtrada (alemtuzumab) prescribing information. Revised November 2014. http://products.sanofi.us/lemtrada/lemtrada.pdf. Accessed April 6, 2016.
McCombe P, Achiron A, Brinar B, et al. Pregnancy outcomes in the alemtuzumab multiple sclerosis clinical development program. Presented at: ACTRIMS-ECTRIMS; September 12, 2014; Boston, Massachusetts, USA. Poster P842.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Lincoln, M.R., Oh, J. (2017). Multiple sclerosis therapies and pregnancy. In: Giacomini, P. (eds) Case Studies in Multiple Sclerosis. Adis, Cham. https://doi.org/10.1007/978-3-319-31190-6_17
Download citation
DOI: https://doi.org/10.1007/978-3-319-31190-6_17
Published:
Publisher Name: Adis, Cham
Print ISBN: 978-3-319-31188-3
Online ISBN: 978-3-319-31190-6
eBook Packages: MedicineMedicine (R0)