Abstract
Faithful and stable inheritance of DNA is coupled with occasional random errors of replication that lead to a change in the DNA code known as mutation. Mutations can be considered as “good” because they are the fuel that drives evolution of species. On the level of the individual they are mostly harmful. In fact, the majority of severe intellectual disabilities derives from such random mutational events. In my experience, the tendency to ascribe all events to definite causes is still highly prevalent. Against this background of presumed guilt, parents who are confronted with the birth of a severely handicapped child tend to take solace form the knowledge that the condition was not their “fault”. Our recent understanding that severe handicaps may strike anyone, may well lead to the acceptance of a more universal offer of prenatal diagnosis than previous strategies which were based on the identification of high risk groups.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Random Mutational Events
- Severe Intellectual Disability
- Presumed Guilt
- First-cousin Marriages
- Craniodiaphyseal Dysplasia
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Fascination
For as long as we know, people have been devastated and fascinated by the birth of a child with severe malformations or disabilities. Collecting malformed foetuses was a popular pastime for the elite during the 17th Century. Rich and educated men built up sizable private collections of curiosities. One such anatomical collection was sold in its entirety to Czar Peter the Great in 1717 by Frederik Ruijsch from Amsterdam (Baljet and Oostra 1998). An anatomical collection from the 18th century that has been preserved and maintained as a museum is that of Willem and Gerard Vrolik. This is now in the AMC hospital in Amsterdam. People with malformations or other visible developmental defects were put on display in “freak shows” and exhibitions. In the 19th century, PT Barnum in the USA and Tom Norman in the UK traveled widely around their respective countries, with shows of supposed freaks of nature. Quite probably, malformations will continue to scare and excite us forever. Certainly, our fascination with physical abnormality has not ceased in the 20th century. The 1980 movie “the elephant man” directed by David Lynch relates the story of John Merrick whose malformations were exploited by the owner of such a freak show. The 1985 movie “Mask” was based on the life of Roy Lee Dennis who died at age 16 from craniodiaphyseal dysplasia, a progressive deforming bone disease of the skull. Another contemporary movie about malformation is Edward Scissorhands (Tim Burton 1990). The image of a boy born with scissors for hands is clearly inspired by inherited ectrodactyly or “lobster claw malformation” where the middle fingers are missing at birth. A fascination with malformations can further be found in many literary tales, notably Homer’s Cyclops in the Odyssey.
2 Divinity and Sorcery
Beyond fascination is the need to find explanations for personal disasters such as the birth of a malformed or handicapped child. In antiquity, and in societies around the world, congenital abnormalities were regarded as omens, or punishment from the gods (Warkany 1959; Beckwith 2012). For example, Tigay (1997) mentions the Babylonian Omen series (Izbu) which lists the predicted significance of women giving birth to children with a wide variety of malformations. “If a woman gives birth (and the child) has two heads: there will be a fierce attack against the land and the king will give up his throne” (Izbu, II, 20 h32) (Pangas 2000). Although divinity was not generally considered a plausible cause after the middle ages, witchcraft and other supernatural phenomena remained serious possibilities until relatively recently. A case cited by Brent and Fawcett (2007) concerns the trial of one George Spencer from Connecticutt, who had a glass eye. When a one-eyed piglet was born on the farm, he was charged with bestiality. He was duly sentenced to death in New England in 1642 for having sired the abnormal pig. George Spencer was hanged. The sow was put to death by the sword.
3 Maternal Impressions
One common belief about malformations which originated very early and appears pervasive in many different cultures is the concept that events and images witnessed by a pregnant woman may somehow imprint themselves on the foetus (e.g. Warkany and Kalter 1962). A positive example of this is the advice given to pregnant women in the Greek city of Sparta, to admire statues of well-formed human beings. The converse idea, that viewing an abnormality can leave an imprint on the developing foetus by some sort of “photographic” effect, remained common until the late 19th century (Fisher 1870). In his book on medical curiosities Jan Bondeson (1997) extensively discusses these so-called maternal impressions. Bondeson relates the story of the Danish anatomist Bartholin who saw a girl with a cat’s head on a visit to Holland in 1738. The explanation given to Bartholin by the locals, was that a cat hiding in her mother’s bed, had dashed out unexpectedly and startled the pregnant woman. Bartholin and his colleague Jaccobaeus were influential at the Danish court. On their advice, King Frederik IV ruled that invalid and malformed people should be kept out of sight in a special hospital in Copenhagen. This was not out of pity for the poor and crippled, but to prevent pregnant women from bearing children exactly like them (Bondeson 1997). The last serious description of maternal impression (“Verzien” in Dutch) as a cause for malformation in the Dutch National Journal of Medicine occurred almost exactly 100 years ago (Formijne 1915). Occasional supporters of the concept remain among those who believe in parapsychology.
4 Infections and Teratogens
The discovery by Gregg in the early 1940s (Gregg 1947) that congenital rubella infection causes cataract, deafness, and other abnormalities and the description of severe malformations due to Thalidomide in the early 1960s by McBride in Australia (1961) and Lenz in Germany (1962), in conjunction with experimental work by Warkany in Cincinnati amongst others established the science of teratology, which studies the influence of harmful substances and infections on the foetus (Warkany and Nelson 1940). This concept of the foetus as a vulnerable developing human being inspired dramatic and effective improvements in prenatal care. It is now generally accepted that prenatal factors are responsible for malformations and handicaps in at most of 5 % in newborns in developed countries. In spite of the apparent rarity of teratogenic causes, all mothers of children with severe abnormalities or disabilities feel guilty. Many consider the possibility that something happened during pregnancy that harmed their child, which should have been avoided. In the case of intellectual disability, it is sometimes assumed that a lack of oxygen during delivery was responsible. However, it would seem that this is also rare and that it cannot begin to account for most cases of intellectual disability in the population at this time.
5 Inherited Factors
Inbreeding is an important factor for malformations, and intellectual disability. This reflects recessive inheritance where a child is affected because it received an abnormal gene from both parents. Because most deleterious gene variants are rare, the chance of these coming together in a child is very low, unless the parents are related. Thus, recessive inheritance has an important role in causing malformations and intellectual disability in countries with a high consanguinity rate. A recent study from the UK suggests that the risk of a baby having a malformation is approximately doubled from 3 to 6 % if the parents are first cousins (Sheridan et al. 2013). A recent study from Germany based on prenatal ultrasound scans came to much the same conclusion but the increase was about 3-fold, from 2.8 to 8.5 % for offspring from first-cousin marriages (Becker et al. 2015). No good estimates are available on their frequency, but there is good evidence for recessive inheritance of intellectual disability from populations with high rates of consanguinity such as Iran (Najmabadi et al. 2011).
The frequency of consanguinity varies enormously across the world, from less than 1 % of all marriage unions in the USA and Russia to over 50 % in Sudan and Pakistan (Romeo and Bittles 2014). This variation is tightly linked with customs and existing religious rules. Notably in Europe, the Roman Catholic church generally prohibited first-cousin marriages, while the protestants took a more liberal view. In the UK, following the marriage of Henry VIII first to his sister in law, Catherine of Aragon, and then to Anne Boleyn who was a cousin of his executed second wife, the church of England decided to legalize all first-cousin marriages (Bittles 2009). A dispute about the possible adverse effects of first-cousin marriage in Great Britain in the late 19th century was settled when George Darwin (son of Charles Darwin who married his first cousin Emma Wedgwood) produced evidence that the negative effects of first-cousin marriages were likely small (Darwin 1875; cited in Bittles 2009). Indeed we find that in outbred populations, the contribution of recessive inheritance to intellectual disability appears of modest importance(Gilissen et al. 2014; Deciphering Developmental Disorders Study 2015).
6 De Novo Mutations in Human Genetic Disease
Mutations are sudden changes in the genetic material. Mutations are the fuel of evolution, and therefore beneficial to the adaptation of species to changes in their environment (Crow 2000). Nonetheless, most mutations are either of no effect to the individual (neutral) or detrimental to health and survival. Truly beneficial mutations are clearly exceptionally rare events. Mutations can involve chromosomes, parts of chromosomes, or single genes.
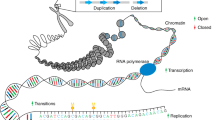
Chromosome abnormalities have been recognized as a cause of severe intellectual disability for many years at least since the discovery of trisomy 21 in Down syndrome 50 years ago. Chromosomal abnormalities are an important cause of severe intellectual disability and explain about 20 % of the total frequency. Techniques for the investigation of chromosomes have become better over time. Still, most individuals with severe intellectual disability have normal chromosomes even when studied by the best available techniques. Patients come from a normal pregnancy, normal birth and from normal families. For these reasons the most common answer to the question why a child has intellectual disability is “I don’t know”. The possibility to characterize the complete DNA sequence at the single base level by whole genome sequencing has radically changed this situation. It now turns out that most people with a severe intellectual disability do not have abnormalities of whole chromosomes. Some have very small chromosomal changes, but most have an abnormal single gene which has mutated (Gilissen et al. 2014). Similar findings have been reported for autism and schizophrenia but in a lower percentage. Analysis of the affected child and both parents demonstrates that the abnormality has arisen spontaneously in the child by a mutation of a single nucleotide in the DNA. This has important implications since DNA mutations are spread more or less equally across the genome, and occur at a relatively fixed rate of one per 100 million nucleotides per generation. Mutations represent random errors of replication during the formation of our germ cells. Thus, the majority of all instances of severe intellectual disability and a large proportion of other diseases such as autism, schizophrenia and birth defects are due to what seem to be essentially random events (Veltman and Brunner 2012).
7 The Randomness of Mutations
It has now been firmly established that the number of DNA mutations in a newborn child is approximately 100. Of these 100 mutations, on average 1 or 2 hit a gene. Since there are 20,000 genes, the impact of the single gene mutation that every newborn child has will be determined by the nature of the gene that was hit, and by the severity of the mutational event. Both of these factors are random. We may say, that the more we improve our lives, our habits, and our pregnancy care, the more the decision to start a family becomes similar to taking part in a genetic lottery. This comes as no great surprise to most parents. We all know and accept that each pregnancy carries risks. On the other hand, we do want explanations when a severely handicapped child is born. In my experience, the information that a disability is due to a chance event is perceived as good news by parents because it absolves them of feelings of guilt and insufficiency about how they handled their pregnancy.
8 Why Mutations Happen
There are two main causes of new mutations, insufficient DNA repair and random errors during DNA replication. DNA repair is necessary, because the DNA in our cells is under constant attack from external factors that may damage it. External damaging factors include radiation, chemicals, as well as various toxic substances that are generated by the cell itself such as oxygen radicals. To protect our existence, our cells have developed an elaborate system of DNA damage protection and especially DNA repair. This means that the large majority of DNA mutations is immediately corrected and repaired. Our germ cells seem to be especially good at preventing or repairing DNA damage. It was a striking and unexpected result from studies that were performed after the Nagasaki and Hiroshima atom bombs during World War II that there was only limited evidence for an increase of inherited genetic mutations. This is not to say that external factors are not relevant to new mutations. They are obviously very important but at the current level of exposure to noxious influences, they do not seem to be the determining factor whether or not a mutation ensues in a child. In fact, studies of the frequency of new mutations in children suggest a random distribution around the mean of 1–2 gene mutation per newborn individual. The driving force for the generation of new mutations is in the replication of DNA when our germ cells are created. Copying DNA is the essence of creating sperm cells and egg cells. All DNA nucleotides need to be copied with very high fidelity. Viewed like this, it is perhaps surprising that the total number of errors in a newborn is just 100 out of the 3 billion nucleotides of DNA that need to be copied. Mutations are a part of all life.
9 Can We Prevent Mutations?
If we view mutations as copy errors, then we must accept that it will not be easy to prevent them from happening. Consequently, it becomes quite difficult to further reduce the occurrence of severe handicaps and diseases. Once we have minimized the negative influences of DNA damaging substances and radiation, the remaining mutations are due to copy errors that reflect an intrinsic function of our cellular machinery. There may be a practical solution however. We may try to reduce the number of cellular divisions in the germ-line as much as possible. More de novo gene mutations happen during spermatogenesis than during oogenesis. This is because sperm cells continue to copy and then divide over a man’s lifetime while the egg cells are already completed by the time a girl is born. In fact, the mutation rate in the child is strongly dependent on the age of the father (Risch et al. 1987; Goriely and Wilkie 2012). While it is probably not practical to try and convince men to have their families young, it is a practical possibility to freeze and store sperm samples at a young age, and then use these later in life. While the impact on an individual may not be immediately apparent, it is clear that if this policy were universally adopted in the face of an increasing age at which men and women start their families, a society could reduce the burden of severe handicaps and autism by a large fraction. Whether this is acceptable or desirable is a different matter and will invite a vigorous societal debate.
10 Accepting Risks
Each pregnancy carries risks and this is a generally accepted fact. Because we cannot prevent mutations from happening, we cannot reduce or eliminate all risk, even if we live healthy lives and provide the best possible pregnancy care. Ultimately, early detection by prenatal diagnosis may be the only real option if we want to prevent severe handicaps. Whether this is acceptable in the form of universal prenatal diagnosis is again a matter for societal debate. It is clear that such discussions carry tremendous societal, ethical and emotional and even personal connotations and that they cannot be solved from the respective perspectives of biology, medicine or genetics. I believe that such a debate will take place over the coming years. In this respect, it may be instructive to read some of the reactions to a recent paper by cancer expert Vogelstein that suggests that most cases of cancer in Western populations are due to random mutations and that their risk is strongly related to the number of cell divisions per tissue (Tomassetti and Vogelstein 2015a). The authors concluded from their findings, that it is probably more worthwhile for society to try to detect cancers at an early stage than it is for society to invest in cancer prevention. Several commentators objected to this generalization, and partly for good scientific reasons. Nonetheless, the perceived dichotomy between external factors (and inherited predispositions) which we can avoid or ameliorate, and the randomness of mutations which strike from nowhere also seems to have inspired some of these comments. Or as Tomassetti and Vogelstein put it in their response: “Replicative mutations are unavoidable. They are in a sense a side-effect of evolution, which cannot proceed without them. That they play a larger role in cancer than previously believed has important scientific and societal implications.” (Tomassetti and Vogelstein 2015b).
All in all, the recent recognition that spontaneous mutations are an important driver of severe illnesses, such as intellectual disability, autism, schizophrenia, and cancer is likely to fuel another nature-nurture debate where random mutation events are contrasted with bad influences from the environment. Nature-nurture debates are never fully solved because the opposing sides are not ready to compromise. Still, such debates are always interesting and instructive, and in the end genome sequencing will provide us with real scientific data to weigh these two respective forces. At the end of the day, we need to come to terms with randomness as an integral part of our biology. This include accepting limits to the extent to which we can and cannot manage our existence.
11 Are Mutations a Necessary Part of Our Existence?
It is often argued that because mutations are the drivers of evolution, we should welcome them as a good thing. In general terms, advantageous mutations may indeed drive improved species adaptation and promote evolution. Nonetheless, since mutations may easily destroy the capacity of the organism to reproduce, there must be an upper limit to the number of random mutations a species can endure. In fact, in humans, the total number of copy errors in a newborn is just 100 out of the 3 billion nucleotides of DNA.
So is there an optimum rate for random mutational events, and how is this determined? First of all, it is clear that the answer to this question varies. In fact, frequency of random mutation can vary 100-fold between species, and each species has its own specific mutation rate. This species-specific mutation rate is not random, as it appears strongly dependent on the size of the genome, with bacteria having the lowest mutation rate and mammals having the highest mutation rate. All this suggests that for each species, there is a relatively constant and likely optimized error-rate of DNA replication.
So if our mutation rate is fixed, why is it what it is? In the absence of a divine plan, we may consider the following possibilities. First, it may be that our current human rate of evolution exactly matches the requirement for adaptation to a changing environment. If this were true then one would expect that there should be some variability of mutation rate within a species over evolutionary time. Simply put: In order to cope with changes in the selection regime, populations should evolve mechanisms that tune the rate of mutation, amongst other things, in order to increase their long-term adaptability (Carja et al. 2014). There is currently not a lot of evidence to support this idea, although it has recently been argued that there are data to support that the rate of human mutation may not be stable over time (Harris 2015).
Another possibility is that the mutation rate is as low as our species can afford. Keeping mutation rates low through high fidelity of DNA replication and reliable repair of mutations, is clearly a strategy that involves considerable cost to the organism. Since resources are limited, there may be a point where it becomes much more rewarding to species overall survival to stop investing in mutation prevention and repair, and rather divert resources and energy to other ways to promote survival and fitness. One weak spot in replication that has not been fixed by evolution, is to do with the defective proofreading capacity of polymerase alpha during replication (Reijns et al. 2012).
12 Conclusion
There may be an inherent tension between the interest of the individual and that of the species it belongs to as to the allowing of randomness. If we go by the “Adapt or die” paradigm, then we need random mutational events to survive as a species. But at the same time such random mutations may kill us before we reproduce. We need a bit of randomness in our existence otherwise our species cannot survive. But we need to dose this randomness very carefully or the resulting chaos will destroy us.
References
Baljet, B., & Oostra, R. J. (1998). Historical aspects of the study of malformations in The Netherlands. American Journal of Medical Genetics, 77(2), 91–99.
Becker, R., Keller, T., Wegner, R. D., Neitzel, H., Stumm, M., Knoll, U., et al. (2015). Consanguinity and pregnancy outcomes in a multi-ethnic, metropolitan European population. Prenatal Diagnosis, 35, 81–89.
Beckwith, J. B. (2012). Congenital malformations: From superstition to understanding. Virchows Archiv, 461, 609–619.
Bittles, A. H. (2009). Commentary: The background and outcomes of the first-cousin marriage controversy in Great Britain. International Journal of Epidemiology, 38, 1453–1458.
Bondeson, J. (1997). A Cabinet of Medical Curiosities. Ithaca: Cornell University Press.
Brent, R. L., & Fawcett, L. B. (2007). Developmental toxicology, drugs, and fetal teratogenesis. In E. Albert Reece & J. C. Hobbins (Ed.), Clinical obstetrics: The fetus and the mother. London: Wiley-Blackwell.
Carja, O., Liberman, U., & Feldman, M. W. (2014). Evolution in changing environments: Modifiers of mutation, recombination, and migration. Proceeding of National Academy of Science United States of America, 111(50), 17935–17940.
Crow, J. F. (2000). The origins, patterns and implications of human spontaneous mutation. Nature Reviews Genetics, 1, 40–47.
Deciphering Developmental Disorders Study (2015). Large-scale discovery of novel genetic causes of developmental disorders. Nature, 519, 223–228.
Darwin, G. H. (1875). Marriages between first cousins in England and Wales and their effects. Fortnight Review, 24, 22–41 (2009) (Reprinted in International Journal of Epidemiology 38, 1429–1439).
Fisher, G. J. (1870). Does maternal mental influence have any constructive or destructive power in the production of malformations or monstrosities at any stage of embryonic development? American Journal of Insanity, 26(24), 1–295.
Formijne, A. J. (1915). Het verzien van zwangeren. Nederlandsch Tijdschrift voor Geneeskunde, 59(1876–1), 877.
Goriely, A., & Wilkie, A. O. (2012). Paternal age effect mutations and selfish spermatogonial selection: Causes and consequences for human disease. American Journal of Human Genetics, 2(90), 175–200.
Gilissen, C., Hehir-Kwa, J. Y., Thung, D. T., van de Vorst, M., van Bon, B. W., Willemsen, M. H., et al. (2014). Genome sequencing identifies major causes of severe intellectual disability. Nature, 511, 344–347.
Gregg, N. M. (1947). Congenital defects associated with maternal rubella. The Australian Hospital, 14(11), 7–9.
Harris, K. (2015). Evidence for recent, population-specific evolution of the human mutation rate. Proceedings of the National Academy of Sciences United States of America, 112(11), 3439–3444.
Lenz, W., & Knapp, K. (1962). Thalidomide embrypopathy. Deutsche Medizinische Wochenschrift, 15(87), 1232–1242.
McBride, W. G. (1961). Thalidomide and congenital abnormalities. Letter to the Editor. The Lancet, 2, 1358.
Najmabadi, H., Hu, H., Garshasbi, M., Zemojtel, T., Abedini, S. S., Chen, W., et al. (2011). Deep sequencing reveals 50 novel genes for recessive cognitive disorders. Nature, 478, 57–63.
Pangas, J. C. (2000). Birth malformations in Babylon and Assyria. American Journal of Medical Genetics, 91(4), 318–321.
Reijns, M. A., Rabe, B., Rigby, R. E., Mill, P., Astell, K. R., Lettice, L. A., et al. (2012). Enzymatic removal of ribonucleotides from DNA is essential for mammalian genome integrity and development. Cell, 149(5), 1008–1022.
Risch, N., Reich, E. W., Wishnick, M. M., & McCarthy, J. G. (1987). Spontaneous mutation and parental age in humans. American Journal of Human Genetics, 41, 218–248.
Romeo, G., & Bittles, A. H. (2014). Consanguinity in the contemporary world. Human Heredity, 77, 6–9.
Sheridan, E., Wright, J., Small, N., Corry, P. C., Oddie, S., Whibley, C., et al. (2013). Risk factors for congenital anomaly in a multiethnic birth cohort: An analysis of the born in Bradford study. The Lancet, 382, 1350–1359.
Tigay, J. (1997). “‘He Begot a Son in His Likeness after His Image’ (Gen. 5:3),” in Tehillah le- Moshe. Biblical and Judaic Studies in Honor of Moshe Greenberg (pp. 139–147). In M. Cogan, B. L. Eichler & J. H. Tigay (Eds.). Winona Lake, Indiana: Eisenbraun’s.
Tomasetti, C., & Vogelstein, B. (2015a). Cancer etiology. Variation in cancer risk among tissues can be explained by the number of stem cell divisions. Science, 347, 78–81.
Tomasetti, C., & Vogelstein, B. (2015b). Cancer risk: Role of environment—response. Science, 347, 729–731.
Veltman, J. A., & Brunner, H. G. (2012). De novo mutations in human genetic disease. Nature Reviews Genetics, 13(8), 565–575.
Warkany, J., & Kalter, H. (1962, December). Maternal impressions and congenital malformations. Plastic and Reconstructive Surgery and the Transplantation Bulletin, 30, 628–637.
Warkany, J. (1959). Congenital malformations in the past. Journal of Chronic Diseases, 10(2), 84–96.
Warkany, J., & Nelson, R. C. (1940). Appearance of skeletal abnormalities in the offspring of rats reared on a deficient diet. Science, 92(2391), 383–384.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
<SimplePara><Emphasis Type="Bold">Open Access</Emphasis> This chapter is licensed under the terms of the Creative Commons Attribution-NonCommercial 2.5 International License (http://creativecommons.org/licenses/by-nc/2.5/), which permits any noncommercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made. </SimplePara> <SimplePara>The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.</SimplePara>
Copyright information
© 2016 The Author(s)
About this chapter
Cite this chapter
Brunner, H.G. (2016). When Chance Strikes: Random Mutational Events as a Cause of Birth Defects and Cancer. In: Landsman, K., van Wolde, E. (eds) The Challenge of Chance. The Frontiers Collection. Springer, Cham. https://doi.org/10.1007/978-3-319-26300-7_10
Download citation
DOI: https://doi.org/10.1007/978-3-319-26300-7_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-26298-7
Online ISBN: 978-3-319-26300-7
eBook Packages: Religion and PhilosophyPhilosophy and Religion (R0)