Abstract
Viruses of the Ebolavirus genus cause sporadic epidemics of severe and systemic febrile disease that are fueled by human-to-human transmission. Despite the notoriety of ebolaviruses, particularly Ebola virus (EBOV), as prominent viral hemorrhagic fever agents, and the international concern regarding Ebola virus disease (EVD) outbreaks, very little is known about the pathophysiology of EVD in humans and, in particular, about the human immune correlates of survival and immune memory. This lack of basic knowledge about physiological characteristics of EVD is probably attributable to the dearth of clinical and laboratory data gathered from past outbreaks. The unprecedented magnitude of the EVD epidemic that occurred in West Africa from 2013 to 2016 has allowed, for the first time, evaluation of clinical, epidemiological, and immunological parameters in a significant number of patients using state-of-the-art laboratory equipment. This review will summarize the data from the literature regarding human pathophysiologic and immunologic responses to filoviral infection.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
1 Introduction
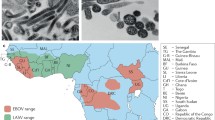
Ebola virus (EBOV) is the prototypic member of the Ebolavirus genus in the Filoviridae family of negative-sense, single-stranded RNA viruses. Discovered in 1976 during the first documented outbreak of Ebola virus disease (EVD) in the town of Yambuku in northern Zaire (today Democratic Republic of the Congo), EBOV has since caused sporadic human disease outbreaks of varying magnitude in Equatorial African countries (Sanchez et al. 2007a). In March 2014, an EBOV variant later named EBOV Makona was first detected in Guinea. This variant was responsible for a 3-year-long epidemic that affected tens of thousands of people in several West African countries, collapsing the healthcare systems of three of them. EBOV Makona rampaged through both rural and urban areas, and underscored previously poorly characterized features of EVD, like sexual transmission and virus persistence after recovery (Bausch et al. 2007; Rowe et al. 1999; Chughtai et al. 2016; Deen et al. 2015; Fischer et al. 2016; Rodriguez et al. 1999; Varkey et al. 2015; Uyeki et al. 2016a).
The scientific and clinical knowledge of human EVD before its appearance in West Africa was very limited. The scarcity of human cases and their occurrence in rural areas of Equatorial Africa limited research, as did confinement of filovirus research to biosafety level 4 containment laboratories. In addition, basic studies on EVD pathophysiology have been hampered by the lack of susceptible small animal models with competent immunity. For example, laboratory mice, a commonly used disease model, are completely resistant to nonadapted EBOV.
Before 2014, EVD was described as an acute hemorrhagic fever, thus earning its former name Ebola hemorrhagic fever (EHF); case fatality rates of up to 90% had been reported. The disease was characterized by lymphopenia, disseminated intravascular coagulation (DIC), immunosuppression, and a systemic inflammatory response resembling septic shock (Feldmann and Geisbert 2011). While many of these observations have been strengthened by findings from the West African EVD outbreak, some of the previous hypotheses have been revised. Perhaps one of the most surprising findings has been the low overall number of human cases presenting with bleeding (Schieffelin et al. 2014), as well as the lack of correlation between bleeding and disease severity (Schieffelin et al. 2014; McElroy et al. 2014a, b). These findings triggered the change in disease nomenclature from Ebola hemorrhagic fever to Ebola virus disease. Moreover, the finding that EVD correlates with robust immune activation rather than immunosuppression (Ruibal et al. 2016; McElroy et al. 2015a), and the ability of the virus to persist in several body fluids long after recovery (Varkey et al. 2015; Uyeki et al. 2016a; Sow et al. 2016; Green et al. 2016; Deen et al. 2015) have changed our current view of EVD and have prompted new directions in research and new public health policies. Here we will aim to integrate these novel findings within the current human EVD model, and will discuss future research directions.
Several ebolaviruses cause EVD, and while differences may exist between the diseases caused by the individual viruses, this review will focus on EVD as a disease caused by all known viruses in the Ebolavirus genus (ebolaviruses) that are pathogenic for humans. The reader will note that most of the available data come from infections caused by EBOV rather than the other pathogenic viruses in this genus: Sudan virus (SUDV), Bundibugyo virus (BDBV), and Taï Forest virus (TAFV). The related marburgviruses, Marburg virus (MARV) and Ravn virus (RAVV), will be mentioned where appropriate data are available, but unfortunately, information on Marburg virus disease (MVD), which is caused by both of them, is still lacking.
2 Portals of Ebolavirus Entry
Epidemiological data collected over the last 40 years indicate that human infection with EBOV occurs mainly through close contact with infected body fluids. This probably occurs during both spillover events (e.g., contact with infected blood during butchering of bushmeat) and human-to-human transmission. There is no evidence that direct contact with bats causes EBOV spillover into humans (Mari Saez et al. 2015; Leroy et al. 2009), but infection with MARV and RAVV via direct or indirect contact with Egyptian rousettes (fruit bats of the species Rousettus aegyptiacus) (Amman et al. 2012; Schuh et al. 2017), has been documented. Human visits to caves or mines in which these bats roost have been directly associated with the development of MVD (Bausch et al. 2003; Centers for Disease and Prevention 2009; Adjemian et al. 2011), strongly indicating that mucosal or skin contact with bat droppings is sufficient to initiate MARV infection in humans.
With the exception of the first EVD outbreaks in Zaire, which were linked to substantial percutaneous needle transmission (Ebola haemorrhagic fever in Zaire 1976/1978), most of the data since the early 1990s suggest that exposing skin and mucosae to EBOV while conducting activities like body washing during traditional funerals or caring for sick relatives in the household is sufficient for human-to-human transmission of EBOV (Bausch et al. 2007; Dowell et al. 1999; Francesconi et al. 2003). Early data collected from a laboratory exposure to SUDV even suggest that skin abrasions may not be necessary to allow ebolavirus entry through the skin (Emond et al. 1977). These findings raise questions regarding how ebolavirus infection takes place in skin and mucosae, and which cells are involved in the primary amplification of the virus.
Antigen-presenting cells are a putative initial target of EBOV infection and previous research in animal models of disease has indicated that dendritic cells (DCs) and macrophages are early and preferred targets of EBOV and support virus replication (Geisbert et al. 2003a). Both DCs and macrophages can also be productively infected by EBOV in vitro (Gupta et al. 2001, 2007; Mahanty et al. 2003; Bosio et al. 2003), and EBOV prevents activation of in vitro-derived DCs, mainly through the action of VP24 and VP35 (Yen et al. 2014; Jin et al. 2010; Ilinykh et al. 2015).
To further complicate things, a great deal of research over the last decade has been devoted to defining the ontogeny and specific function of DC subsets in mice and humans. The emerging picture is that several cell subsets exist with overlapping and nonoverlapping functions, and these subsets can be roughly classified into classical, plasmacytoid, and inflammatory DCs in humans (see (Haniffa et al. 2013) for an excellent review). Whether EBOV can equally infect different DC subsets is not known, but some of the existing evidence suggest that it cannot (Leung et al. 2011). For example, a number of cellular receptors have been involved in the attachment of EBOV virions to target cells. These receptors include several C-type lectins present on the surface of DCs, such as dendritic cell-specific ICAM-3-grabbing non-integrin (DC-SIGN) (Simmons et al. 2003) and liver/lymph node SIGN (L-SIGN) (Alvarez et al. 2002). DCs of the epidermis and mucosal epithelium do not express these molecules, but Langerhans cells in the skin and CD141+ DCs in mucosal epithelium do express the C-type lectin langerin (Merad et al. 2008). In fact, studies in monkeys and pigs have indicated that DC-SIGN+ cells are scarce in the dermis and the lamina propria or submucosa in the steady state (Schwartz et al. 2002; Huang et al. 2009), suggesting that other cell types may be targets for early EBOV replication. Initiating EBOV infection may depend on attachment to target cells via TIM-1 and TIM-4, which are highly expressed in mucosal epithelia (Rhein et al. 2016; Kondratowicz et al. 2011). Initial virus amplification could then lead to inflammation and infiltration of a high number of myeloid cells expressing DC-SIGN and other described EBOV attachment factors, like triggering receptor expressed on myeloid cells 1 (TREM-1) expressed by neutrophils (Mohamadzadeh et al. 2006), and human macrophage C-type lectin specific for galactose/N-acetylgalactosamine (hMGL) expressed by macrophages (Takada et al. 2004). The elucidation of the initial steps by which EBOV establishes productive infection in a host organism is highly needed to understand the mechanisms by which the virus disseminates from the initial site of entry to the body, and perhaps to design medical countermeasures aimed at preventing virus spread.
3 Virus Dissemination and Initiation of EBOV-Specific Immunity
As mentioned above, DCs and macrophages are early targets of EBOV infection. Due to the migratory potential of DCs, these immune cells may participate in disseminating EBOV from the initial points of entry to the draining lymph nodes (Geisbert et al. 2003a). This strategy is commonly used by other viruses, including SARS coronavirus (Liu et al. 2015), Toscana virus (Cusi et al. 2016), and measles virus (Mesman et al. 2012), for dissemination in the host. However, perhaps due to the lack of suitable in vivo models for kinetic studies of EBOV, the involvement of DCs in EBOV dissemination has not been experimentally addressed. In any case, the specific subsets of cells responsible for EBOV dissemination remain to be identified. It is plausible that tissue-resident DCs or inflammatory DCs derived from infiltrating monocytes are important for EBOV dissemination. Both myeloid DC populations are migratory and can transport a variety of antigens from inflamed tissues to the draining lymph nodes (Leon et al. 2007; Ersland et al. 2010). Conversely, macrophages and neutrophils are less likely to participate in EBOV dissemination due to their low mobility and nonproductive infection, respectively (Mohamadzadeh et al. 2006).
Recent studies have demonstrated that human EVD is associated with loss of peripheral blood monocytes, in particular nonclassical CD16+ monocytes (Ludtke et al. 2016) which have been proposed as the main antiviral monocyte subset (Cros et al. 2010). Even though this study did not demonstrate direct infection of CD16+ by EBOV, it raised the possibility that this cell subset could be involved in virus dissemination. In fact, CD16+ monocytes are also called patrolling monocytes due to their ability to attach to endothelial cells in a LFA-1-dependent manner and to extravasate into inflamed tissues where they differentiate into inflammatory DCs and macrophages (Cros et al. 2010; Auffray et al. 2007). This hypothesis is also substantiated by a previous study that demonstrated that EBOV particles can attach to monocytes and enter these cells only when the monocyte differentiation program has started, that is, during their differentiation into DCs and macrophages in inflamed tissues (Martinez et al. 2013).
The identification of the DC subsets specifically involved in filovirus dissemination is a highly relevant topic of study, because the function of DCs can be enhanced or inhibited, ex vivo or in vivo, by antigen delivery or use of molecules and antibodies. Therefore, DCs are putative immunotherapeutic targets for postexposure EVD treatment (Klechevsky and Banchereau 2013). For example, the ligand of the DC co-stimulatory molecule CD40 (sCD40L) is commonly used to enhance DC-mediated antigen presentation (Kornbluth and Bot 2012), and previous studies have demonstrated a correlation between circulating levels of sCD40L and survival after SUDV infection (McElroy et al. 2014a, b). In addition, poor activation profiles of circulating antigen-presenting cells have been correlated with severe EVD (Ludtke et al. 2016). These findings provide a rationale for the use of DC enhancers as immunotherapy candidates in filovirus disease.
Another key driver of EBOV dissemination may be the cytokine microenvironment, since these innate immune signaling molecules play an important role in recruiting myeloid cells, which are putative EBOV targets, to sites of inflammation. A considerable body of research exists on cytokine and chemokine responses during EVD. Despite some conflicts, in general, these data correlate fatal outcomes during EVD with high concentrations of pro-inflammatory cytokines (e.g., IL-6), pro-inflammatory chemokines (e.g., IP-10), and anti-inflammatory cytokines (IL-RA and IL-10), overall suggesting a general dysregulation in the expression of these key immune signaling molecules (Hutchinson and Rollin 2007; Gupta et al. 2012; Wauquier et al. 2010; Baize et al. 2002; Villinger et al. 1999). An inability to control viral replication is likely leading to continued innate immune stimulation. Data from asymptomatic human cases have shown an even greater magnitude of cytokine and chemokine upregulation, followed by rapid downregulation of this response in association with control of viral replication (Leroy et al. 2000, 2001), suggesting that cytokine/chemokine dysregulation is a consequence of uncontrolled viral replication rather than a primary mediator of pathogenesis.
Type I interferons (IFN-I) are key antiviral cytokines, and perhaps one of the more conflicting aspects of comparing EBOV experimental and clinical human data is the role of IFN-I in EBOV immunity and pathogenesis. Importantly, IFN-I not only induces an antiviral state in infected and bystander cells during early virus infection, but also is a key modulator in the transition between innate and adaptive immunity. By enhancing natural killer cell function, antigen presentation by DCs, and expansion of effector T cells, IFN-I bridges natural and acquired antiviral immunity (see (McNab et al. 2015) for a review), so elucidating its functions during human EVD is highly relevant to understanding disease pathogenesis. IFN is critical in protecting laboratory mice from EBOV (Brannan et al. 2015; Bray et al. 2002), but data are somewhat conflicting in nonhuman primates (NHPs) and humans (McElroy et al. 2016; Smith et al. 2013; Villinger et al. 1999; Yen et al. 2011). Higher levels of IFN-alpha were associated with fatal EVD cases (Villinger et al. 1999), but higher IFN-beta was associated with less severe EVD (McElroy et al. 2016) and IFN-beta administration prolonged survival in nonhuman primates (Smith et al. 2013). However, as IFN responses are highly dynamic, drawing conclusions regarding human pathogenesis is difficult. For example, EVD survivors may mount early and robust IFN responses that keep viral replication at bay, while patients who succumb to EVD may display higher IFN levels later on due to increased viral replication and inflammation.
Interestingly, recent studies have shown that patients who survive and patients who succumb to EVD both show robust T cell activation (McElroy et al. 2015a; Ruibal et al. 2016). Since DCs are the only antigen-presenting cells capable of priming naïve T cells (Banchereau and Steinman 1998), these results suggest at least two possibilities. On one hand, infected DCs may retain their capacity to initiate T cell-specific responses, as has been shown in other viral infections (Wahid et al. 2005; Rivera and McGuire 2005; Kvale et al. 2006). On the other hand, some DC subsets may be spared from infection and thus able to prime EBOV-specific T cells. The generation of this EBOV-specific adaptive immunity is the topic of the next two sections.
4 Adaptive Immunity: Human Antibody Responses
While innate immune responses may play a chief role in controlling early EBOV replication in humans, the current model identifies the character (though not necessarily the magnitude) of adaptive immunity as the main factor driving viral clearance and recovery. Both humoral and cellular immunity seem to be required for EBOV clearance in humans, a hypothesis strengthened by the finding that EVD patients mount robust adaptive immune responses (McElroy et al. 2015a; Ruibal et al. 2016) with high numbers of circulating plasmablasts and EBOV-specific T cells.
The more difficult task is assessing whether adaptive immune responses mark substantial differences between fatal and surviving patients. Initial studies supported the idea that early development of IgM and isotype switching to IgG correlated with positive outcome. Indeed, a high percentage of patients with fatal outcomes do not seem to develop IgM (Ksiazek et al. 1999). These field studies are also in agreement with findings in patients evacuated into Europe or the US for medical treatment during the recent West African EVD outbreak. Surviving EVD patients mounted early IgM responses and showed upregulation of serum IgG over the course of the disease, which was correlated with viral clearance (Kreuels et al. 2014; Wolf et al. 2015). Conversely, deficient or diminished IgM and IgG responses have been reported in fatal cases of both EVD and MVD (van Paassen et al. 2012; Baize et al. 1999).
However, limited field data also indicate survivors who did not develop IgG, as well as patients who died after developing detectable circulating anti-EBOV antibodies (Onyango et al. 2007). In addition, limited clinical data obtained from the SUDV-caused EVD outbreak in Gulu, Uganda, did not reveal significant differences between the humoral responses in fatal and nonfatal EVD, with very late expression of IgG in both groups that was unrelated with viral clearance (Towner et al. 2004). As in many other aspects of EVD immunology, the kinetics of antibody responses in a statistically relevant cohort of acute-stage patients with defined outcomes must be studied.
Antibodies play many roles during the immune response to pathogens, including neutralization and antibody-dependent killing of virus-infected cells by targeting them to Fc receptor bearing cells (ADCC) or complement (CMC). Neutralizing antibodies (NAB) probably play a small role in recovery from acute EVD, since in many survivors NAB are not detectable until weeks or even months after recovery (Luczkowiak et al. 2016; Sobarzo et al. 2012). This is a puzzling and as yet unexplained finding. One of the plausible hypotheses is that disrupting lymphoid architecture during acute EVD infection may compromise germinal center formation and B cell affinity maturation, a feature that has been observed during Lassa fever (Carrion et al. 2007). However, this hypothesis does not reconcile easily with the levels of circulating plasmablasts in patients in the acute stage of illness (McElroy et al. 2015a), or with the limited focal necrosis observed in human biopsy samples of lymphoid tissues (Martines et al. 2015). One interesting possibility is that the long filament shape of filovirus virions may require a highly diverse repertoire of NAB for effective neutralization. In fact, studies have found greater B cell clonality in EVD survivors than in individuals with B cell memory against HIV-1 or influenza A virus (Bornholdt et al. 2016b). Still, this cannot be the whole story, as similar delays in NAB production have been described in other viral hemorrhagic fevers (e.g., Lassa fever). Perhaps more importantly, delayed NAB production strongly suggests long-term virus or antigen persistence, which is in agreement with duration of post-EVD sequelae (see below).
Nevertheless, long-term survivors develop effective NAB, mainly directed against several epitopes of the ebolaviral GP1,2 (Bornholdt et al. 2016a, b; Misasi et al. 2016; Corti et al. 2016). Many of the described NAB isolated from survivors are directed against the GP1,2 glycan cap as well as against the region bridging GP1 and GP2, which seem to be epitopes amenable for antibody-based therapeutics like ZMapp (Murin et al. 2014). Importantly, a number of studies in surviving patients have highlighted the presence of naturally occurring NAB with cross-reactivity against other ebolaviruses and even against MARV (Olal et al. 2012; Misasi et al. 2016; Bornholdt et al. 2016a). These findings strongly suggest that antibody-mediated immune memory may provide long-term protection against secondary infection with filoviruses, and may have important implications for public health measures (e.g., recruiting survivors as caregivers in future outbreaks).
Currently, the relative importance of NAB versus other antibody-mediated mechanisms, such as ADCC and CMC, is unclear, even though most protective antibodies probably act through both neutralization and ADCC/CMC activation (Schmaljohn and Lewis 2016). Antibodies with both neutralizing and ADCC capacity have been detected in EVD survivors more than a decade after recovery (Corti et al. 2016), and ADCC is probably an important mechanistic feature of ZMapp (Olinger et al. 2012). In addition to dissecting whether or not neutralization and ADCC specifically contribute to EVD immunity, it is important to determine the kinetics of antibody-mediated immunity from acute infection to long-term recovery. Lack of information on EVD antibody kinetics and EBOV-specific quantitative activity is probably largely responsible for the lack of protective effect demonstrated by convalescent plasma therapy (van Griensven et al. 2016). This therapeutic strategy has putative applicability for field outbreak conditions, but requires characterization of the virus specific activity in the product as well as optimization to ensure transfer of sufficient quantities of protective antibodies.
5 Adaptive Immunity: Human T Cell Responses
T cells, particularly CD8 T cells, are essential for clearance of acute viral infections. Naïve T cells react to stimulation with pathogen-specific peptides by massively expanding, differentiating into effector cells and migrating to peripheral infection sites for elimination of infected cells (see (Zhang and Bevan 2011) for a review). Because naïve CD8 T cells can be activated only by DCs (Banchereau and Steinman 1998), the initial assumption was that EBOV-induced DC inactivation would in turn result in poor T cell priming and overall inability of the host to eliminate infection. This hypothesis was substantiated by early studies demonstrating that, despite being spared from infection, many T cells underwent apoptosis during human EVD (Baize et al. 1999; Wauquier et al. 2010). While this observation is still valid, data gathered mainly during the recent West African outbreak have suggested that lymphocytes in general display very dynamic kinetics during EVD, which may include early proliferation followed by lymphopenia (Kreuels et al. 2014; Wolf et al. 2015). Similar dynamics occur during other systemic viral infections and during pro-inflammatory disorders such as sepsis (Luan et al. 2015), suggesting that perhaps lymphopenia is not a differential characteristic of EVD.
A substantial difference between earlier studies and those carried out in the context of the recent outbreak has been the application of multiparametric flow cytometry, which has allowed for the first time collection of phenotypic and functional information from single cells in unprecedented detail. These studies have revealed that, in fact, EVD is characterized by massive T cell activation rather than inhibition in both surviving and fatal cases. Co-expression of activation markers such as CD38 and HLA-DR, as well as proliferation markers like Ki-67, were detected in a significant percentage of CD4 and CD8 T cells in EVD patients (McElroy et al. 2015a; Ruibal et al. 2016) and were comparable with the magnitude of activation observed in other acute infections or after vaccination (Lindgren et al. 2011; Miller et al. 2008). Of note, since co-expression of CD38 and HLA-DR is correlated with engagement of the T cell receptor (Appay et al. 2002), these findings strongly suggest that proper T cell priming by antigen-presenting cells occurs during EVD in humans. Additionally, these findings were comparable between patients receiving experimental therapy (McElroy et al. 2015a) and those who received supportive care in the field (Ruibal et al. 2016), indicating that robust T cell activation is a characteristic of EVD unrelated to treatment.
A paramount question, therefore, is why robust T cell activation does not lead to viral clearance during EVD. To some extent, this lack of T cell effectiveness may be related to defects in negative immune checkpoints, namely the molecular mechanisms that control the transition from activation to immune homeostasis and that are essential for autoimmune control (Buchbinder and Desai 2016). Two such mechanisms are triggered by the T cell co-inhibitor molecules, programmed cell death-1 (PD-1) and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4). An earlier review already hypothesized that T cell dysfunction during filovirus infection could be related to high expression of PD-1 and CTLA-4 in T cells (Mohamadzadeh et al. 2007), which leads to a nonfunctional but reversible status termed T cell exhaustion (Wherry 2011). Studies from the recent West African EVD outbreak found that peripheral blood T cells from EVD patients expressed high levels of PD-1 and CTLA-4 (McElroy et al. 2015a; Ruibal et al. 2016), which were significantly higher in fatal cases (Ruibal et al. 2016). As a follow-up to these observational studies, determining the correlation between high expression of T cell inhibitory molecules and T cell function and apoptosis will be important. Determining this correlation will most likely require relevant in vivo models that can reproduce this T cell phenotype. Utilizing immunotherapeutic approaches to block PD-1 and CTLA-4 function during postexposure filovirus infection treatment may provide an interesting opportunity. Several therapeutic products are licensed extensively to block PD-1 and CTLA-4 in several types of cancer, thereby restoring T cell function (see (Sharma and Allison 2015) for a review).
Another important and related question is whether broad and polyfunctional T cell responses lead to increased disease manifestations, like in hantavirus (cardio)pulmonary syndrome (Terajima and Ennis 2011), or to decreased susceptibility, as in dengue virus 1–4 infections (Weiskopf et al. 2013). To answer this question, an exhaustive analysis of EBOV T cell immunodominance in humans must be performed, which is still not available. Previous evidence shows that the viral nucleoprotein (NP) drives most of the CD8 T cell response (McElroy et al. 2015a; Sundar et al. 2007; Wilson and Hart 2001). This finding is consistent with the observation that HLA alleles recognizing conserved filovirus NP epitopes provide protection against SUDV infection (Sanchez et al. 2007b). Additional studies of HLA association with EVD outcomes in a statistically significant cohort of patients are highly needed to strengthen these initial observations. The finding that NP drives most of the CD8 T cell response also has significant implications for vaccine design and may explain, at least to some extent, why most GP1,2-based vaccines induce poor T cell immunity (Agnandji et al. 2016; Ewer et al. 2016; Zhu et al. 2015).
6 Electrolyte Imbalances
The degree to which electrolyte abnormalities contribute to EVD pathogenesis was not appreciated in earlier outbreak responses because real-time serum electrolyte data were not available. The ability to acquire these measurements in patients during the West African EVD outbreak, as well as the degree of profuse watery diarrhea that was reported, have brought to the forefront the severity of electrolyte imbalances in EVD and the impact electrolytes could have on patient outcome. Such data were first collected during the SUDV outbreak in Gulu, Uganda, in 2000–2001, during which elevated BUN/Cre levels and hypocalcemia were associated with severe disease and fatal outcomes (Rollin et al. 2007). Data from African cohorts (Hunt et al. 2015) combined with data from repatriated patients who were cared for in developing nations (Uyeki et al. 2016b) have revealed potassium abnormalities, hyponatremia, hypomagnesemia, and hypocalcemia. Some of these alterations may be related to acute renal injury that is also common among severely ill patients; others might be related to volume and electrolyte imbalance secondary to profuse watery diarrhea. The clinical consequences of electrolyte imbalances could include cardiac arrhythmias, seizures, or coma. Indeed, 41% of the repatriated patients exhibited arrhythmia or electrocardiographic changes, one patient had seizures, and three were in a coma (Uyeki et al. 2016b). Electrolyte levels are easily measured blood chemistry parameters that can be corrected with electrolyte and fluid administration. Such measures may have contributed to improved outcomes during the recent outbreak, as one EVD treatment center that incorporated these data into patient management had a case fatality rate of only 37%, significantly lower than the 50 and 74% rates reported from two other treatment centers in Sierra Leone (Hunt et al. 2015; Schieffelin et al. 2014; Lanini et al. 2015).
7 Endothelial Dysfunction
Multiple lines of evidence have suggested that the endothelium is dysfunctional during EVD. While endothelial cells are directly infected, they do not show significant cytopathic effect, and endothelial infection is thought to occur during the terminal phase of the illness (Martines et al. 2015; Geisbert et al. 2003c). The overall dysfunction of the endothelium is thought to be an indirect effect of pro-inflammatory cytokines like TNF-alpha (Villinger et al. 1999; Feldmann et al. 1996), or other molecules, like nitric oxide, that increase the permeability of the endothelium during inflammation (Sanchez et al. 2004). Increased levels of several pro-inflammatory cytokines and chemokines are associated with EVD-related deaths (Baize et al. 2002; Wauquier et al. 2010; Gupta et al. 2012; Hutchinson and Rollin 2007; McElroy et al. 2014a, b). Infected antigen-presenting cells, such as macrophages, DCs, or monocytes are the presumed source of these cytokines (Feldmann et al. 1996; Gupta et al. 2001), and these cytokines lead to endothelial activation.
Increased vascular permeability due to loosening of the endothelial barrier is a normal and necessary physiologic function that allows cells and biomolecules to reach sites of inflammation, but widespread activation in many inflammatory diseases results in fluid movement that can be detrimental to the host. Clinical and laboratory findings in EVD, including tachypnea (with or without pulmonary edema), hypotension, oliguria, tachycardia, impaired distal perfusion, hypoalbuminemia, and hemoconcentration, are consistent with fluid extravasation into extravascular spaces secondary to increased vascular permeability (Rollin et al. 2007; Uyeki et al. 2016b; Hunt et al. 2015; Chertow et al. 2014). This constellation of clinical findings is thought ultimately to lead to hypovolemic shock in fatal cases.
In recent years, additional evidence that dysfunctional endothelia contribute to the disease process include the findings of increased levels of sICAM, thrombomodulin, PE-CAM, and P-selectin in patients with severe or fatal disease (McElroy et al. 2014a, b). All of these biomarkers, when released into the plasma, indicate an activated endothelium and/or breakdown of endothelial intercellular junctions. An activated endothelium is both pro-inflammatory and pro-coagulant, and likely contributes both to the ongoing inflammatory response that characterizes severe EVD and to the coagulopathy that has been observed in some patients (discussed in more detail below).
Also noteworthy is the sometimes conflicting body of evidence implicating the viral glycoprotein (GP1,2) in endothelial dysfunction. The EBOV GP gene coding region produces two proteins based on a transcriptional editing site, the soluble GP (sGP) and the full-length structural GP1,2 (Sanchez et al. 1996). The full-length GP1,2 produced by pseudotyped retrovirus or virion-like particles (VLPs) can bind to and activate endothelial cells, leading to increased endothelial permeability (Wahl-Jensen et al. 2005; Yang et al. 1998). sGP has been detected in the plasma of infected individuals (Sanchez et al. 1999), and, in fact, inhibits TNF-mediated increases in vascular permeability in vitro, perhaps suggesting a compensatory mechanism to control virus-induced inflammation. A third form of the protein, known as shed GP, is shed from the surface of infected cells in vitro and increases permeability of cultured endothelial cells (Escudero-Perez et al. 2014). While shed GP was detected in infected guinea pigs, it has not yet been detected in vivo in humans. Finally, overexpression of GP1,2 in explanted human, porcine, or NHP blood vessels leads to increased endothelial permeability and endothelial cytotoxicity mediated by the mucin domain of the protein (Yang et al. 2000). The relevance of this finding to EVD is unclear, since endothelial cells are infected long after endothelial function has already been compromised, and do not show cytopathic effects when infected in vitro (Geisbert et al. 2003c). Taken together, these data suggest that the various forms of EBOV GP may modulate endothelial function, but the precise role of the protein in human EVD pathogenesis is unclear.
8 Coagulopathy
The moniker “viral hemorrhagic fever” was applied to EBOV EVD during the first outbreak identified in 1976, and was appropriate because 78% of fatal cases had hemorrhagic manifestations, mostly melena (Ebola haemorrhagic fever in Zaire 1976/1978). This 1976 outbreak was unique, because the route of virus transmission was via injection in approximately one-third of the 288 patients, and this mode of entry could have contributed to the manifestations and severity of disease (the authors note that all patients who were infected by injection died). Notably, a concurrent outbreak of SUDV-caused EVD also had high frequencies (71%) of hemorrhagic manifestations. However, in several of the larger subsequent outbreaks where appropriate data were available, significantly fewer patients had hemorrhagic manifestations of disease: 41% (EBOV 1995), 30% (SUDV 2000), and 47% (BDBV 2007) (Okware et al. 2002; MacNeil et al. 2010; Bwaka et al. 1999). Additionally, in these three outbreaks, no association was observed between bleeding and death, arguing against the commonly held belief that hemorrhage equates to a fatal outcome. Furthermore, in the Western African outbreak, hemorrhagic manifestations were rarely reported; fewer than 15% of all patients from Liberia and Sierra Leone had any bleeding symptom recorded (Chertow et al. 2014; Schieffelin et al. 2014; Lado et al. 2015; Yan et al. 2015; Dallatomasina et al. 2015; Li et al. 2016; Qin et al. 2015), but two reports from a single center in Guinea reported bleeding in 51 and 26% of patients (Barry et al. 2014; Bah et al. 2015). Perhaps reported differences in hemorrhage frequency are related to genetic or nutritional factors that cannot be controlled for in observational reports. Regardless, hemorrhaging can occur during EVD, but is not the most prominent feature. In contrast, hemorrhage does seem to be a common (50–80% of patients) feature of MVD, based upon limited data from the two largest outbreaks to date (Colebunders et al. 2007; Roddy et al. 2010).
Hemorrhaging is a clinical sign that can be secondary to multiple types of hematologic disorders. In the simplest terms, two general categories of hematologic disorders manifest clinically as bleeding: low levels of platelets and coagulation factor deficiencies (Hunt 2014). Platelet counts have not been routinely measured in patients with EVD, but in one study of 150 patients with EVD during the West African outbreak, platelet counts were not especially low, ranging 119–247 × 109/L (normal range is 150–300) (Hunt et al. 2015). Interestingly, the same phenomenon was noted years ago in the NHP model, and while absolute platelet counts were not very low, platelet function was severely affected as a result of in vivo activation and degranulation (Fisher-Hoch et al. 1985). No measurements of platelet function have been reported to date in humans. However, elevated levels of sCD40L were observed in surviving patients with EVD caused by SUDV (McElroy et al. 2014a, b). Since platelets are the major source of sCD40L in the bloodstream (Henn et al. 2001), this finding suggests platelet activation in humans during EVD. These data suggest a process that consumes platelet functional activity in severe or fatal EVD; this process would be consistent with the finding that in lethal NHP studies of EVD, sCD40L levels are elevated initially, but decline to undetectable at the time of death (Ebihara et al. 2011).
MARV might be a bit different, since in the original report of the first outbreak in Europe in 1967, most patients had severe thrombocytopenia, sometimes less than 10 × 109/L (Martini 1973), coincident with significant hemorrhage in about half of the patients. Finally, the type of bleeding often described in EVD (and MVD) patients—epistaxis, conjunctival hemorrhages, bleeding into the GI tract and from the oral cavity—is mostly mucosal in nature, consistent with loss of platelet numbers or function.
The second general category of hematologic disorders that manifest as bleeding is deficiency in coagulation factors. Coagulation factors are quantitated clinically by measuring partial thromboplastin time (PTT) and prothrombin time (PT) to evaluate the intrinsic and extrinsic coagulation pathways. Unfortunately, these measurements have only been reported in case studies, and provide no consensus regarding the levels of PT and PTT during EVD. This is a clear information gap that needs to be addressed. An early report on MARV states that PTT and PT were measured in 10 patients, but the values obtained did not explain the observed severity of the hemorrhaging (Martini 1973).
DIC is often seen in critically ill patients, especially those with sepsis, and involves both low platelet counts and coagulation factor deficiencies. The bleeding seen in patients with EVD is often reported as due to DIC, although whether the criteria for DIC are met is unknown because the necessary laboratory tests are not routinely available. DIC laboratory features include thrombocytopenia, elevated fibrin split products, prolonged PT, and consumption of fibrinogen (Levi et al. 2009). As noted above, the level of thrombocytopenia seen in EVD patients rarely meets the criteria to assign a DIC score, but elevated fibrin split products (such as D-dimer) have been measured retrospectively, are elevated in EVD patients, and are associated with fatal outcomes (Rollin et al. 2007). PT measurements are normal in the few available case reports (Sueblinvong et al. 2015), and fibrinogen levels were not associated with outcome or hemorrhagic manifestations (McElroy et al. 2014a, b, 2016). Measurement of DIC markers is clearly an area that requires additional study for clarification.
Less conventional evaluations of factors involved in coagulation pathways have also been conducted. Thrombomodulin, a protein expressed on endothelial cells, has anticoagulant properties in the microenvironment of the cell surface. When present in the plasma, thrombomodulin can act more globally, as shown in one family with a genetic deficiency that results in elevated levels of free plasma thrombomodulin in association with a bleeding disorder (Langdown et al. 2014). Endothelial cells also release thrombomodulin when they become activated. Elevated plasma levels of thrombomodulin were associated with both hemorrhage and death in SUDV patients, and with more severe disease in a cohort of EVD patients (McElroy et al. 2014a, b, 2016), suggesting that loss of this protein from the endothelial surface exacerbates both endothelial dysfunction and coagulopathy during EVD. Additionally, tissue factor, which is implicated in coagulopathy observed in NHPs (Geisbert et al. 2003b), was also elevated in patients with severe EVD (McElroy et al. 2016). Von Willebrand factor (vWF), a protein that is present in both platelets and endothelial cells and mediates interactions between platelets and the damaged endothelium, was elevated both in SUDV-infected patients with hemorrhage and in pediatric SUDV-infected patients with fatal outcomes. It was also elevated in EBOV-infected patients with severe disease (McElroy et al. 2015b, 2016).
A complex interplay of activated endothelial cells, activated platelets, inflammation, and coagulopathy is clearly at work during EVD. How intervening in any one aspect of the network impacts human disease is still unknown. It would be invaluable to determine the effects on EVD outcome of readily available clinical products that affect aspects of these processes. Some compounds of interest are statins, which stabilize the endothelium; soluble GPIbα, which inhibits the interaction between platelets and vWF; and sCD40L, which appears to be consumed during severe disease.
9 The Roles of Co-infections, Co-morbidities, and Age
One key and largely unaddressed question is the role of co-infections and co-morbidities in EVD pathogenesis. Especially relevant to patient populations in the affected African nations are the possible contributions of malnutrition and malarial co-infection in the disease process. Malnutrition is prevalent in the regions affected by EVD (Wirth et al. 2016), and long-standing malnutrition leads to defects in both innate and adaptive cellular immune responses (Schaible and Kaufmann 2007). Malnutrition may contribute to the high case fatality rates observed during EVD outbreaks in Africa as compared to filoviral infections in patients repatriated to the US and Europe (Uyeki et al. 2016b; Martini 1973).
Malaria co-infection is likely to increase EVD-related mortality, although this has not been rigorously evaluated, a study of the effects of various antimalarial drugs has been conducted during the West African outbreak. Antimalarial drugs were routinely given to EVD-positive patients at the ETC in Foya, Liberia; during a time of artesunate-lumefantrine shortage, artesunate-amodiaquine was prescribed. While the amodiaquine preparation was associated with improved survival in malaria-negative patients, interestingly suggesting a direct antiviral effect, this effect was lost in the malaria-positive patients, suggesting that malaria and EVD co-infection lead to worse outcomes even when malaria is treated (Gignoux et al. 2016).
Also of potential consequence are co-infections with HIV or other hepatotropic viruses. HIV co-infection has only been examined in one study; during the SUDV outbreak in Gulu, 18% of the tested patients were HIV-1-positive by antibody testing. No differences in EVD outcome were observed based upon HIV status in this study (McElroy et al. 2014a, b), but no CD4 counts were obtained, so it is possible that all cases were newly acquired and the patients were not yet immune-compromised enough for HIV-1 infection to influence EVD outcome. One study evaluated publicly available next-generation sequencing data, and using a cohort of 49 patients, posited that co-infection with GB virus C (a common, clinically innocuous pegivirus infection) results in improved outcomes during EVD infection (Lauck et al. 2015). The results were somewhat confounded by the fact that age is a major determinant of both GB virus C infection and outcome during EVD.
A special mention must be made that early data regarding the effect of age on EBOV susceptibility (Dowell 1996) and outcome (Mupere et al. 2001; McElroy et al. 2014a, b) have been repeatedly observed in large cohorts during the West African outbreak (Team et al. 2015; Faye et al. 2015; Li et al. 2016; Schieffelin et al. 2014; Bower et al. 2016). Case fatality rates are high in children under 5, lowest in school-aged children, reaching a nadir around puberty, and increase again to peak in the elderly. This phenomenon has been seen in other infectious diseases in children, and suggests perhaps that school-aged children are in the perfect immunologic window of life, with a fully mature and functioning immune system without the alterations that occur secondary to the influence of sex hormones. To date, only one study has examined pediatric patients for laboratory evidence of this protective effect in EVD; this study demonstrated that pediatric patients have viral loads similar to adult patients (McElroy et al. 2014a, b) and thus do not appear to control the viremia better. However, higher levels of RANTES, a T cell chemokine, were associated with pediatric survival, an association not seen in adults. Thus, stronger immune responses in pediatric patients might contribute to better outcomes, but this remains to be proven definitively and will require additional research efforts.
10 Post-EVD Syndrome
Perhaps one of the most striking findings during the Western African EVD outbreak has been the identification of severe sequelae in EVD survivors long after recovery. These sequelae have important implications both for medical treatment and for public health. In 1975, one year before the identification of EBOV in Zaire, MARV was successfully isolated from the ocular fluid of a convalescent patient with uveitis (Gear et al. 1975). A later follow-up study described arthralgia, myalgia, and abdominal pain as common sequelae in EVD survivors of the Kikwit outbreak (Rowe et al. 1999). Similar findings in individuals long after recovery from BDBV infection (Clark et al. 2015) suggest that post-recovery sequelae may be common in filovirus infections.
To date, EBOV RNA has been detected in semen, ocular fluid, cerebrospinal fluid, breast milk, and other body fluids in EVD survivors for several weeks or even months after discharge (Green et al. 2016; Chughtai et al. 2016). Moreover, infectious virus has been isolated from semen (Uyeki et al. 2016a), ocular fluid (Varkey et al. 2015), saliva, and breast milk (Bausch et al. 2007), and epidemiological evidence of sexual EBOV transmission has been established (Mate et al. 2015). An important task will be determining the pathogenic potential of virus isolates from semen compared to those of blood from the same patient. Because sexual transmission seems to be uncommon (based on the large numbers of male survivors and few sexual transmission events), virus isolates from semen may be less infectious, either due to attenuating mutations or inactivation by EBOV-specific antibodies secreted at the mucosal surface. In general, the pathogenic features of EVD sequelae and their putative physiological mechanisms are poorly understood.
Importantly, many of the symptoms reported by EVD survivors, as well as some of the observed signs like uveitis and skin desquamation, suggest an inflammatory syndrome. Indeed, immune activation persists after the acute phase of EVD (McElroy et al. 2015a; Rowe et al. 1999), strongly suggesting continuous immune stimulation and postinfection autoimmunity. These hypotheses still need to be experimentally tested, but are consistent with virus persistence in immunoprivileged sites. Alternatively, sustained inflammation could be due to deposition of immune complexes in the joints or to viral antigen persistence, as seen commonly in alphavirus infections and influenza A virus infection respectively (Hoarau et al. 2010; Tamburini et al. 2014; Tappe et al. 2016).
Since some, but not all, EVD survivors suffer sequelae, the factors leading to post-EVD syndrome must be determined. Co-morbidities and co-infections are likely contributing to sequelae development, particularly those involving immune phenomena like bystander T cell activation (Fujinami et al. 2006). Also, a positive correlation has been described between viremia levels during the acute phase of EVD and increased risk of sequelae (Mattia et al. 2016). These findings suggest that during infection, EBOV may be confined to immunoprivileged sites by treatment or by the host immune response, leading to viral persistence, and that this phenomenon would be favored by high viral loads. Consistent with this hypothesis, one surviving patient with very high virus loads had virus persistence even within the central nervous system, followed by virus reactivation and meningoencephalitis (Jacobs et al. 2016).
In the future, the causes and molecular mechanisms of post-EVD syndrome must be determined. In particular, assessing whether reactivation of infectious virus is involved in sequelae development is a key. Due to the magnitude of the West African outbreak and the high number of surviving individuals, this is an important issue that needs to be addressed to establish adequate medical countermeasures and public health policies.
References
Adjemian J, Farnon EC, Tschioko F, Wamala JF, Byaruhanga E, Bwire GS, Kansiime E, Kagirita A, Ahimbisibwe S, Katunguka F, Jeffs B, Lutwama JJ, Downing R, Tappero JW, Formenty P, Amman B, Manning C, Towner J, Nichol ST, Rollin PE (2011) Outbreak of Marburg hemorrhagic fever among miners in Kamwenge and Ibanda Districts, Uganda, 2007. J Infect Dis 204(Suppl 3):S796–799. doi:10.1093/infdis/jir312
Agnandji ST, Huttner A, Zinser ME, Njuguna P, Dahlke C, Fernandes JF, Yerly S, Dayer JA, Kraehling V, Kasonta R, Adegnika AA, Altfeld M, Auderset F, Bache EB, Biedenkopf N, Borregaard S, Brosnahan JS, Burrow R, Combescure C, Desmeules J, Eickmann M, Fehling SK, Finckh A, Goncalves AR, Grobusch MP, Hooper J, Jambrecina A, Kabwende AL, Kaya G, Kimani D, Lell B, Lemaitre B, Lohse AW, Massinga-Loembe M, Matthey A, Mordmuller B, Nolting A, Ogwang C, Ramharter M, Schmidt-Chanasit J, Schmiedel S, Silvera P, Stahl FR, Staines HM, Strecker T, Stubbe HC, Tsofa B, Zaki S, Fast P, Moorthy V, Kaiser L, Krishna S, Becker S, Kieny MP, Bejon P, Kremsner PG, Addo MM, Siegrist CA (2016) Phase 1 Trials of rVSV Ebola Vaccine in Africa and Europe. N Engl J Med 374(17):1647–1660. doi:10.1056/NEJMoa1502924
Alvarez CP, Lasala F, Carrillo J, Muniz O, Corbi AL, Delgado R (2002) C-type lectins DC-SIGN and L-SIGN mediate cellular entry by Ebola virus in cis and in trans. J Virol 76(13):6841–6844
Amman BR, Carroll SA, Reed ZD, Sealy TK, Balinandi S, Swanepoel R, Kemp A, Erickson BR, Comer JA, Campbell S, Cannon DL, Khristova ML, Atimnedi P, Paddock CD, Crockett RJ, Flietstra TD, Warfield KL, Unfer R, Katongole-Mbidde E, Downing R, Tappero JW, Zaki SR, Rollin PE, Ksiazek TG, Nichol ST, Towner JS (2012) Seasonal pulses of Marburg virus circulation in juvenile Rousettus aegyptiacus bats coincide with periods of increased risk of human infection. PLoS Pathog 8(10):e1002877. doi:10.1371/journal.ppat.1002877
Appay V, Dunbar PR, Callan M, Klenerman P, Gillespie GM, Papagno L, Ogg GS, King A, Lechner F, Spina CA, Little S, Havlir DV, Richman DD, Gruener N, Pape G, Waters A, Easterbrook P, Salio M, Cerundolo V, McMichael AJ, Rowland-Jones SL (2002) Memory CD8+ T cells vary in differentiation phenotype in different persistent virus infections. Nat Med 8(4):379–385. doi:10.1038/nm0402-379
Auffray C, Fogg D, Garfa M, Elain G, Join-Lambert O, Kayal S, Sarnacki S, Cumano A, Lauvau G, Geissmann F (2007) Monitoring of blood vessels and tissues by a population of monocytes with patrolling behavior. Science 317(5838):666–670. doi:10.1126/science.1142883
Bah EI, Lamah MC, Fletcher T, Jacob ST, Brett-Major DM, Sall AA, Shindo N, Fischer WA 2nd, Lamontagne F, Saliou SM, Bausch DG, Moumie B, Jagatic T, Sprecher A, Lawler JV, Mayet T, Jacquerioz FA, Mendez Baggi MF, Vallenas C, Clement C, Mardel S, Faye O, Faye O, Soropogui B, Magassouba N, Koivogui L, Pinto R, Fowler RA (2015) Clinical presentation of patients with Ebola virus disease in Conakry, Guinea. N Engl J Med 372(1):40–47. doi:10.1056/NEJMoa1411249
Baize S, Leroy EM, Georges AJ, Georges-Courbot MC, Capron M, Bedjabaga I, Lansoud-Soukate J, Mavoungou E (2002) Inflammatory responses in Ebola virus-infected patients. Clin Exp Immunol 128(1):163–168
Baize S, Leroy EM, Georges-Courbot MC, Capron M, Lansoud-Soukate J, Debre P, Fisher-Hoch SP, McCormick JB, Georges AJ (1999) Defective humoral responses and extensive intravascular apoptosis are associated with fatal outcome in Ebola virus-infected patients. Nat Med 5(4):423–426. doi:10.1038/7422
Banchereau J, Steinman RM (1998) Dendritic cells and the control of immunity. Nature 392(6673):245–252. doi:10.1038/32588
Barry M, Traore FA, Sako FB, Kpamy DO, Bah EI, Poncin M, Keita S, Cisse M, Toure A (2014) Ebola outbreak in Conakry, Guinea: epidemiological, clinical, and outcome features. Med Mal Infect 44(11–12):491–494. doi:10.1016/j.medmal.2014.09.009
Bausch DG, Borchert M, Grein T, Roth C, Swanepoel R, Libande ML, Talarmin A, Bertherat E, Muyembe-Tamfum JJ, Tugume B, Colebunders R, Konde KM, Pirad P, Olinda LL, Rodier GR, Campbell P, Tomori O, Ksiazek TG, Rollin PE (2003) Risk factors for Marburg hemorrhagic fever, Democratic Republic of the Congo. Emerg Infect Dis 9(12):1531–1537. doi:10.3201/eid0912.030355
Bausch DG, Towner JS, Dowell SF, Kaducu F, Lukwiya M, Sanchez A, Nichol ST, Ksiazek TG, Rollin PE (2007) Assessment of the risk of Ebola virus transmission from bodily fluids and fomites. J Infect Dis 196(Suppl 2):S142–147. doi:10.1086/520545
Bornholdt ZA, Ndungo E, Fusco ML, Bale S, Flyak AI, Crowe JE Jr, Chandran K, Saphire EO (2016a) Host-primed Ebola Virus GP exposes a hydrophobic NPC1 receptor-binding pocket, revealing a target for broadly neutralizing antibodies. mBio 7(1):e02154–02115. doi:10.1128/mBio.02154-15
Bornholdt ZA, Turner HL, Murin CD, Li W, Sok D, Souders CA, Piper AE, Goff A, Shamblin JD, Wollen SE, Sprague TR, Fusco ML, Pommert KB, Cavacini LA, Smith HL, Klempner M, Reimann KA, Krauland E, Gerngross TU, Wittrup KD, Saphire EO, Burton DR, Glass PJ, Ward AB, Walker LM (2016b) Isolation of potent neutralizing antibodies from a survivor of the 2014 Ebola virus outbreak. Science 351(6277):1078–1083. doi:10.1126/science.aad5788
Bosio CM, Aman MJ, Grogan C, Hogan R, Ruthel G, Negley D, Mohamadzadeh M, Bavari S, Schmaljohn A (2003) Ebola and Marburg viruses replicate in monocyte-derived dendritic cells without inducing the production of cytokines and full maturation. J Infect Dis 188(11):1630–1638. doi:10.1086/379199
Bower H, Johnson S, Bangura MS, Kamara AJ, Kamara O, Mansaray SH, Sesay D, Turay C, Checchi F, Glynn JR (2016) Exposure-specific and age-specific attack rates for Ebola Virus disease in Ebola-affected households, Sierra Leone. Emerg Infect Dis 22(8). doi:10.3201/eid2208.160163
Brannan JM, Froude JW, Prugar LI, Bakken RR, Zak SE, Daye SP, Wilhelmsen CE, Dye JM (2015) Interferon alpha/beta receptor-deficient mice as a model for Ebola Virus disease. J Infect Dis 212(Suppl 2):S282–294. doi:10.1093/infdis/jiv215
Bray M, Raymond JL, Geisbert T, Baker RO (2002) 3-deazaneplanocin A induces massively increased interferon-alpha production in Ebola virus-infected mice. Antiviral Res 55(1):151–159
Buchbinder EI, Desai A (2016) CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am J Clin Oncol 39(1):98–106. doi:10.1097/COC.0000000000000239
Bwaka MA, Bonnet MJ, Calain P, Colebunders R, De Roo A, Guimard Y, Katwiki KR, Kibadi K, Kipasa MA, Kuvula KJ, Mapanda BB, Massamba M, Mupapa KD, Muyembe-Tamfum JJ, Ndaberey E, Peters CJ, Rollin PE, Van den Enden E, Van den Enden E (1999) Ebola hemorrhagic fever in Kikwit, Democratic Republic of the Congo: clinical observations in 103 patients. J Infect Dis 179(Suppl 1):S1–7. doi:10.1086/514308
Carrion R Jr, Brasky K, Mansfield K, Johnson C, Gonzales M, Ticer A, Lukashevich I, Tardif S, Patterson J (2007) Lassa virus infection in experimentally infected marmosets: liver pathology and immunophenotypic alterations in target tissues. J Virol 81(12):6482–6490. doi:10.1128/JVI.02876-06
Centers for Disease C, Prevention (2009) Imported case of Marburg hemorrhagic fever—Colorado, 2008. MMWR Morb Mortal Wkly Rep 58(49):1377–1381
Chertow DS, Kleine C, Edwards JK, Scaini R, Giuliani R, Sprecher A (2014) Ebola virus disease in West Africa–clinical manifestations and management. N Engl J Med 371(22):2054–2057. doi:10.1056/NEJMp1413084
Chughtai AA, Barnes M, Macintyre CR (2016) Persistence of Ebola virus in various body fluids during convalescence: evidence and implications for disease transmission and control. Epidemiol Infect 144(8):1652–1660. doi:10.1017/S0950268816000054
Clark DV, Kibuuka H, Millard M, Wakabi S, Lukwago L, Taylor A, Eller MA, Eller LA, Michael NL, Honko AN, Olinger GG Jr, Schoepp RJ, Hepburn MJ, Hensley LE, Robb ML (2015) Long-term sequelae after Ebola virus disease in Bundibugyo, Uganda: a retrospective cohort study. Lancet Infect Dis 15(8):905–912. doi:10.1016/S1473-3099(15)70152-0
Colebunders R, Tshomba A, Van Kerkhove MD, Bausch DG, Campbell P, Libande M, Pirard P, Tshioko F, Mardel S, Mulangu S, Sleurs H, Rollin PE, Muyembe-Tamfum JJ, Jeffs B, Borchert M, International S, Technical Committee DRCWDMOIG (2007) Marburg hemorrhagic fever in Durba and Watsa, Democratic Republic of the Congo: clinical documentation, features of illness, and treatment. J Infect Dis 196(Suppl 2):S148–153. doi:10.1086/520543
Corti D, Misasi J, Mulangu S, Stanley DA, Kanekiyo M, Wollen S, Ploquin A, Doria-Rose NA, Staupe RP, Bailey M, Shi W, Choe M, Marcus H, Thompson EA, Cagigi A, Silacci C, Fernandez-Rodriguez B, Perez L, Sallusto F, Vanzetta F, Agatic G, Cameroni E, Kisalu N, Gordon I, Ledgerwood JE, Mascola JR, Graham BS, Muyembe-Tamfun JJ, Trefry JC, Lanzavecchia A, Sullivan NJ (2016) Protective monotherapy against lethal Ebola virus infection by a potently neutralizing antibody. Science 351(6279):1339–1342. doi:10.1126/science.aad5224
Cros J, Cagnard N, Woollard K, Patey N, Zhang SY, Senechal B, Puel A, Biswas SK, Moshous D, Picard C, Jais JP, D’Cruz D, Casanova JL, Trouillet C, Geissmann F (2010) Human CD14dim monocytes patrol and sense nucleic acids and viruses via TLR7 and TLR8 receptors. Immunity 33(3):375–386. doi:10.1016/j.immuni.2010.08.012
Cusi MG, Gandolfo C, Terrosi C, Gori Savellini G, Belmonte G, Miracco C (2016) Toscana virus infects dendritic and endothelial cells opening the way for the central nervous system. J Neurovirol 22(3):307–315. doi:10.1007/s13365-015-0395-2
Dallatomasina S, Crestani R, Sylvester Squire J, Declerk H, Caleo GM, Wolz A, Stinson K, Patten G, Brechard R, Gbabai OB, Spreicher A, Van Herp M, Zachariah R (2015) Ebola outbreak in rural West Africa: epidemiology, clinical features and outcomes. Trop Med Int Health TM & IH 20(4):448–454. doi:10.1111/tmi.12454
Deen GF, Knust B, Broutet N, Sesay FR, Formenty P, Ross C, Thorson AE, Massaquoi TA, Marrinan JE, Ervin E, Jambai A, McDonald SL, Bernstein K, Wurie AH, Dumbuya MS, Abad N, Idriss B, Wi T, Bennett SD, Davies T, Ebrahim FK, Meites E, Naidoo D, Smith S, Banerjee A, Erickson BR, Brault A, Durski KN, Winter J, Sealy T, Nichol ST, Lamunu M, Stroher U, Morgan O, Sahr F (2015) Ebola RNA Persistence in Semen of Ebola Virus disease survivors—preliminary report. N Engl J Med. doi:10.1056/NEJMoa1511410
Dowell SF (1996) Ebola hemorrhagic fever: why were children spared? Pediatr Infect Dis J 15(3):189–191
Dowell SF, Mukunu R, Ksiazek TG, Khan AS, Rollin PE, Peters CJ (1999) Transmission of Ebola hemorrhagic fever: a study of risk factors in family members, Kikwit, Democratic Republic of the Congo, 1995. Commission de Lutte contre les Epidemies a Kikwit. J Infect Dis 179(Suppl 1):S87–91. doi:10.1086/514284
Ebihara H, Rockx B, Marzi A, Feldmann F, Haddock E, Brining D, LaCasse RA, Gardner D, Feldmann H (2011) Host response dynamics following lethal infection of rhesus macaques with Zaire ebolavirus. J Infect Dis 204(Suppl 3):S991–999. doi:10.1093/infdis/jir336
Ebola haemorrhagic fever in Zaire. (1976/1978). Bulletin of the World Health Organization 56 (2):271–293
Emond RT, Evans B, Bowen ET, Lloyd G (1977) A case of Ebola virus infection. Br Med J 2(6086):541–544
Ersland K, Wuthrich M, Klein BS (2010) Dynamic interplay among monocyte-derived, dermal, and resident lymph node dendritic cells during the generation of vaccine immunity to fungi. Cell Host Microbe 7(6):474–487. doi:10.1016/j.chom.2010.05.010
Escudero-Perez B, Volchkova VA, Dolnik O, Lawrence P, Volchkov VE (2014) Shed GP of Ebola virus triggers immune activation and increased vascular permeability. PLoS Pathog 10(11):e1004509. doi:10.1371/journal.ppat.1004509
Ewer K, Rampling T, Venkatraman N, Bowyer G, Wright D, Lambe T, Imoukhuede EB, Payne R, Fehling SK, Strecker T, Biedenkopf N, Krahling V, Tully CM, Edwards NJ, Bentley EM, Samuel D, Labbe G, Jin J, Gibani M, Minhinnick A, Wilkie M, Poulton I, Lella N, Roberts R, Hartnell F, Bliss C, Sierra-Davidson K, Powlson J, Berrie E, Tedder R, Roman F, De Ryck I, Nicosia A, Sullivan NJ, Stanley DA, Mbaya OT, Ledgerwood JE, Schwartz RM, Siani L, Colloca S, Folgori A, Di Marco S, Cortese R, Wright E, Becker S, Graham BS, Koup RA, Levine MM, Volkmann A, Chaplin P, Pollard AJ, Draper SJ, Ballou WR, Lawrie A, Gilbert SC, Hill AV (2016) A monovalent Chimpanzee Adenovirus Ebola Vaccine boosted with MVA. N Engl J Med 374(17):1635–1646. doi:10.1056/NEJMoa1411627
Faye O, Andronico A, Faye O, Salje H, Boelle PY, Magassouba N, Bah EI, Koivogui L, Diallo B, Diallo AA, Keita S, Konde MK, Fowler R, Fall G, Cauchemez S, Sall AA (2015) Use of Viremia to evaluate the baseline case fatality ratio of Ebola Virus disease and inform treatment studies: a retrospective cohort study. PLoS Med 12(12):e1001908. doi:10.1371/journal.pmed.1001908
Feldmann H, Bugany H, Mahner F, Klenk HD, Drenckhahn D, Schnittler HJ (1996) Filovirus-induced endothelial leakage triggered by infected monocytes/macrophages. J Virol 70(4):2208–2214
Feldmann H, Geisbert TW (2011) Ebola haemorrhagic fever. Lancet 377(9768):849–862. doi:10.1016/S0140-6736(10)60667-8
Fischer RJ, Judson S, Miazgowicz K, Bushmaker T, Munster VJ (2016) Ebola Virus persistence in Semen Ex Vivo. Emerg Infect Dis 22(2):289–291. doi:10.3201/eid2202.151278
Fisher-Hoch SP, Platt GS, Neild GH, Southee T, Baskerville A, Raymond RT, Lloyd G, Simpson DI (1985) Pathophysiology of shock and hemorrhage in a fulminating viral infection (Ebola). J Infect Dis 152(5):887–894
Francesconi P, Yoti Z, Declich S, Onek PA, Fabiani M, Olango J, Andraghetti R, Rollin PE, Opira C, Greco D, Salmaso S (2003) Ebola hemorrhagic fever transmission and risk factors of contacts, Uganda. Emerg Infect Dis 9(11):1430–1437. doi:10.3201/eid0911.030339
Fujinami RS, von Herrath MG, Christen U, Whitton JL (2006) Molecular mimicry, bystander activation, or viral persistence: infections and autoimmune disease. Clin Microbiol Rev 19(1):80–94. doi:10.1128/CMR.19.1.80-94.2006
Gear JS, Cassel GA, Gear AJ, Trappler B, Clausen L, Meyers AM, Kew MC, Bothwell TH, Sher R, Miller GB, Schneider J, Koornhof HJ, Gomperts ED, Isaacson M, Gear JH (1975) Outbreake of Marburg virus disease in Johannesburg. Br Med J 4(5995):489–493
Geisbert TW, Hensley LE, Larsen T, Young HA, Reed DS, Geisbert JB, Scott DP, Kagan E, Jahrling PB, Davis KJ (2003a) Pathogenesis of Ebola hemorrhagic fever in cynomolgus macaques: evidence that dendritic cells are early and sustained targets of infection. Am J Pathol 163(6):2347–2370. doi:10.1016/S0002-9440(10)63591-2
Geisbert TW, Young HA, Jahrling PB, Davis KJ, Kagan E, Hensley LE (2003b) Mechanisms underlying coagulation abnormalities in ebola hemorrhagic fever: overexpression of tissue factor in primate monocytes/macrophages is a key event. J Infect Dis 188(11):1618–1629. doi:10.1086/379724
Geisbert TW, Young HA, Jahrling PB, Davis KJ, Larsen T, Kagan E, Hensley LE (2003c) Pathogenesis of Ebola hemorrhagic fever in primate models: evidence that hemorrhage is not a direct effect of virus-induced cytolysis of endothelial cells. Am J Pathol 163(6):2371–2382. doi:10.1016/S0002-9440(10)63592-4
Gignoux E, Azman AS, de Smet M, Azuma P, Massaquoi M, Job D, Tiffany A, Petrucci R, Sterk E, Potet J, Suzuki M, Kurth A, Cannas A, Bocquin A, Strecker T, Logue C, Pottage T, Yue C, Cabrol JC, Serafini M, Ciglenecki I (2016) Effect of Artesunate-Amodiaquine on mortality related to Ebola Virus disease. N Engl J Med 374(1):23–32. doi:10.1056/NEJMoa1504605
Green E, Hunt L, Ross JC, Nissen NM, Curran T, Badhan A, Sutherland KA, Richards J, Lee JS, Allen SH, Laird S, Blackman M, Collacott I, Parker PA, Walbridge A, Phillips R, Sellu SJ, Dama A, Sheriff AK, Zombo J, Ngegba D, Wurie AH, Checchi F, Brooks TJ (2016) Viraemia and Ebola virus secretion in survivors of Ebola virus disease in Sierra Leone: a cross-sectional cohort study. Lancet Infect Dis. doi:10.1016/S1473-3099(16)30060-3
Gupta M, MacNeil A, Reed ZD, Rollin PE, Spiropoulou CF (2012) Serology and cytokine profiles in patients infected with the newly discovered Bundibugyo ebolavirus. Virology 423(2):119–124. doi:10.1016/j.virol.2011.11.027
Gupta M, Mahanty S, Ahmed R, Rollin PE (2001) Monocyte-derived human macrophages and peripheral blood mononuclear cells infected with ebola virus secrete MIP-1alpha and TNF-alpha and inhibit poly-IC-induced IFN-alpha in vitro. Virology 284(1):20–25. doi:10.1006/viro.2001.0836
Gupta M, Spiropoulou C, Rollin PE (2007) Ebola virus infection of human PBMCs causes massive death of macrophages, CD4 and CD8 T cell sub-populations in vitro. Virology 364(1):45–54. doi:10.1016/j.virol.2007.02.017
Haniffa M, Collin M, Ginhoux F (2013) Ontogeny and functional specialization of dendritic cells in human and mouse. Adv Immunol 120:1–49. doi:10.1016/B978-0-12-417028-5.00001-6
Henn V, Steinbach S, Buchner K, Presek P, Kroczek RA (2001) The inflammatory action of CD40 ligand (CD154) expressed on activated human platelets is temporally limited by coexpressed CD40. Blood 98(4):1047–1054
Hoarau JJ, Jaffar Bandjee MC, Krejbich Trotot P, Das T, Li-Pat-Yuen G, Dassa B, Denizot M, Guichard E, Ribera A, Henni T, Tallet F, Moiton MP, Gauzere BA, Bruniquet S, Jaffar Bandjee Z, Morbidelli P, Martigny G, Jolivet M, Gay F, Grandadam M, Tolou H, Vieillard V, Debre P, Autran B, Gasque P (2010) Persistent chronic inflammation and infection by Chikungunya arthritogenic alphavirus in spite of a robust host immune response. J Immunol 184(10):5914–5927. doi:10.4049/jimmunol.0900255
Huang YW, Dryman BA, Li W, Meng XJ (2009) Porcine DC-SIGN: molecular cloning, gene structure, tissue distribution and binding characteristics. Dev Comp Immunol 33(4):464–480. doi:10.1016/j.dci.2008.09.010
Hunt BJ (2014) Bleeding and coagulopathies in critical care. N Engl J Med 370(22):2153. doi:10.1056/NEJMc1403768
Hunt L, Gupta-Wright A, Simms V, Tamba F, Knott V, Tamba K, Heisenberg-Mansaray S, Tamba E, Sheriff A, Conteh S, Smith T, Tobin S, Brooks T, Houlihan C, Cummings R, Fletcher T (2015) Clinical presentation, biochemical, and haematological parameters and their association with outcome in patients with Ebola virus disease: an observational cohort study. Lancet Infect Dis 15(11):1292–1299. doi:10.1016/S1473-3099(15)00144-9
Hutchinson KL, Rollin PE (2007) Cytokine and chemokine expression in humans infected with Sudan Ebola virus. J Infect Dis 196(Suppl 2):S357–363. doi:10.1086/520611
Ilinykh PA, Lubaki NM, Widen SG, Renn LA, Theisen TC, Rabin RL, Wood TG, Bukreyev A (2015) Different temporal effects of Ebola Virus VP35 and VP24 proteins on global gene expression in human dendritic cells. J Virol 89(15):7567–7583. doi:10.1128/JVI.00924-15
Jacobs M, Rodger A, Bell DJ, Bhagani S, Cropley I, Filipe A, Gifford RJ, Hopkins S, Hughes J, Jabeen F, Johannessen I, Karageorgopoulos D, Lackenby A, Lester R, Liu RS, MacConnachie A, Mahungu T, Martin D, Marshall N, Mepham S, Orton R, Palmarini M, Patel M, Perry C, Peters SE, Porter D, Ritchie D, Ritchie ND, Seaton RA, Sreenu VB, Templeton K, Warren S, Wilkie GS, Zambon M, Gopal R, Thomson EC (2016) Late Ebola virus relapse causing meningoencephalitis: a case report. Lancet. doi:10.1016/S0140-6736(16)30386-5
Jin H, Yan Z, Prabhakar BS, Feng Z, Ma Y, Verpooten D, Ganesh B, He B (2010) The VP35 protein of Ebola virus impairs dendritic cell maturation induced by virus and lipopolysaccharide. J Gen Virol 91(Pt 2):352–361. doi:10.1099/vir.0.017343-0
Klechevsky E, Banchereau J (2013) Human dendritic cells subsets as targets and vectors for therapy. Ann N Y Acad Sci 1284:24–30. doi:10.1111/nyas.12113
Kondratowicz AS, Lennemann NJ, Sinn PL, Davey RA, Hunt CL, Moller-Tank S, Meyerholz DK, Rennert P, Mullins RF, Brindley M, Sandersfeld LM, Quinn K, Weller M, McCray PB Jr, Chiorini J, Maury W (2011) T-cell immunoglobulin and mucin domain 1 (TIM-1) is a receptor for Zaire Ebolavirus and Lake Victoria Marburgvirus. Proc Natl Acad Sci U S A 108(20):8426–8431. doi:10.1073/pnas.1019030108
Kornbluth RS, Bot A (2012) In this issue: harnessing immunity against difficult targets, through the CD40-CD40L pathway. Int Rev Immunol 31(4):243–245. doi:10.3109/08830185.2012.702511
Kreuels B, Wichmann D, Emmerich P, Schmidt-Chanasit J, de Heer G, Kluge S, Sow A, Renne T, Gunther S, Lohse AW, Addo MM, Schmiedel S (2014) A case of severe Ebola virus infection complicated by gram-negative septicemia. N Engl J Med 371(25):2394–2401. doi:10.1056/NEJMoa1411677
Ksiazek TG, Rollin PE, Williams AJ, Bressler DS, Martin ML, Swanepoel R, Burt FJ, Leman PA, Khan AS, Rowe AK, Mukunu R, Sanchez A, Peters CJ (1999) Clinical virology of Ebola hemorrhagic fever (EHF): virus, virus antigen, and IgG and IgM antibody findings among EHF patients in Kikwit, Democratic Republic of the Congo, 1995. J Infect Dis 179(Suppl 1):S177–187. doi:10.1086/514321
Kvale EO, Dalgaard J, Lund-Johansen F, Rollag H, Farkas L, Midtvedt K, Jahnsen FL, Brinchmann JE, Olweus J (2006) CD11c+ dendritic cells and plasmacytoid DCs are activated by human cytomegalovirus and retain efficient T cell-stimulatory capability upon infection. Blood 107(5):2022–2029. doi:10.1182/blood-2005-05-2016
Lado M, Walker NF, Baker P, Haroon S, Brown CS, Youkee D, Studd N, Kessete Q, Maini R, Boyles T, Hanciles E, Wurie A, Kamara TB, Johnson O, Leather AJ (2015) Clinical features of patients isolated for suspected Ebola virus disease at Connaught Hospital, Freetown, Sierra Leone: a retrospective cohort study. Lancet Infect Dis 15(9):1024–1033. doi:10.1016/S1473-3099(15)00137-1
Langdown J, Luddington RJ, Huntington JA, Baglin TP (2014) A hereditary bleeding disorder resulting from a premature stop codon in thrombomodulin (p.Cys537Stop). Blood 124(12):1951–1956. doi:10.1182/blood-2014-02-557538
Lanini S, Portella G, Vairo F, Kobinger GP, Pesenti A, Langer M, Kabia S, Brogiato G, Amone J, Castilletti C, Miccio R, Zumla A, Capobianchi MR, Di Caro A, Strada G, Ippolito G, Group I-EESLS (2015) Blood kinetics of Ebola virus in survivors and nonsurvivors. J Clin Invest 125(12):4692–4698. doi:10.1172/JCI83111
Lauck M, Bailey AL, Andersen KG, Goldberg TL, Sabeti PC, O’Connor DH (2015) GB virus C coinfections in west African Ebola patients. J Virol 89(4):2425–2429. doi:10.1128/JVI.02752-14
Leon B, Lopez-Bravo M, Ardavin C (2007) Monocyte-derived dendritic cells formed at the infection site control the induction of protective T helper 1 responses against Leishmania. Immunity 26(4):519–531. doi:10.1016/j.immuni.2007.01.017
Leroy EM, Baize S, Debre P, Lansoud-Soukate J, Mavoungou E (2001) Early immune responses accompanying human asymptomatic Ebola infections. Clin Exp Immunol 124(3):453–460
Leroy EM, Baize S, Volchkov VE, Fisher-Hoch SP, Georges-Courbot MC, Lansoud-Soukate J, Capron M, Debre P, McCormick JB, Georges AJ (2000) Human asymptomatic Ebola infection and strong inflammatory response. Lancet 355(9222):2210–2215
Leroy EM, Epelboin A, Mondonge V, Pourrut X, Gonzalez JP, Muyembe-Tamfum JJ, Formenty P (2009) Human Ebola outbreak resulting from direct exposure to fruit bats in Luebo, Democratic Republic of Congo, 2007. Vector Borne Zoonotic Dis 9(6):723–728. doi:10.1089/vbz.2008.0167
Leung LW, Martinez O, Reynard O, Volchkov VE, Basler CF (2011) Ebola virus failure to stimulate plasmacytoid dendritic cell interferon responses correlates with impaired cellular entry. J Infect Dis 204(Suppl 3):S973–977. doi:10.1093/infdis/jir331
Levi M, Toh CH, Thachil J, Watson HG (2009) Guidelines for the diagnosis and management of disseminated intravascular coagulation. British Committee for standards in haematology. Br J Haematol 145(1):24–33. doi:10.1111/j.1365-2141.2009.07600.x
Li J, Duan HJ, Chen HY, Ji YJ, Zhang X, Rong YH, Xu Z, Sun LJ, Zhang JY, Liu LM, Jin B, Zhang J, Du N, Su HB, Teng GJ, Yuan Y, Qin EQ, Jia HJ, Wang S, Guo TS, Wang Y, Mu JS, Yan T, Li ZW, Dong Z, Nie WM, Jiang TJ, Li C, Gao XD, Ji D, Zhuang YJ, Li L, Wang LF, Li WG, Duan XZ, Lu YY, Sun ZQ, Kanu AB, Koroma SM, Zhao M, Ji JS, Wang FS (2016) Age and Ebola viral load correlate with mortality and survival time in 288 Ebola virus disease patients. Int J Infect Dis IJID Official Publ Int Soc Infect Dis 42:34–39. doi:10.1016/j.ijid.2015.10.021
Lindgren T, Ahlm C, Mohamed N, Evander M, Ljunggren HG, Bjorkstrom NK (2011) Longitudinal analysis of the human T cell response during acute hantavirus infection. J Virol 85(19):10252–10260. doi:10.1128/JVI.05548-11
Liu L, Wei Q, Nishiura K, Peng J, Wang H, Midkiff C, Alvarez X, Qin C, Lackner A, Chen Z (2015) Spatiotemporal interplay of severe acute respiratory syndrome coronavirus and respiratory mucosal cells drives viral dissemination in rhesus macaques. Mucosal Immunol. doi:10.1038/mi.2015.127
Luan YY, Yao YM, Xiao XZ, Sheng ZY (2015) Insights into the apoptotic death of immune cells in sepsis. J Interferon Cytokine Res Official J Int Soc Interferon Cytokine Res 35(1):17–22. doi:10.1089/jir.2014.0069
Luczkowiak J, Arribas JR, Gomez S, Jimenez-Yuste V, de la Calle F, Viejo A, Delgado R (2016) Specific neutralizing response in plasma from convalescent patients of Ebola Virus disease against the West Africa Makona variant of Ebola virus. Virus Res 213:224–229. doi:10.1016/j.virusres.2015.12.019
Ludtke A, Ruibal P, Becker-Ziaja B, Rottstegge M, Wozniak DM, Cabeza-Cabrerizo M, Thorenz A, Weller R, Kerber R, Idoyaga J, Magassouba N, Gabriel M, Gunther S, Oestereich L, Munoz-Fontela C (2016) Ebola Virus disease is characterized by Poor activation and reduced levels of circulating CD16+ Monocytes. J Infect Dis 214(suppl 3):S275–S280. doi:10.1093/infdis/jiw260
MacNeil A, Farnon EC, Wamala J, Okware S, Cannon DL, Reed Z, Towner JS, Tappero JW, Lutwama J, Downing R, Nichol ST, Ksiazek TG, Rollin PE (2010) Proportion of deaths and clinical features in Bundibugyo Ebola virus infection, Uganda. Emerg Infect Dis 16(12):1969–1972. doi:10.3201/eid1612.100627
Mahanty S, Hutchinson K, Agarwal S, McRae M, Rollin PE, Pulendran B (2003) Cutting edge: impairment of dendritic cells and adaptive immunity by Ebola and Lassa viruses. J Immunol 170(6):2797–2801
Mari Saez A, Weiss S, Nowak K, Lapeyre V, Zimmermann F, Dux A, Kuhl HS, Kaba M, Regnaut S, Merkel K, Sachse A, Thiesen U, Villanyi L, Boesch C, Dabrowski PW, Radonic A, Nitsche A, Leendertz SA, Petterson S, Becker S, Krahling V, Couacy-Hymann E, Akoua-Koffi C, Weber N, Schaade L, Fahr J, Borchert M, Gogarten JF, Calvignac-Spencer S, Leendertz FH (2015) Investigating the zoonotic origin of the West African Ebola epidemic. EMBO Mol Med 7(1):17–23. doi:10.15252/emmm.201404792
Martines RB, Ng DL, Greer PW, Rollin PE, Zaki SR (2015) Tissue and cellular tropism, pathology and pathogenesis of Ebola and Marburg viruses. J Pathol 235(2):153–174. doi:10.1002/path.4456
Martinez O, Johnson JC, Honko A, Yen B, Shabman RS, Hensley LE, Olinger GG, Basler CF (2013) Ebola virus exploits a monocyte differentiation program to promote its entry. J Virol 87(7):3801–3814. doi:10.1128/JVI.02695-12
Martini GA (1973) Marburg virus disease. Postgrad Med J 49(574):542–546
Mate SE, Kugelman JR, Nyenswah TG, Ladner JT, Wiley MR, Cordier-Lassalle T, Christie A, Schroth GP, Gross SM, Davies-Wayne GJ, Shinde SA, Murugan R, Sieh SB, Badio M, Fakoli L, Taweh F, de Wit E, van Doremalen N, Munster VJ, Pettitt J, Prieto K, Humrighouse BW, Stroher U, DiClaro JW, Hensley LE, Schoepp RJ, Safronetz D, Fair J, Kuhn JH, Blackley DJ, Laney AS, Williams DE, Lo T, Gasasira A, Nichol ST, Formenty P, Kateh FN, De Cock KM, Bolay F, Sanchez-Lockhart M, Palacios G (2015) Molecular evidence of sexual transmission of Ebola Virus. N Engl J Med 373(25):2448–2454. doi:10.1056/NEJMoa1509773
Mattia JG, Vandy MJ, Chang JC, Platt DE, Dierberg K, Bausch DG, Brooks T, Conteh S, Crozier I, Fowler RA, Kamara AP, Kang C, Mahadevan S, Mansaray Y, Marcell L, McKay G, O’Dempsey T, Parris V, Pinto R, Rangel A, Salam AP, Shantha J, Wolfman V, Yeh S, Chan AK, Mishra S (2016) Early clinical sequelae of Ebola virus disease in Sierra Leone: a cross-sectional study. Lancet Infect Dis 16(3):331–338. doi:10.1016/S1473-3099(15)00489-2
McElroy AK, Erickson BR, Flietstra TD, Rollin PE, Nichol ST, Towner JS, Spiropoulou CF (2014a) Biomarker correlates of survival in pediatric patients with Ebola virus disease. Emerging infectious diseases
McElroy AK, Erickson BR, Flietstra TD, Rollin PE, Nichol ST, Towner JS, Spiropoulou CF (2014b) Ebola hemorrhagic Fever: novel biomarker correlates of clinical outcome. J Infect Dis 210(4):558–566. doi:10.1093/infdis/jiu088
McElroy AK, Akondy RS, Davis CW, Ellebedy AH, Mehta AK, Kraft CS, Lyon GM, Ribner BS, Varkey J, Sidney J, Sette A, Campbell S, Stroher U, Damon I, Nichol ST, Spiropoulou CF, Ahmed R (2015a) Human Ebola virus infection results in substantial immune activation. Proc Natl Acad Sci U S A 112(15):4719–4724. doi:10.1073/pnas.1502619112
McElroy AK, Erickson BR, Flietstra TD, Rollin PE, Towner JS, Nichol ST, Spiropoulou CF (2015b) Von Willebrand factor is elevated in individuals infected with Sudan virus and is associated with adverse clinical outcomes. Viral Immunol 28(1):71–73. doi:10.1089/vim.2014.0072
McElroy AK, Harmon JR, Flietstra TD, Campbell S, Mehta AK, Kraft CS, Lyon MG, Varkey JB, Ribner BS, Kratochvil CJ, Iwen PC, Smith PW, Ahmed R, Nichol ST, Spiropoulou CF (2016) Kinetic analysis of biomarkers in a Cohort of US patients with Ebola Virus disease. Clin Infect Dis Official Publ Infect Dis Soc Am 63(4):460–467. doi:10.1093/cid/ciw334
McNab F, Mayer-Barber K, Sher A, Wack A, O’Garra A (2015) Type I interferons in infectious disease. Nat Rev Immunol 15(2):87–103. doi:10.1038/nri3787
Merad M, Ginhoux F, Collin M (2008) Origin, homeostasis and function of Langerhans cells and other langerin-expressing dendritic cells. Nat Rev Immunol 8(12):935–947. doi:10.1038/nri2455
Mesman AW, de Vries RD, McQuaid S, Duprex WP, de Swart RL, Geijtenbeek TB (2012) A prominent role for DC-SIGN+ dendritic cells in initiation and dissemination of measles virus infection in non-human primates. PLoS ONE 7(12):e49573. doi:10.1371/journal.pone.0049573
Miller JD, van der Most RG, Akondy RS, Glidewell JT, Albott S, Masopust D, Murali-Krishna K, Mahar PL, Edupuganti S, Lalor S, Germon S, Del Rio C, Mulligan MJ, Staprans SI, Altman JD, Feinberg MB, Ahmed R (2008) Human effector and memory CD8+ T cell responses to smallpox and yellow fever vaccines. Immunity 28(5):710–722. doi:10.1016/j.immuni.2008.02.020
Misasi J, Gilman MS, Kanekiyo M, Gui M, Cagigi A, Mulangu S, Corti D, Ledgerwood JE, Lanzavecchia A, Cunningham J, Muyembe-Tamfun JJ, Baxa U, Graham BS, Xiang Y, Sullivan NJ, McLellan JS (2016) Structural and molecular basis for Ebola virus neutralization by protective human antibodies. Science 351(6279):1343–1346. doi:10.1126/science.aad6117
Mohamadzadeh M, Chen L, Schmaljohn AL (2007) How Ebola and Marburg viruses battle the immune system. Nat Rev Immunol 7(7):556–567. doi:10.1038/nri2098
Mohamadzadeh M, Coberley SS, Olinger GG, Kalina WV, Ruthel G, Fuller CL, Swenson DL, Pratt WD, Kuhns DB, Schmaljohn AL (2006) Activation of triggering receptor expressed on myeloid cells-1 on human neutrophils by marburg and ebola viruses. J Virol 80(14):7235–7244. doi:10.1128/JVI.00543-06
Mupere E, Kaducu OF, Yoti Z (2001) Ebola haemorrhagic fever among hospitalised children and adolescents in northern Uganda: epidemiologic and clinical observations. Afr Health Sci 1(2):60–65
Murin CD, Fusco ML, Bornholdt ZA, Qiu X, Olinger GG, Zeitlin L, Kobinger GP, Ward AB, Saphire EO (2014) Structures of protective antibodies reveal sites of vulnerability on Ebola virus. Proc Natl Acad Sci U S A 111(48):17182–17187. doi:10.1073/pnas.1414164111
Okware SI, Omaswa FG, Zaramba S, Opio A, Lutwama JJ, Kamugisha J, Rwaguma EB, Kagwa P, Lamunu M (2002) An outbreak of Ebola in Uganda. Trop Med Int Health TM & IH 7(12):1068–1075
Olal D, Kuehne AI, Bale S, Halfmann P, Hashiguchi T, Fusco ML, Lee JE, King LB, Kawaoka Y, Dye JM Jr, Saphire EO (2012) Structure of an antibody in complex with its mucin domain linear epitope that is protective against Ebola virus. J Virol 86(5):2809–2816. doi:10.1128/JVI.05549-11
Olinger GG Jr, Pettitt J, Kim D, Working C, Bohorov O, Bratcher B, Hiatt E, Hume SD, Johnson AK, Morton J, Pauly M, Whaley KJ, Lear CM, Biggins JE, Scully C, Hensley L, Zeitlin L (2012) Delayed treatment of Ebola virus infection with plant-derived monoclonal antibodies provides protection in rhesus macaques. Proc Natl Acad Sci U S A 109(44):18030–18035. doi:10.1073/pnas.1213709109
Onyango CO, Opoka ML, Ksiazek TG, Formenty P, Ahmed A, Tukei PM, Sang RC, Ofula VO, Konongoi SL, Coldren RL, Grein T, Legros D, Bell M, De Cock KM, Bellini WJ, Towner JS, Nichol ST, Rollin PE (2007) Laboratory diagnosis of Ebola hemorrhagic fever during an outbreak in Yambio, Sudan, 2004. J Infect Dis 196(Suppl 2):S193–198. doi:10.1086/520609
Qin E, Bi J, Zhao M, Wang Y, Guo T, Yan T, Li Z, Sun J, Zhang J, Chen S, Wu Y, Li J, Zhong Y (2015) Clinical Features of Patients With Ebola Virus Disease in Sierra Leone. Clin Infect Dis Official Publ Infect Dis Soc Am 61(4):491–495. doi:10.1093/cid/civ319
Rhein BA, Brouillette RB, Schaack GA, Chiorini JA, Maury W (2016) Characterization of human and murine T-cell immunoglobulin mucin domain 4 (TIM-4) IgV domain residues critical for Ebola Virus Entry. J Virol 90(13):6097–6111. doi:10.1128/JVI.00100-16
Rivera JA, McGuire TC (2005) Equine infectious anemia virus-infected dendritic cells retain antigen presentation capability. Virology 335(2):145–154. doi:10.1016/j.virol.2005.02.013
Roddy P, Thomas SL, Jeffs B, Nascimento Folo P, Pablo Palma P, Moco Henrique B, Villa L, Damiao Machado FP, Bernal O, Jones SM, Strong JE, Feldmann H, Borchert M (2010) Factors associated with Marburg hemorrhagic fever: analysis of patient data from Uige, Angola. J Infect Dis 201(12):1909–1918. doi:10.1086/652748
Rodriguez LL, De Roo A, Guimard Y, Trappier SG, Sanchez A, Bressler D, Williams AJ, Rowe AK, Bertolli J, Khan AS, Ksiazek TG, Peters CJ, Nichol ST (1999) Persistence and genetic stability of Ebola virus during the outbreak in Kikwit, Democratic Republic of the Congo, 1995. J Infect Dis 179(Suppl 1):S170–176. doi:10.1086/514291
Rollin PE, Bausch DG, Sanchez A (2007) Blood chemistry measurements and D-Dimer levels associated with fatal and nonfatal outcomes in humans infected with Sudan Ebola virus. J Infect Dis 196(Suppl 2):S364–371. doi:10.1086/520613
Rowe AK, Bertolli J, Khan AS, Mukunu R, Muyembe-Tamfum JJ, Bressler D, Williams AJ, Peters CJ, Rodriguez L, Feldmann H, Nichol ST, Rollin PE, Ksiazek TG (1999) Clinical, virologic, and immunologic follow-up of convalescent Ebola hemorrhagic fever patients and their household contacts, Kikwit, Democratic Republic of the Congo. Commission de Lutte contre les Epidemies a Kikwit. J Infect Dis 179(Suppl 1):S28–35. doi:10.1086/514318
Ruibal P, Oestereich L, Ludtke A, Becker-Ziaja B, Wozniak DM, Kerber R, Korva M, Cabeza-Cabrerizo M, Bore JA, Koundouno FR, Duraffour S, Weller R, Thorenz A, Cimini E, Viola D, Agrati C, Repits J, Afrough B, Cowley LA, Ngabo D, Hinzmann J, Mertens M, Vitoriano I, Logue CH, Boettcher JP, Pallasch E, Sachse A, Bah A, Nitzsche K, Kuisma E, Michel J, Holm T, Zekeng EG, Garcia-Dorival I, Wolfel R, Stoecker K, Fleischmann E, Strecker T, Di Caro A, Avsic-Zupanc T, Kurth A, Meschi S, Mely S, Newman E, Bocquin A, Kis Z, Kelterbaum A, Molkenthin P, Carletti F, Portmann J, Wolff S, Castilletti C, Schudt G, Fizet A, Ottowell LJ, Herker E, Jacobs T, Kretschmer B, Severi E, Ouedraogo N, Lago M, Negredo A, Franco L, Anda P, Schmiedel S, Kreuels B, Wichmann D, Addo MM, Lohse AW, De Clerck H, Nanclares C, Jonckheere S, Van Herp M, Sprecher A, Xiaojiang G, Carrington M, Miranda O, Castro CM, Gabriel M, Drury P, Formenty P, Diallo B, Koivogui L, Magassouba N, Carroll MW, Gunther S, Munoz-Fontela C (2016) Unique human immune signature of Ebola virus disease in Guinea. Nature 533(7601):100–104. doi:10.1038/nature17949
Sanchez A, Geisbert T, Feldmann H (2007a) Filoviridae: Marburg and Ebola Viruses, vol 1. Fields’ virology, 5th edn. Wolters kluwer/Lippincott Williams & Wilkins, Philadelphia
Sanchez A, Ksiazek TG, Rollin PE, Miranda ME, Trappier SG, Khan AS, Peters CJ, Nichol ST (1999) Detection and molecular characterization of Ebola viruses causing disease in human and nonhuman primates. J Infect Dis 179(Suppl 1):S164–169. doi:10.1086/514282
Sanchez A, Lukwiya M, Bausch D, Mahanty S, Sanchez AJ, Wagoner KD, Rollin PE (2004) Analysis of human peripheral blood samples from fatal and nonfatal cases of Ebola (Sudan) hemorrhagic fever: cellular responses, virus load, and nitric oxide levels. J Virol 78(19):10370–10377. doi:10.1128/JVI.78.19.10370-10377.2004
Sanchez A, Trappier SG, Mahy BW, Peters CJ, Nichol ST (1996) The virion glycoproteins of Ebola viruses are encoded in two reading frames and are expressed through transcriptional editing. Proc Natl Acad Sci U S A 93(8):3602–3607
Sanchez A, Wagoner KE, Rollin PE (2007b) Sequence-based human leukocyte antigen-B typing of patients infected with Ebola virus in Uganda in 2000: identification of alleles associated with fatal and nonfatal disease outcomes. J Infect Dis 196(Suppl 2):S329–336. doi:10.1086/520588
Schaible UE, Kaufmann SH (2007) Malnutrition and infection: complex mechanisms and global impacts. PLoS Med 4(5):e115. doi:10.1371/journal.pmed.0040115
Schieffelin JS, Shaffer JG, Goba A, Gbakie M, Gire SK, Colubri A, Sealfon RS, Kanneh L, Moigboi A, Momoh M, Fullah M, Moses LM, Brown BL, Andersen KG, Winnicki S, Schaffner SF, Park DJ, Yozwiak NL, Jiang PP, Kargbo D, Jalloh S, Fonnie M, Sinnah V, French I, Kovoma A, Kamara FK, Tucker V, Konuwa E, Sellu J, Mustapha I, Foday M, Yillah M, Kanneh F, Saffa S, Massally JL, Boisen ML, Branco LM, Vandi MA, Grant DS, Happi C, Gevao SM, Fletcher TE, Fowler RA, Bausch DG, Sabeti PC, Khan SH, Garry RF, Program KGHLF, Viral Hemorrhagic Fever C, Team WHOCR (2014) Clinical illness and outcomes in patients with Ebola in Sierra Leone. N Engl J Med 371(22):2092–2100. doi:10.1056/NEJMoa1411680
Schmaljohn A, Lewis GK (2016) Cell-targeting antibodies in immunity to Ebola. Pathog Dis 74(4). doi:10.1093/femspd/ftw021
Schuh AJ, Amman BR, Jones ME, Sealy TK, Uebelhoer LS, Spengler JR, Martin BE, Coleman-McCray JA, Nichol ST, Towner JS (2017) Modelling filovirus mainteinance in nature by experimental transmission of Marburg virus between Egyptian rousette bats. Nat Commun 8:14446. doi:10.1038/ncomms14446
Schwartz AJ, Alvarez X, Lackner AA (2002) Distribution and immunophenotype of DC-SIGN-expressing cells in SIV-infected and uninfected macaques. AIDS Res Hum Retroviruses 18(14):1021–1029. doi:10.1089/08892220260235380
Sharma P, Allison JP (2015) Immune checkpoint targeting in cancer therapy: toward combination strategies with curative potential. Cell 161(2):205–214. doi:10.1016/j.cell.2015.03.030
Simmons G, Reeves JD, Grogan CC, Vandenberghe LH, Baribaud F, Whitbeck JC, Burke E, Buchmeier MJ, Soilleux EJ, Riley JL, Doms RW, Bates P, Pohlmann S (2003) DC-SIGN and DC-SIGNR bind ebola glycoproteins and enhance infection of macrophages and endothelial cells. Virology 305(1):115–123
Smith LM, Hensley LE, Geisbert TW, Johnson J, Stossel A, Honko A, Yen JY, Geisbert J, Paragas J, Fritz E, Olinger G, Young HA, Rubins KH, Karp CL (2013) Interferon-beta therapy prolongs survival in rhesus macaque models of Ebola and Marburg hemorrhagic fever. J Infect Dis 208(2):310–318. doi:10.1093/infdis/jis921
Sobarzo A, Perelman E, Groseth A, Dolnik O, Becker S, Lutwama JJ, Dye JM, Yavelsky V, Lobel L, Marks RS (2012) Profiling the native specific human humoral immune response to Sudan Ebola virus strain Gulu by chemiluminescence enzyme-linked immunosorbent assay. Clin Vaccine Immunol CVI 19(11):1844–1852. doi:10.1128/CVI.00363-12
Sow MS, Etard JF, Baize S, Magassouba N, Faye O, Msellati P, Toure AI, Savane I, Barry M, Delaporte E, Postebogui Study G (2016) New evidence of long-lasting persistence of Ebola Virus genetic material in Semen of survivors. J Infect Dis. doi:10.1093/infdis/jiw078
Sueblinvong V, Johnson DW, Weinstein GL, Connor MJ Jr, Crozier I, Liddell AM, Franch HA, Wall BR, Kalil AC, Feldman M, Lisco SJ, Sevransky JE (2015) Critical care for multiple organ failure secondary to Ebola Virus disease in the United States. Crit Care Med 43(10):2066–2075. doi:10.1097/CCM.0000000000001197
Sundar K, Boesen A, Coico R (2007) Computational prediction and identification of HLA-A2.1-specific Ebola virus CTL epitopes. Virology 360(2):257–263. doi:10.1016/j.virol.2006.09.042
Takada A, Fujioka K, Tsuiji M, Morikawa A, Higashi N, Ebihara H, Kobasa D, Feldmann H, Irimura T, Kawaoka Y (2004) Human macrophage C-type lectin specific for galactose and N-acetylgalactosamine promotes filovirus entry. J Virol 78(6):2943–2947
Tamburini BA, Burchill MA, Kedl RM (2014) Antigen capture and archiving by lymphatic endothelial cells following vaccination or viral infection. Nat Commun 5:3989. doi:10.1038/ncomms4989
Tappe D, Perez-Giron JV, Just-Nubling G, Schuster G, Gomez-Medina S, Gunther S, Munoz-Fontela C, Schmidt-Chanasit J (2016) Sustained elevated cytokine levels during recovery phase of Mayaro Virus infection. Emerg Infect Dis 22(4):750–752. doi:10.3201/eid2204.151502
Team WHOER, Agua-Agum J, Ariyarajah A, Blake IM, Cori A, Donnelly CA, Dorigatti I, Dye C, Eckmanns T, Ferguson NM, Fowler RA, Fraser C, Garske T, Hinsley W, Jombart T, Mills HL, Murthy S, Nedjati Gilani G, Nouvellet P, Pelletier L, Riley S, Schumacher D, Shah A, Van Kerkhove MD (2015) Ebola virus disease among children in West Africa. N Engl J Med 372(13):1274–1277. doi:10.1056/NEJMc1415318
Terajima M, Ennis FA (2011) T cells and pathogenesis of hantavirus cardiopulmonary syndrome and hemorrhagic fever with renal syndrome. Viruses 3(7):1059–1073. doi:10.3390/v3071059
Towner JS, Rollin PE, Bausch DG, Sanchez A, Crary SM, Vincent M, Lee WF, Spiropoulou CF, Ksiazek TG, Lukwiya M, Kaducu F, Downing R, Nichol ST (2004) Rapid diagnosis of Ebola hemorrhagic fever by reverse transcription-PCR in an outbreak setting and assessment of patient viral load as a predictor of outcome. J Virol 78(8):4330–4341
Uyeki TM, Erickson BR, Brown S, McElroy AK, Cannon D, Gibbons A, Sealy T, Kainulainen MH, Schuh AJ, Kraft CS, Mehta AK, Lyon GM 3rd, Varkey JB, Ribner BS, Ellison RT 3rd, Carmody E, Nau GJ, Spiropoulou C, Nichol ST, Stroher U (2016) Ebola Virus persistence in Semen of male survivors. Clin Infect Dis Official Publ Infect Dis Soc Am 62(12):1552–1555. doi:10.1093/cid/ciw202
Uyeki TM, Mehta AK, Davey RT, Jr., Liddell AM, Wolf T, Vetter P, Schmiedel S, Grunewald T, Jacobs M, Arribas JR, Evans L, Hewlett AL, Brantsaeter AB, Ippolito G, Rapp C, Hoepelman AI, Gutman J, Working Group of the USECNoCMoEVDPitUS, Europe (2016) Clinical management of Ebola Virus disease in the United States and Europe. N Engl J Med 374(7):636–646. doi:10.1056/NEJMoa1504874
van Griensven J, Edwards T, de Lamballerie X, Semple MG, Gallian P, Baize S, Horby PW, Raoul H, Magassouba N, Antierens A, Lomas C, Faye O, Sall AA, Fransen K, Buyze J, Ravinetto R, Tiberghien P, Claeys Y, De Crop M, Lynen L, Bah EI, Smith PG, Delamou A, De Weggheleire A, Haba N, Ebola-Tx C (2016) Evaluation of convalescent plasma for Ebola Virus disease in Guinea. N Engl J Med 374(1):33–42. doi:10.1056/NEJMoa1511812
van Paassen J, Bauer MP, Arbous MS, Visser LG, Schmidt-Chanasit J, Schilling S, Olschlager S, Rieger T, Emmerich P, Schmetz C, van de Berkmortel F, van Hoek B, van Burgel ND, Osterhaus AD, Vossen AC, Gunther S, van Dissel JT (2012) Acute liver failure, multiorgan failure, cerebral oedema, and activation of proangiogenic and antiangiogenic factors in a case of Marburg haemorrhagic fever. Lancet Infect Dis 12(8):635–642. doi:10.1016/S1473-3099(12)70018-X
Varkey JB, Shantha JG, Crozier I, Kraft CS, Lyon GM, Mehta AK, Kumar G, Smith JR, Kainulainen MH, Whitmer S, Stroher U, Uyeki TM, Ribner BS, Yeh S (2015) Persistence of Ebola Virus in ocular fluid during convalescence. N Engl J Med 372(25):2423–2427. doi:10.1056/NEJMoa1500306
Villinger F, Rollin PE, Brar SS, Chikkala NF, Winter J, Sundstrom JB, Zaki SR, Swanepoel R, Ansari AA, Peters CJ (1999) Markedly elevated levels of interferon (IFN)-gamma, IFN-alpha, interleukin (IL)-2, IL-10, and tumor necrosis factor-alpha associated with fatal Ebola virus infection. J Infect Dis 179(Suppl 1):S188–191. doi:10.1086/514283
Wahid R, Cannon MJ, Chow M (2005) Dendritic cells and macrophages are productively infected by poliovirus. J Virol 79(1):401–409. doi:10.1128/JVI.79.1.401-409.2005
Wahl-Jensen VM, Afanasieva TA, Seebach J, Stroher U, Feldmann H, Schnittler HJ (2005) Effects of Ebola virus glycoproteins on endothelial cell activation and barrier function. J Virol 79(16):10442–10450. doi:10.1128/JVI.79.16.10442-10450.2005
Wauquier N, Becquart P, Padilla C, Baize S, Leroy EM (2010) Human fatal zaire ebola virus infection is associated with an aberrant innate immunity and with massive lymphocyte apoptosis. PLoS Neglected Trop Dis 4(10). doi:10.1371/journal.pntd.0000837
Weiskopf D, Angelo MA, de Azeredo EL, Sidney J, Greenbaum JA, Fernando AN, Broadwater A, Kolla RV, De Silva AD, de Silva AM, Mattia KA, Doranz BJ, Grey HM, Shresta S, Peters B, Sette A (2013) Comprehensive analysis of dengue virus-specific responses supports an HLA-linked protective role for CD8+ T cells. Proc Natl Acad Sci U S A 110(22):E2046–2053. doi:10.1073/pnas.1305227110
Wherry EJ (2011) T cell exhaustion. Nat Immunol 12(6):492–499
Wilson JA, Hart MK (2001) Protection from Ebola virus mediated by cytotoxic T lymphocytes specific for the viral nucleoprotein. J Virol 75(6):2660–2664. doi:10.1128/JVI.75.6.2660-2664.2001
Wirth JP, Rohner F, Woodruff BA, Chiwile F, Yankson H, Koroma AS, Russel F, Sesay F, Dominguez E, Petry N, Shahab-Ferdows S, de Onis M, Hodges MH (2016) Anemia, micronutrient deficiencies, and Malaria in children and women in Sierra Leone prior to the Ebola outbreak—findings of a cross-sectional study. PLoS ONE 11(5):e0155031. doi:10.1371/journal.pone.0155031
Wolf T, Kann G, Becker S, Stephan C, Brodt HR, de Leuw P, Grunewald T, Vogl T, Kempf VA, Keppler OT, Zacharowski K (2015) Severe Ebola virus disease with vascular leakage and multiorgan failure: treatment of a patient in intensive care. Lancet 385(9976):1428–1435. doi:10.1016/S0140-6736(14)62384-9
Yan T, Mu J, Qin E, Wang Y, Liu L, Wu D, Jia H, Li Z, Guo T, Wang X, Qin Y, Li Y, Chen S, Zhang Y, Zhang J, Wu Y, Wang S, Li J (2015) Clinical characteristics of 154 patients suspected of having Ebola virus disease in the Ebola holding center of Jui Government Hospital in Sierra Leone during the 2014 Ebola outbreak. Eur J Clin Microbiol Infect Dis 34(10):2089–2095. doi:10.1007/s10096-015-2457-z
Yang Z, Delgado R, Xu L, Todd RF, Nabel EG, Sanchez A, Nabel GJ (1998) Distinct cellular interactions of secreted and transmembrane Ebola virus glycoproteins. Science 279(5353):1034–1037
Yang ZY, Duckers HJ, Sullivan NJ, Sanchez A, Nabel EG, Nabel GJ (2000) Identification of the Ebola virus glycoprotein as the main viral determinant of vascular cell cytotoxicity and injury. Nat Med 6(8):886–889. doi:10.1038/78645
Yen B, Mulder LC, Martinez O, Basler CF (2014) Molecular basis for ebolavirus VP35 suppression of human dendritic cell maturation. J Virol 88(21):12500–12510. doi:10.1128/JVI.02163-14
Yen JY, Garamszegi S, Geisbert JB, Rubins KH, Geisbert TW, Honko A, Xia Y, Connor JH, Hensley LE (2011) Therapeutics of ebola hemorrhagic Fever: whole-genome transcriptional analysis of successful disease mitigation. J Infect Dis 204(Suppl 3):S1043–1052. doi:10.1093/infdis/jir345
Zhang N, Bevan MJ (2011) CD8(+) T cells: foot soldiers of the immune system. Immunity 35(2):161–168. doi:10.1016/j.immuni.2011.07.010
Zhu FC, Hou LH, Li JX, Wu SP, Liu P, Zhang GR, Hu YM, Meng FY, Xu JJ, Tang R, Zhang JL, Wang WJ, Duan L, Chu K, Liang Q, Hu JL, Luo L, Zhu T, Wang JZ, Chen W (2015) Safety and immunogenicity of a novel recombinant adenovirus type-5 vector-based Ebola vaccine in healthy adults in China: preliminary report of a randomised, double-blind, placebo-controlled, phase 1 trial. Lancet 385(9984):2272–2279. doi:10.1016/S0140-6736(15)60553-0
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Muñoz-Fontela, C., McElroy, A.K. (2017). Ebola Virus Disease in Humans: Pathophysiology and Immunity. In: Mühlberger, E., Hensley, L., Towner, J. (eds) Marburg- and Ebolaviruses. Current Topics in Microbiology and Immunology, vol 411. Springer, Cham. https://doi.org/10.1007/82_2017_11
Download citation
DOI: https://doi.org/10.1007/82_2017_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-68946-3
Online ISBN: 978-3-319-68948-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)