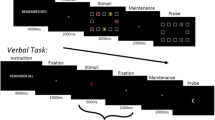
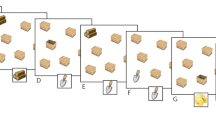
Abstract
Working memory impairments are frequently observed in patients with Alzheimer’s disease (AD) and Parkinson’s disease (PD). Recent research suggests that the mechanisms underlying these deficits might be dissociable using sensitive tasks, specifically those that rely on the reproduction of the exact quality of features held in memory.
In patients with AD, working memory impairments are mainly due to an increase in misbinding errors. They arise when patients misremember which features (e.g., color, orientation, shape, and location) belong to different objects held in memory. Hence, they erroneously report features that belong to items in memory other than the one they are probed on. This misbinding of features that belong to different objects in memory can be considered a form of interference between stored items. Such binding errors are evident even in presymptomatic individuals with familial AD (due to gene mutations) who do not have AD yet. Overall, these findings are in line with the role of the medial temporal lobes, and specifically the hippocampus, in retention of feature bindings, regardless of retention duration, i.e., in both short- or long-term memory.
Patients with PD, on the other hand, do not show increased misbinding. Their working memory deficits are associated with making more random errors or guesses. These random responses are not modulated by manipulations of their dopaminergic medication and hence may reflect involvement of non-dopaminergic neurotransmitters in this deficit. In addition, patients with PD demonstrate impairments in gating of information into relevant vs. irrelevant items in memory, a cognitive operation that is modulated by dopaminergic manipulation in line with a frontal executive effect of this neurotransmitter. Thus, although AD and PD are both associated with working memory impairments, these surface manifestations appear to be underpinned by very different mechanisms.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Baddeley A (2003) Working memory: looking back and looking forward. Nat Rev Neurosci 4:829–839. https://doi.org/10.1038/nrn1201
Bays PM, Catalao RFG, Husain M (2009) The precision of visual working memory is set by allocation of a shared resource. J Vis 9:7. https://doi.org/10.1167/9.10.7
Becker JT (1988) Working memory and secondary memory deficits in Alzheimer’s disease. J Clin Exp Neuropsychol 10:739–753. https://doi.org/10.1080/01688638808402811
Belleville S, Peretz I, Malenfant D (1996) Examination of the working memory components in normal aging and in dementia of the Alzheimer type. Neuropsychologia 34:195–207
Blackwell AD, Sahakian BJ, Vesey R, Semple JM, Robbins TW, Hodges JR (2004) Detecting dementia: novel neuropsychological markers of preclinical Alzheimer’s disease. Dement Geriatr Cogn Disord 17:42–48. https://doi.org/10.1159/000074081
Boeve BF (2010) REM sleep behavior disorder: updated review of the core features, the REM sleep behavior disorder-neurodegenerative disease association, evolving concepts, controversies, and future directions. Ann N Y Acad Sci 1184:15–54. https://doi.org/10.1111/j.1749-6632.2009.05115.x
Boot BP, Boeve BF, Roberts RO, Ferman TJ, Geda YE, Pankratz VS, Ivnik RJ, Smith GE, McDade E, Christianson TJH, Knopman DS, Tangalos EG, Silber MH, Petersen RC (2012) Probable rapid eye movement sleep behavior disorder increases risk for mild cognitive impairment and Parkinson disease: a population-based study. Ann Neurol 71:49–56. https://doi.org/10.1002/ana.22655
Braak H, Braak E (1998) Evolution of neuronal changes in the course of Alzheimer’s disease. J Neural Transm Suppl 53:127–140
Brier MR, Gordon B, Friedrichsen K, McCarthy J, Stern A, Christensen J, Owen C, Aldea P, Su Y, Hassenstab J, Cairns NJ, Holtzman DM, Fagan AM, Morris JC, Benzinger TLS, Ances BM (2016) Tau and Aβ imaging, CSF measures, and cognition in Alzheimer’s disease. Sci Transl Med 8:338ra66. https://doi.org/10.1126/scitranslmed.aaf2362
Carlesimo GA, Fadda L, Lorusso S, Caltagirone C (1994) Verbal and spatial memory spans in Alzheimer’s and multi-infarct dementia. Acta Neurol Scand 89:132–138
Cashdollar N, Duncan JS, Duzel E (2011) Challenging the classical distinction between long-term and short-term memory: reconsidering the role of the hippocampus. Future Neurol 6:351–362. https://doi.org/10.2217/fnl.11.12
Clark LN, Kartsaklis LA, Wolf Gilbert R, Dorado B, Ross BM, Kisselev S, Verbitsky M, Mejia-Santana H, Cote LJ, Andrews H, Vonsattel J-P, Fahn S, Mayeux R, Honig LS, Marder K (2009) Association of glucocerebrosidase mutations with dementia with lewy bodies. Arch Neurol 66:578–583. https://doi.org/10.1001/archneurol.2009.54
Cools R, Miyakawa A, Sheridan M, D’Esposito M (2010) Enhanced frontal function in Parkinson’s disease. Brain J Neurol 133:225–233. https://doi.org/10.1093/brain/awp301
Dalrymple-Alford JC, Kalders AS, Jones RD, Watson RW (1994) A central executive deficit in patients with Parkinson’s disease. J Neurol Neurosurg Psychiatry 57:360–367
Davachi L (2006) Item, context and relational episodic encoding in humans. Curr Opin Neurobiol 16:693–700. https://doi.org/10.1016/j.conb.2006.10.012
Della Sala S, Parra MA, Fabi K, Luzzi S, Abrahams S (2012) Short-term memory binding is impaired in AD but not in non-AD dementias. Neuropsychologia 50:833–840. https://doi.org/10.1016/j.neuropsychologia.2012.01.018
Dubois B, Ruberg M, Javoy-Agid F, Ploska A, Agid Y (1983) A subcortico-cortical cholinergic system is affected in Parkinson’s disease. Brain Res 288:213–218
Dujardin K, Degreef JF, Rogelet P, Defebvre L, Destee A (1999) Impairment of the supervisory attentional system in early untreated patients with Parkinson’s disease. J Neurol 246:783–788
Eichenbaum H, Yonelinas AP, Ranganath C (2007) The medial temporal lobe and recognition memory. Annu Rev Neurosci 30:123–152. https://doi.org/10.1146/annurev.neuro.30.051606.094328
Emir UE, Tuite PJ, Öz G (2012) Elevated pontine and putamenal GABA levels in mild-moderate Parkinson disease detected by 7 tesla proton MRS. PLoS One 7:e30918
Esfahani-Bayerl N, Finke C, Braun M, Düzel E, Heekeren HR, Holtkamp M, Hasper D, Storm C, Ploner CJ (2016) Visuo-spatial memory deficits following medial temporal lobe damage: a comparison of three patient groups. Neuropsychologia 81:168–179. https://doi.org/10.1016/j.neuropsychologia.2015.12.024
Fallon SJ, Zokaei N, Husain M (2016) Causes and consequences of limitations in visual working memory. Ann N Y Acad Sci 1369:40–54. https://doi.org/10.1111/nyas.12992
Fallon SJ, Mattiesing RM, Muhammed K, Manohar S, Husain M (2017) Fractionating the neurocognitive mechanisms underlying working memory: independent effects of dopamine and Parkinson’s disease. Cereb Cortex 27:5727–5738. https://doi.org/10.1093/cercor/bhx242
Ferini-Strambi L, Di Gioia MR, Castronovo V, Oldani A, Zucconi M, Cappa SF (2004) Neuropsychological assessment in idiopathic REM sleep behavior disorder (RBD): does the idiopathic form of RBD really exist? Neurology 62:41–45
Fox NC, Warrington EK, Seiffer AL, Agnew SK, Rossor MN (1998) Presymptomatic cognitive deficits in individuals at risk of familial Alzheimer’s disease. A longitudinal prospective study. Brain J Neurol 121(Pt 9):1631–1639
Gorgoraptis N, Catalao RFG, Bays PM, Husain M (2011) Dynamic updating of working memory resources for visual objects. J Neurosci 31:8502–8511. https://doi.org/10.1523/JNEUROSCI.0208-11.2011
Gratwicke J, Jahanshahi M, Foltynie T (2015) Parkinson’s disease dementia: a neural networks perspective. Brain J Neurol 138:1454–1476. https://doi.org/10.1093/brain/awv104
Gruszka A, Bor D, Barker RR, Necka E, Owen AM (2016) The role of executive processes in working memory deficits in Parkinson’s disease. Pol Psychol Bull 47:123–130. https://doi.org/10.1515/ppb-2016-0013
Hannula DE, Tranel D, Cohen NJ (2006) The long and the short of it: relational memory impairments in amnesia, even at short lags. J Neurosci 26:8352–8359
Hartley T, Bird CM, Chan D, Cipolotti L, Husain M, Vargha-Khadem F, Burgess N (2007) The hippocampus is required for short-term topographical memory in humans. Hippocampus 17:34–48. https://doi.org/10.1002/hipo.20240
Hasselmo ME, Sarter M (2011) Modes and models of forebrain cholinergic neuromodulation of cognition. Neuropsychopharmacology 36:52–73. https://doi.org/10.1038/npp.2010.104
Heise V, Filippini N, Trachtenberg AJ, Suri S, Ebmeier KP, Mackay CE (2014) Apolipoprotein E genotype, gender and age modulate connectivity of the hippocampus in healthy adults. NeuroImage 98:23–30. https://doi.org/10.1016/j.neuroimage.2014.04.081
Hughes LE, Altena E, Barker RA, Rowe JB (2013) Perseveration and choice in Parkinson’s disease: the impact of progressive frontostriatal dysfunction on action decisions. Cereb Cortex 23:1572–1581
Hulme C, Lee G, Brown GD (1993) Short-term memory impairments in Alzheimer-type dementia: evidence for separable impairments of articulatory rehearsal and long-term memory. Neuropsychologia 31:161–172
Iranzo A, Molinuevo JL, Santamaría J, Serradell M, Martí MJ, Valldeoriola F, Tolosa E (2006) Rapid-eye-movement sleep behaviour disorder as an early marker for a neurodegenerative disorder: a descriptive study. Lancet Neurol 5:572–577. https://doi.org/10.1016/S1474-4422(06)70476-8
Jeneson A, Mauldin KN, Squire LR (2010) Intact working memory for relational information after medial temporal lobe damage. J Neurosci 30:13624–13629. https://doi.org/10.1523/JNEUROSCI.2895-10.2010
Joseph S, Teki S, Kumar S, Husain M, Griffiths TD (2016) Resource allocation models of auditory working memory. Brain Res 1640:183–192. https://doi.org/10.1016/j.brainres.2016.01.044
Kehagia AA, Barker RA, Robbins TW (2010) Neuropsychological and clinical heterogeneity of cognitive impairment and dementia in patients with Parkinson’s disease. Lancet Neurol 9:1200–1213. https://doi.org/10.1016/S1474-4422(10)70212-X
Kish SJ, Tong J, Hornykiewicz O, Rajput A, Chang L-J, Guttman M, Furukawa Y (2008) Preferential loss of serotonin markers in caudate versus putamen in Parkinson’s disease. Brain 131:120–131
Koen JD, Borders AA, Petzold MT, Yonelinas AP (2016) Visual short-term memory for high resolution associations is impaired in patients with medial temporal lobe damage. Hippocampus 27:184–193. https://doi.org/10.1002/hipo.22682
Konkel A, Cohen NJ (2009) Relational memory and the hippocampus: representations and methods. Front Neurosci 3:166–174. https://doi.org/10.3389/neuro.01.023.2009
Kroener S, Chandler LJ, Phillips PEM, Seamans JK (2009) Dopamine modulates persistent synaptic activity and enhances the signal-to-noise ratio in the prefrontal cortex. PLoS One 4:e6507. https://doi.org/10.1371/journal.pone.0006507
Lange KW, Robbins TW, Marsden CD, James M, Owen AM, Paul GM (1992) L-dopa withdrawal in Parkinson’s disease selectively impairs cognitive performance in tests sensitive to frontal lobe dysfunction. Psychopharmacology 107:394–404
Lau KM, Parikh M, Harvey DJ, Huang C-J, Farias ST (2015) Early cognitively based functional limitations predict loss of independence in instrumental activities of daily living in older adults. J Int Neuropsychol Soc 21:688–698. https://doi.org/10.1017/S1355617715000818
Lawton M, Baig F, Rolinski M, Ruffman C, Nithi K, May MT, Ben-Shlomo Y, Hu MTM (2015) Parkinson’s disease subtypes in the Oxford Parkinson disease Centre (OPDC) discovery cohort. J Park Dis 5:269–279. https://doi.org/10.3233/JPD-140523
Lewis SJG, Foltynie T, Blackwell AD, Robbins TW, Owen AM, Barker RA (2005) Heterogeneity of Parkinson’s disease in the early clinical stages using a data driven approach. J Neurol Neurosurg Psychiatry 76:343–348
Liang Y, Pertzov Y, Nicholas JM, Henley SMD, Crutch S, Woodward F, Leung K, Fox NC, Husain M (2016) Visual short-term memory binding deficit in familial Alzheimer’s disease. Cortex 78:150–164. https://doi.org/10.1016/j.cortex.2016.01.015
Libby LA, Hannula DE, Ranganath C (2014) Medial temporal lobe coding of item and spatial information during relational binding in working memory. J Neurosci 34:14233–14242. https://doi.org/10.1523/JNEUROSCI.0655-14.2014
Linn RT, Wolf PA, Bachman DL, Knoefel JE, Cobb JL, Belanger AJ, Kaplan EF, D’Agostino RB (1995) The “preclinical phase” of probable Alzheimer’s disease. A 13-year prospective study of the Framingham cohort. Arch Neurol 52:485–490
Luck V (1997) The capacity of visual working memory for features and conjunctions. Nature 390:279–281. https://doi.org/10.1038/36846
Ma WJ, Husain M, Bays PM (2014) Changing concepts of working memory. Nat Neurosci 17:347–356. https://doi.org/10.1038/nn.3655
Martin A, Brouwers P, Cox C, Fedio P (1985) On the nature of the verbal memory deficit in Alzheimer’s disease. Brain Lang 25:323–341
Miller E (1973) Short- and long-term memory in patients with presenile dementia (Alzheimer’s disease). Psychol Med 3:221–224
Muslimovic D, Post B, Speelman JD, Schmand B (2005) Cognitive profile of patients with newly diagnosed Parkinson disease. Neurology 65:1239–1245. https://doi.org/10.1212/01.wnl.0000180516.69442.95
Myers RH, Schaefer EJ, Wilson PW, D’Agostino R, Ordovas JM, Espino A, Au R, White RF, Knoefel JE, Cobb JL, McNulty KA, Beiser A, Wolf PA (1996) Apolipoprotein E epsilon4 association with dementia in a population-based study: the Framingham study. Neurology 46:673–677
Nadel L, Peterson MA (2013) The hippocampus: part of an interactive posterior representational system spanning perceptual and memorial systems. J Exp Psychol Gen 142:1242–1254. https://doi.org/10.1037/a0033690
Neumann J, Bras J, Deas E, O’Sullivan SS, Parkkinen L, Lachmann RH, Li A, Holton J, Guerreiro R, Paudel R, Segarane B, Singleton A, Lees A, Hardy J, Houlden H, Revesz T, Wood NW (2009) Glucocerebrosidase mutations in clinical and pathologically proven Parkinson’s disease. Brain 132:1783–1794. https://doi.org/10.1093/brain/awp044
Olson IR, Moore KS, Stark M, Chatterjee A (2006) Visual working memory is impaired when the medial temporal lobe is damaged. J Cogn Neurosci 18:1087–1097. https://doi.org/10.1162/jocn.2006.18.7.1087
Owen AM, Beksinska M, James M, Leigh PN, Summers BA, Marsden CD, Quinn NP, Sahakian BJ, Robbins TW (1993) Visuospatial memory deficits at different stages of Parkinson’s disease. Neuropsychologia 31:627–644
Owen AM, Iddon JL, Hodges JR, Summers BA, Robbins TW (1997) Spatial and non-spatial working memory at different stages of Parkinson’s disease. Neuropsychologia 35:519–532
Parra MA, Abrahams S, Fabi K, Logie R, Luzzi S, Della Sala S (2009) Short-term memory binding deficits in Alzheimer’s disease. Brain J Neurol 132:1057–1066. https://doi.org/10.1093/brain/awp036
Parra MA, Abrahams S, Logie RH, Méndez LG, Lopera F, Della Sala S (2010) Visual short-term memory binding deficits in familial Alzheimer’s disease. Brain J Neurol 133:2702–2713. https://doi.org/10.1093/brain/awq148
Parra MA, Sala SD, Abrahams S, Logie RH, Méndez LG, Lopera F (2011) Specific deficit of colour-colour short-term memory binding in sporadic and familial Alzheimer’s disease. Neuropsychologia 49:1943–1952. https://doi.org/10.1016/j.neuropsychologia.2011.03.022
Pastores GM, Hughes DA (1993) Gaucher disease. In: Pagon RA, Adam MP, Bird TD, Dolan CR, Fong C-T, Stephens K (eds) GeneReviews™. University of Washington, Seattle
Perry RJ, Hodges JR (2000) Fate of patients with questionable (very mild) Alzheimer’s disease: longitudinal profiles of individual subjects’ decline. Dement Geriatr Cogn Disord 11:342–349. https://doi.org/10.1159/000017264
Pertzov Y, Miller TD, Gorgoraptis N, Caine D, Schott JM, Butler C, Husain M (2013) Binding deficits in memory following medial temporal lobe damage in patients with voltage-gated potassium channel complex antibody-associated limbic encephalitis. Brain J Neurol 136:2474–2485. https://doi.org/10.1093/brain/awt129
Poewe W, Berger W, Benke TH, Schelosky L (1991) High-speed memory scanning in Parkinson’s disease: adverse effects of levodopa. Ann Neurol 29:670–673
Postle BR, Jonides J, Smith EE, Corkin S, Growdon JH (1997) Spatial, but not object, delayed response is impaired in early Parkinson’s disease. Neuropsychology 11:171–179
Postuma RB, Gagnon JF, Vendette M, Montplaisir JY (2009) Markers of neurodegeneration in idiopathic rapid eye movement sleep behaviour disorder and Parkinson’s disease. Brain J Neurol 132:3298–3307. https://doi.org/10.1093/brain/awp244
Ridha BH, Barnes J, Bartlett JW, Godbolt A, Pepple T, Rossor MN, Fox NC (2006) Tracking atrophy progression in familial Alzheimer’s disease: a serial MRI study. Lancet Neurol 5:828–834. https://doi.org/10.1016/S1474-4422(06)70550-6
Rolinski M, Zokaei N, Baig F, Giehl K, Quinnell T, Zaiwalla Z, Mackay CE, Husain M, Hu MTM (2015) Visual short-term memory deficits in REM sleep behaviour disorder mirror those in Parkinson’s disease. Brain 139:47–53. https://doi.org/10.1093/brain/awv334
Sando SB, Melquist S, Cannon A, Hutton ML, Sletvold O, Saltvedt I, White LR, Lydersen S, Aasly JO (2008) APOE epsilon 4 lowers age at onset and is a high risk factor for Alzheimer’s disease; a case control study from Central Norway. BMC Neurol 8:9. https://doi.org/10.1186/1471-2377-8-9
Sardi SP, Clarke J, Kinnecom C, Tamsett TJ, Li L, Stanek LM, Passini MA, Grabowski GA, Schlossmacher MG, Sidman RL, Cheng SH, Shihabuddin LS (2011) CNS expression of glucocerebrosidase corrects α-synuclein pathology and memory in a mouse model of Gaucher-related synucleinopathy. Proc Natl Acad Sci 108:12101–12106. https://doi.org/10.1073/pnas.1108197108
Sato C, Morgan A, Lang AE, Salehi-Rad S, Kawarai T, Meng Y, Ray PN, Farrer LA, St George-Hyslop P, Rogaeva E (2005) Analysis of the glucocerebrosidase gene in Parkinson’s disease. Mov Disord 20:367–370. https://doi.org/10.1002/mds.20319
Saunders AM, Strittmatter WJ, Schmechel D, George-Hyslop PHS, Pericak-Vance MA, Joo SH, Rosi BL, Gusella JF, Crapper-MacLachlan DR, Alberts MJ, Hulette C, Crain B, Goldgaber D, Roses AD (1993) Association of apolipoprotein E allele ϵ4 with late-onset familial and sporadic Alzheimer’s disease. Neurology 43:1467–1467. https://doi.org/10.1212/WNL.43.8.1467
Savica R, Rocca WA, Ahlskog J (2010) When does Parkinson disease start? Arch Neurol 67:798–801. https://doi.org/10.1001/archneurol.2010.135
Sawaguchi T, Goldman-Rakic P (1991) D1 dopamine receptors in prefrontal cortex: involvement in working memory. Science 251:947–950. https://doi.org/10.1126/science.1825731
Schenck CH, Boeve BF, Mahowald MW (2013) Delayed emergence of a parkinsonian disorder or dementia in 81% of older men initially diagnosed with idiopathic rapid eye movement sleep behavior disorder: a 16-year update on a previously reported series. Sleep Med 14:744–748. https://doi.org/10.1016/j.sleep.2012.10.009
Selikhova M, Williams DR, Kempster PA, Holton JL, Revesz T, Lees AJ (2009) A clinico-pathological study of subtypes in Parkinson’s disease. Brain J Neurol 132:2947–2957. https://doi.org/10.1093/brain/awp234
Sidransky E, Nalls MA, Aasly JO, Aharon-Peretz J, Annesi G, Barbosa ER, Bar-Shira A, Berg D, Bras J, Brice A, Chen C-M, Clark LN, Condroyer C, de Marco EV, Dürr A, Eblan MJ, Fahn S, Farrer MJ, Fung H-C, Gan-Or Z, Gasser T, Gershoni-Baruch R, Giladi N, Griffith A, Gurevich T, Januario C, Kropp P, Lang AE, Lee-Chen G-J, Lesage S, Marder K, Mata IF, Mirelman A, Mitsui J, Mizuta I, Nicoletti G, Oliveira C, Ottman R, Orr-Urtreger A, Pereira LV, Quattrone A, Rogaeva E, Rolfs A, Rosenbaum H, Rozenberg R, Samii A, Samaddar T, Schulte C, Sharma M, Singleton A, Spitz M, Tan E-K, Tayebi N, Toda T, Troiano AR, Tsuji S, Wittstock M, Wolfsberg TG, Wu Y-R, Zabetian CP, Zhao Y, Ziegler SG (2009) Multicenter analysis of glucocerebrosidase mutations in Parkinson’s disease. N Engl J Med 361:1651–1661. https://doi.org/10.1056/NEJMoa0901281
Spinnler H, Sala SD, Bandera R, Baddeley A (1988) Dementia, ageing, and the structure of human memory. Cogn Neuropsychol 5:193–211. https://doi.org/10.1080/02643298808252933
Suri S, Heise V, Trachtenberg AJ, Mackay CE (2013) The forgotten APOE allele: a review of the evidence and suggested mechanisms for the protective effect of APOE ɛ2. Neurosci Biobehav Rev 37:2878–2886. https://doi.org/10.1016/j.neubiorev.2013.10.010
Uitvlugt MG, Pleskac TJ, Ravizza SM (2016) The nature of working memory gating in Parkinson’s disease: a multi-domain signal detection examination. Cogn Affect Behav Neurosci 16:289–301
van Geldorp B, Bouman Z, Hendriks MPH, Kessels RPC (2014) Different types of working memory binding in epilepsy patients with unilateral anterior temporal lobectomy. Brain Cogn 85:231–238. https://doi.org/10.1016/j.bandc.2013.12.009
Verbaan D, Marinus J, Visser M, van Rooden SM, Stiggelbout AM, Middelkoop HAM, van Hilten JJ (2007) Cognitive impairment in Parkinson’s disease. J Neurol Neurosurg Psychiatry 78:1182–1187. https://doi.org/10.1136/jnnp.2006.112367
Watrous AJ, Tandon N, Conner CR, Pieters T, Ekstrom AD (2013) Frequency-specific network connectivity increases underlie accurate spatiotemporal memory retrieval. Nat Neurosci 16:349–356. https://doi.org/10.1038/nn.3315
Williams GC (1957) Pleiotropy, natural selection, and the evolution of senescence. Evolution 11:398–411. https://doi.org/10.2307/2406060
Winterer G, Weinberger DR (2004) Genes, dopamine and cortical signal-to-noise ratio in schizophrenia. Trends Neurosci 27:683–690. https://doi.org/10.1016/j.tins.2004.08.002
Wolk DA, Dickerson BC, Alzheimer’s Disease Neuroimaging Initiative (2010) Apolipoprotein E (APOE) genotype has dissociable effects on memory and attentional-executive network function in Alzheimer’s disease. Proc Natl Acad Sci U S A 107:10256–10261. https://doi.org/10.1073/pnas.1001412107
Wong K, Sidransky E, Verma A, Mixon T, Sandberg GD, Wakefield LK, Morrison A, Lwin A, Colegial C, Allman JM, Schiffmann R (2004) Neuropathology provides clues to the pathophysiology of Gaucher disease. Mol Genet Metab 82:192–207. https://doi.org/10.1016/j.ymgme.2004.04.011
Yau W-YW, Tudorascu DL, McDade EM, Ikonomovic S, James JA, Minhas D, Mowrey W, Sheu LK, Snitz BE, Weissfeld L, Gianaros PJ, Aizenstein HJ, Price JC, Mathis CA, Lopez OL, Klunk WE (2015) Longitudinal assessment of neuroimaging and clinical markers in autosomal dominant Alzheimer’s disease: a prospective cohort study. Lancet Neurol 14:804–813. https://doi.org/10.1016/S1474-4422(15)00135-0
Yonelinas AP (2013) The hippocampus supports high-resolution binding in the service of perception, working memory and long-term memory. Behav Brain Res 254:34–44. https://doi.org/10.1016/j.bbr.2013.05.030
Zarow C, Lyness SA, Mortimer JA, Chui HC (2003) Neuronal loss is greater in the locus coeruleus than nucleus basalis and substantia nigra in Alzheimer and Parkinson diseases. Arch Neurol 60:337–341
Zhang W, Luck SJ (2008) Discrete fixed-resolution representations in visual working memory. Nature 453:233–235
Zimmermann JF, Butler CR (2018) Accelerated long-term forgetting in asymptomatic APOE ε4 carriers. Lancet Neurol 17:394–395. https://doi.org/10.1016/S1474-4422(18)30078-4
Zokaei N, Gorgoraptis N, Bahrami B, Bays PM, Husain M (2011) Precision of working memory for visual motion sequences and transparent motion surfaces. J Vis 11:2. https://doi.org/10.1167/11.14.2
Zokaei N, Burnett Heyes S, Gorgoraptis N, Budhdeo S, Husain M (2014a) Working memory recall precision is a more sensitive index than span. J Neuropsychol 9:319–329. https://doi.org/10.1111/jnp.12052
Zokaei N, McNeill A, Proukakis C, Beavan M, Jarman P, Korlipara P, Hughes D, Mehta A, Hu MTM, Schapira AHV, Husain M (2014b) Visual short-term memory deficits associated with GBA mutation and Parkinson’s disease. Brain J Neurol 137:2303–2311. https://doi.org/10.1093/brain/awu143
Zokaei N, Giehl K, Sillence A, Neville MJ, Karpe F, Nobre AC, Husain M (2017) Sex and APOE: a memory advantage in male APOE ε4 carriers in midlife. Cortex 88:98–105. https://doi.org/10.1016/j.cortex.2016.12.016
Zokaei N, Nour MM, Sillence A, Drew D, Adcock J, Stacey R, Voets N, Sen A, Husain M (2018) Binding deficits in visual short-term memory in patients with temporal lobe lobectomy. Hippocampus 29:63–67. https://doi.org/10.1002/hipo.22998
Zokaei N, Čepukaitytė G, Board AG, Mackay CE, Husain M, Nobre AC (2019a) Dissociable effects of the apolipoprotein-E (APOE) gene on short- and long-term memories. Neurobiol Aging 73:115–122. https://doi.org/10.1016/j.neurobiolaging.2018.09.017
Zokaei N, Nour MN, Sillence A, Drew D, Adcock J, Stacey R, Voets N, Sen A, Husain M (2019b) Binding deficits in visual short-term memory in patients with temporal lobe lobectomy. Hippocampus 29:63–67
Acknowledgments
This work is supported by the Wellcome Trust, British Academy, NIHR Oxford Biomedical Research Centre, and Wellcome Centre for Integrative Neuroscience.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Zokaei, N., Husain, M. (2019). Working Memory in Alzheimer’s Disease and Parkinson’s Disease. In: Hodgson, T. (eds) Processes of Visuospatial Attention and Working Memory. Current Topics in Behavioral Neurosciences, vol 41. Springer, Cham. https://doi.org/10.1007/7854_2019_103
Download citation
DOI: https://doi.org/10.1007/7854_2019_103
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-31025-7
Online ISBN: 978-3-030-31026-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)