Abstract
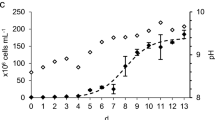
Microalgae are unicellular organisms capable of photosynthesis, turning sunlight and carbon dioxide (CO2) into rich biomass, precisely because of this definition in recent years, various sectors have been targeting the ability to reduce CO2 emissions and the capacity of simultaneously synthesize biomass which can later be used to produce bio-fuels. In respect to the capturing and utilization of CO2 emitted by different industries (cement, generation of electric energy, cellulose and ethanol production by fermentation of sugars) for the cultivation of microalgae destined to the production of bio-fuels, for example, biodiesel, ethanol or bio-oil. However, this research was developed in the Green Technologies Laboratory - Greentec / EQ / UFRJ, which focussed on the monitoring of CO2 consumption in microalgae Monoraphidium sp. cultivated in closed-window type photobioreactor, as well as, the characterization of the microalgal biomass produced in relation to the total lipid content (TL), convertible lipids into biodiesel (CLB), carbohydrates and proteins.The overall procedure involved the evaluation of the following parameters: injection of CO2, temperature (°C), lighting (μE m-2 s-1), pH, cell density (cells ml-1) and dry biomass. From the results of this study, it was observed that at the beginning of the culture (day 0) 0.79 g of CO2 were consumed per each gram of biomass produced. On the last day (12), 0.3 g of CO2 were consumed per gram of culture. On the sixth day of cultivation, the consumption of CO2 per gram of biomass increased, resulting in a CO2 consumption of 0.61 g. The best result was obtained on the second day of cultivation, when for each gram of biomass produced approximately 1.2 grams of CO2 were consumed. The biomass Monoraphidium sp. produced, contained 17,37 ± 3,27% of total lipid content, approximately 8.36 ± 2.69% of convertible lipids into biodiesel, 32% ± 3.37 of carbohydrates and 34.26% ± 0.41 of protein. The analysis performed by -GC-MS Gas chromatography showed the following composition of saturated fatty acids (SAFAs) mainly the C16:0 (palmitic), as in monounsaturated acids (MUFAs), in high quantity C18:1 (oleic) and polyunsaturated acids (PUFAs) mainly represented by C18:2 (Linoleic) and C18:3 (linolenic).
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Aresta, Michele, Angela Di benedetto, Maria Carone, Teresa Colonna and Carlo Fragale. 2005. “Production of Biodiesel from Macroalgae by Supercritical CO2 Extraction and Thermochemical Liquefaction,” 136-39.
Association of Official Analytical Chemists (A.O.A.C). 1995. Official methods of analysis of the Association of Official Analytical Chemists (method 991.20). 33. p. 10-12.
Bligh, G. and Dyer, W. 1959 A Rapid Method for Total Lipid Extraction and Purification. Canadian Journal of Bio- chemistry and Physiology, 37, 911-917.
Brindley C, Garcia-Malea MC, Acien FG, Fernandez JM, Garcia JL, Molina E. 2002 Influence of power supply in the feasibility of Phaeodactylum tricornutum cultures. Biotechnology and Bioengineering; 87:723-33.
Díaz, Gisel Chenard, Yordanka Reyes Cruz, René González Carliz, Rosa C Vitorino De Paula, Donato A Gomes Aranda, Marcellus A G Dario, Gustavo Saraiva Marassi, and Nelson C Furtado. 2015. Cultivation of Microalgae Monoraphidium sp, in the Plant Pilot the Grand Valle Bio Energy, for Biodiesel Production. 376, 370-378.
Holbrook, Gabriel P., Zachary Davidson, Robert a. Tatara, Norbert L. Ziemer, Kurt a. Rosentrater, W. Scott Grayburn,. 2014. “Use of the Microalga Monoraphidium sp. Grown in Wastewater as a Feedstock for Biodiesel: Cultivation and Fuel Characteristics.” Applied Energy 131: 386-93.
Kambourova R, Petkov G, Bankova V. 2006. Extracellular polar substances in cultures of the green alga Scenedesmus. 119:155-62.
Myklestad, s. &Haug, a. 1972. Production of carbohydrates by the marine Chaetocerosaffinis var. O Willei (Gran.)Hustedt. I. Effect of the concentration of nutrients in the cu1ture medium. J. exp mar. Biol. Ecol (9):125-136.
Razzak, Shaikh A., Mohammad M. Hossain, Rahima A. Lucky, Amarjeet S. Bassi, and Hugo de Lasa. 2013. “Integrated CO2 Capture, Wastewater Treatment and Biofuel Production by Microalgae culturing—A Review.” Renewable and Sustainable Energy Reviews 27. Elsevier. 630, 643: 622-53.
Richmond, A. (Ed), 1988. CRC Handbook of microalgal mass culture. Florida: CRC. 528p.
Sánchez-Saavedra. M. P &Voltolina, D., 2005. The growth rate, biomass production and composition of Chaetoceros sp. grown with different light sources. Aquacultural engineering 35: 161-165.
Schmid-Bondzynski-Ratzlaff. 2012. Extração e quantificação de lipídeos em microalgas. Modificado pela Universidade Federal de Viçosa. Departamento de Tecnologia de Alimentos.
Tavares, V.R; Costa, M; Medeiros, S.I.G and Souto, C.R.O. 2015. Analises de microscopia eletrônica e termogravimetriaparaacompanhamento da extração de lipídios da Monoraphidium sp. Congresso Brasileiro de Química. p 1.
The Emissions Gap Report 2015. A UNEP Synthesis Report. ISBN: 978-92-807-3491-1. http://uneplive.unep.org/media/docs/theme/13/EGR_2015_301115_lores.pdf.
Yoo, C., Jun, S.Y. and Lee, J.Y. 2010. Selection of Microalgae for Lipid Production under High Levels Carbon Dioxide. Bioresource Technology, 101, S71–S74. http://dx.doi.org/10.1016/j.biortech.2009.03.030
Yu, Xuya, Peng Zhao, Cian He, Junjun Li, Xianhua Tang, Junpei Zhou and Zunxi Huang. 2012. “Bioresource Technology Isolation of a Novel Strain of Monoraphidium sp. and Characterization of Its Potential Application as Biodiesel Feedstock.” Bioresource Technology 121. Elsevier Ltd: 256-62.
Acknowledgements
The authors acknowledge the support in terms of infrastructure provided by Greens Technologies Laboratory Greentec / EQ / UFRJ and for financing this research. The support of the National Counsel of Technological and Scientific Development (CNPQ) due to a scholarship offered. Also to the Food Technology Laboratory / EQ / UFRJ which provided support on some tests performed, and the National Center of Technology (CNTQ) in Venezuela which promotes the development of new technologies in the area of renewable energy.
Author information
Authors and Affiliations
Corresponding author
Additional information
1.Greentec Laboratory, School of Chemistry, Federal University of Rio de Janeiro, Rio de Janeiro,Brazil (*Corresponding author: figuera.andreina@gmail.com)
2.ECO 100 Company, Rio de Janeiro Brazil (luciano@ivig.coppe.ufrj.br).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Figuera, A., Reyes, Y., González, R. et al. Monitoring the CO2 consumption of Monoraphidium sp. microalgae: Characterization of algal biomass produced. Rev Latinoam Biotecnol Ambient Algal 7, 4 (2016). https://doi.org/10.7603/s40682-016-0004-y
Published:
DOI: https://doi.org/10.7603/s40682-016-0004-y