Abstract
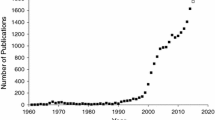
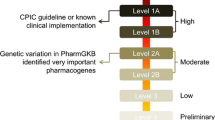
Adverse drug reactions (ADRs) constitute key factors in determining successful medication therapy in clinical situations. Integrative analysis of electronic medical record (EMR) data and use of proper analytical tools are requisite to conduct retrospective surveillance of clinical decisions on medications. Thus, we suggest that electronic medical recording and human genetic databases are considered together in future directions of pharmacovigilance. We analyzed EMR-based ADR studies indexed on PubMed during the period from 2005 to 2017 and retrospectively acquired 1161 (29.6%) articles describing drug-induced adverse reactions (e.g., liver, kidney, nervous system, immune system, and inflammatory responses). Of them, only 102 (8.79%) articles contained useful information to detect or predict ADRs in the context of clinical medication alerts. Since insufficiency of EMR datasets and their improper analyses may provide false warnings on clinical decision, efforts should be made to overcome possible problems on data-mining, analysis, statistics, and standardization. Thus, we address the characteristics and limitations on retrospective EMR database studies in hospital settings. Since gene expression and genetic variations among individuals impact ADRs, pharmacokinetics, and pharmacodynamics, appropriate paths for pharmacovigilance may be optimized using suitable databases available in public domain (e.g., genome-wide association studies (GWAS), non-coding RNAs, microRNAs, proteomics, and genetic variations), novel targets, and biomarkers. These efforts with new validated biomarker analyses would be of help to repurpose clinical and translational research infrastructure and ultimately future personalized therapy considering ADRs.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wang, S.D. (2013) Opportunities and challenges of clinical research in the bigdataera: from RCT to BCT. J. Thorac. Dis., 5, 721–723.
Liu, M., Melton, B.L., Ator, G. and Waitman, L.R. (2017) Integrating medication alert data into a clinical data repository to enable retrospective study of drug interaction alerts in clinical practice. AMIA Jt. Summits Transl. Sci. Proc., 2017, 213–220.
Jaspers, M.W., Smeulers, M., Vermeulen, H. and Peute, L.W. (2011) Effects of clinical decision-support systems on practitioner performance and patient outcomes: a synthesis of high-quality systematic review findings. J. Am. Med. Inform. Assoc., 18, 327–334.
Murdoch, T. and Detsky, A. (2013) The inevitable application of big data to healthcare. JAMA, 309, 1351–1352.
Springate, D.A., Kontopantelis, E., Ashcroft, D.M., Olier, I., Rosa Parisi, R., Chamapiwa, E. and Reeves, D. (2014) Clinical codes: an online clinical codes repository toimprove the validity and reproducibility of researchusing electronic medical records. PLoS ONE, 9, e99825.
Cheetham, T.C., Lee, J., Hunt, C.M., Niu, F., Reisinger, S., Murray, R., Powell, G. and Papay, J. (2014) An automated causality assessment algorithm to detect drug-induced liver injury in electronic medical record data. Pharmacoepidemiol. Drug Saf., 23, 601–608.
Björnsson, E.S. (2016) Hepatotoxicity by drugs: the most common implicated agents. Int. J. Mol. Sci., 17, 224.
Kullak-Ublick, G.A., Andrade, R.J., Merz, M., End, P. and Benesic, A. (2017) Drug-induced liver injury: recent advances in diagnosis and risk assessment. Gut, 66, 1154–1164.
Pirmohamed, M., James, S. and Meakin, S. (2004) Adverse drug reaction as cause of admission to hospital: prospective analysis of 18820 patients. BMJ, 329, 15–19.
Lazarous, J., Pomeranz, B. and Corey, P.N. (1998) Incidence of adverse drug reactions in hospitalized patients. A meta-analysis of prospective studies. JAMA, 279, 1200–1205.
Kohn, L.T., Corrgan, J.M. and Donaldson, M.S. (2000) To Error in Human; Building a Safer Health System, National Academy Press, Washington, DC.
Shin, J., Hunt, D.M., Suzuki, A., Papay, J.I., Beach, K.J. and Cheetham, T.C. (2013) Characterizing phenotypes and outcomes of drug-associated liver injury using electronic medical record data. Pharmacoepidemiol. Drug Saf., 22, 190–198.
Lin, K.J. and Schneeweiss, S. (2016) Considerations for the analysis of longitudinal electronic health records linked to claims data to study the effectiveness and safety of drugs. Clin. Pharmacol. Ther., 100, 147–157.
Andreea, F. and Marius, B. (2009) Adverse drug reactions in clinical practice: a causality assessment of a case of drug-induced pancreatitis. J. Gastrointest. Liver Dis., 18, 353–358.
Jha, A.K., Kuperman, G.J., Teich, J.M., Leape, L., SHea, B., Rittenberg, E., Burdick, E., Seger, D.L., Vander Vliet, M., Bates, D.W. (1998) Identifying adverse drug events: development of a computer-based monitor and comparison with chart review and stimulated voluntary report. J. Am. Med. Inform. Assoc., 5, 305–314.
Tisdale, J.E. and Miller, D.A. (2005) Drug-Induced Diseases: Prevention, Detection and Management, American Society of Health-System Pharmacist Press, Bethesda, Maryland, pp. 1004–1007.
Lee, Y.H., Kang, U.G. and Park, R.W. (2008) Development of adverse drug event surveillance system using BI technology. Int. J. Contents, 9, 106–113.
Miller, T.P., Ki, Y., Getz, K.D., Dudley, J., Burrows, E., Pennington, J., Ibrahimova, A., Fisher, B.T., Baqatell, R., Seif, A.E., Grundmeier, R. and Aplenc, R. (2017) Using electronic medical record data to report laboratory adverse events. Br. J. Haematol., 177, 283–286.
Pandit, A., Sachdeva, T. and Bafna, P. (2012) Drug-induced hepatotoxicity: a review. J. Appl. Pharm. Sci., 02, 233–243.
Mauben, M., Madigan, D. and Gerritis, C.M. (2007) The role of data mining in pharmacovigilance. Exp. Opin. Drug Saf., 14, 929–948.
Phansalkar, S., Hoffman, J.M., Nebeker, J.R. and Hurdle, J.F. (2007) Pharmacists versus nonpharmacists in adverse drug event detection: a meta-analysis and systematic review. Am. J. Health Syst. Pharm., 64, 842–849.
Phansalkar, S., Hoffman, J.M., Hurdle, J.F. and Patel, V.L. (2009) Understanding pharmacist decision making for adverse drug event (ADE) detection. J. Eval. Clin. Pract., 15, 266–275.
Tojios, S. and Fontana, R.J. (2011) Mechanisms of drug-induced liver injury: from bedside to bench. Nat. Rev. Gastroenterol. Hepatol., 8, 202–211.
Manuel, D.G., Rosella, L.C. and Stukel, T.A. (2010) Importance of accurately identifying disease in studies using electronic health records. BMJ, 341, c4226.
Pratt, N., Cham, E.W., Choi, N.K., Kimura, M., Kimura, T., Kubota, K., Lai, E.C., Man, L.L., Ooba, N., Park, B.J., Sato, T., Shin, J.Y., Wong, I.C., Kao Yang, Y.H. and Roughead, E.E. (2015) Prescription sequence symmetry analysis: assessing risk, temporality, and consistency for adverse drug reactions across datasets in five countries. Pharmacoepidemiol. Drug Saf., 24, 858–864.
Scholl, J.H.G., van Hunsel, F.P.A.M., Hak, E. and van Puijenbroek, E.P. (2017) A prediction model-based algorithm for computer-assisted database screening of adverse drug reactions in the Netherlands. Pharmacoepidemiol. Drug Saf., 27, 199–205.
Chen, X., Wang, Y., Wang, P., Lian, B., Li, C., Wamg, J., Li, X. and Jiang, W. (2015) Systematic analysis of the associations between adverse drug reactions and pathways. Biomed. Res. Int., 2015, 670949.
Kohonen, P., Parkkinen, J.A., Willinghaegen, E.L., Ceder, R., Wennerbern, K., Kaski, S. and Grafstrom, R.C. (2017) A transcriptomics data-driven gene space accurately predicts liver cytopathology and drug-induced liver injury. Nat. Commun., 8, 15932.
Koutkias, V.G. and Jaulent, M.-C. (2015) Computational approaches for pharmacovigilance signaldetection: toward integrated and semantically-enriched frameworks. Drug Saf., 38, 219–232.
Carr, D.F. and Pirmohamed, M. (2017) Biomarkers of adverse drug reactions. Exp. Biol. Med. (Maywood), 243, 291–299.
Xu, Q., Higgins, T. and Cembrowski, G.S. (2015) Limiting the testing of AST: a diagnostically nonspecific enzyme. Am. J. Clin. Pathol., 144, 423–426.
Alfirevic, A., Neely, D., Armitage, J., Chinoy, H., Cooper, R.G., Laaksonen, R., Carr, D.F., Bloch, K.M., Fahy, J., Hanson, A., Yue, Q.Y., Wadelius, M., Maitland-van Der Zee, A.H., Voora, D., Psaty, B.M., Paimer, C.N. and Pirmohamed, M. (2014) Phenotype standardization for statin-induced myotoxicity. Clin. Pharmacol. Ther., 96, 470–476.
Duma, R.J. and Siegel, A.L. (1965) Serum creatinine phohphokinase in acute myocardical infarction: diagnostic value. Arch. Intern. Med., 115, 443–451.
Kindermann, W. (2016) Creatine kinase levels after exercise. Dtsch. Arztebl. Int., 113, 344.
Wilke, R.A., Lin, D.W., Roden, D.M., Watkins, P.B., Flockhart, D., Zineh, I., Giacomini, K.M. and Krauss, R.M. (2007) Identifying genetic risk factors for serious adverse drug reactions: current progress and challenges. Nat. Rev. Drug Discov., 6, 904–916.
Redwood, A.J., Pavlos, R.K., White, K.D. and Phillips, E.J. (2018) Human leukocyte antigens: key regulators of T-cell mediated drug hypersensitivity. HLA, 91, 3–16.
Wang, C.-W., Chung, W.-H. and Hung, S.-I. (2017) Genetics of adverse drug reactions. eLS, 1–10.
Lea, J.D., Clarke, J.I., McGuire, N. and Antoine, D.J. (2016) Redox-dependent HMGB1 isoforms as pivotal co-ordinators of drug-induced liver injury: mechanistic biomarkers and therapeutic targets. Antioxid. Redox. Signal., 24, 652–665.
Chan, S.L., Jin, S., Loh, M. and Brunham, L.R. (2015) Progress in understanding the genomic basis for adverse drug reactions: a comprehensive review and focus on the role of ethnicity. Pharmacogenomics, 16, 1161–1178.
Nelson, M.R., Bacanu, S.A., Mosteller, M., Li, L., Bowman, C.E., Roses, A.D., Lai, E.H. and Ehm, M.G. (2009) Genome-wide approaches to identify pharmacogenetic contributions to adverse drug reactions. Pharmacogenomics J., 9, 23–33.
Pichler, W.J. (2003) Delayed drug hypersensitivity reactions. Ann. Intern. Med., 139, 683–693.
Ling, H., Fabbri, M. and Calin, G.A. (2013) MicroRNAs and other non-coding RNAs as targets for anticancer drug development. Nat. Rev. Drug Discov., 12, 847–865.
Matsui, M. and Corey, D.R. (2017) Non-coding RNAs as drug targets. Nat. Rev. Drug Discov., 16, 167–179.
Fu, X.D. (2014) Non-coding RNA: a new frontier in regulatory biology. Natl. Sci. Rev., 1, 190–204.
Maurano, M.T., Humbert, R., Rynes, E., Thurman, R.E., Haugen, E., Wang, H., Reynolds, A.P., Sandstrom, R., Qu, H., Brody, J., Shafer, A., Neri, F., Lee, K., Kutyavin, T., Stehling-Sun, S., Johnson, A.K., Canfield, T.K., Giste, E., Diegel, M., Bates, D., Hansen, R.S., Neph, S., Sabo, P.J., Heimfeld, S., Raubitschek, A., Ziegler, S., Cotsapas, C., Sotoodehnia, N., Glass, I., Sunyaev, S.R., Kaul, R. and Stamatoyannopoulos, J.A. (2012) Systematic localization of common disease-associated variation in regulatory DNA. Science, 337, 1190–1195.
Esteller, M. (2011) Non-coding RNAs in human disease. Nat. Rev. Genet., 12, 861–874.
Croce, C.M. (2009) Causes and consequences of microRNA dysregulation in cancer. Nat. Rev. Genet., 10, 704–714.
Nakajima, M. and Yokoi, T. (2011) MicroRNAs from biology to future pharmacotherapy: regulation of cytochrome P450s and nuclear receptors. Pharmacol. Ther., 131, 330–337.
Yu, A.M. (2009) Role of microRNAs in the regulation of drug metabolism and disposition. Exp. Opin. Drug Metab. Toxicol., 5, 1513–1528.
Zanger, U.M. and Schwab, M. (2013) Cytochrome P450 enzymes in drug metabolism: regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol. Ther., 138, 103–141.
Pan, Y.Z., Gao, W. and Yu, A.M. (2009) MicroRNAs regulate CYP3A4 expression via direct and indirect targeting. Drug Metab. Dispos., 37, 2112–2117.
Lu, Y. and Cederbaum, A.I. (2008) CYP2E1 and oxidative liver injury by alcohol. Free Radic. Biol. Med., 44, 723–738.
Mohri, T., Nakajima, M., Fukami, T., Takamiya, M., Aoki, Y. and Yokoi, T. (2010) Human CYP2E1 is regulated by miR-378. Biochem. Pharmacol., 79, 1045–1052.
Song, K.H., Li, T., Owsley, E. and Chiang, J.Y. (2010) A putative role of micro RNA in regulation of cholesterol 7alpha-hydroxylase expression in human hepatocytes. J. Lipid Res., 51, 2223–2233.
Komagata, S., Nakajima, M., Takagi, S., Mohri, T., Taniya, T. and Yokoi, T. (2009) Human CYP24 catalyzing the inactivation of calcitriol is post-transcriptionally regulated by miR-125b. Mol. Pharmacol., 76, 702–709.
Lin, H., Ewing, L.E., Koturbash, I., Gurley, B.J. and Miousse, I.R. (2017) MicroRNAs as biomarkers for liver injury: current knowledge, challenges and future prospects. Food Chem. Toxicol., 110, 229–239.
Marrone, A.K., Beland, F.A. and Pogribny, I.P. (2015) The role for microRNAs in drug toxicity and in safety assessment. Exp. Opin. Drug Metab. Toxicol., 11, 601–611.
Antoine, D.J., Dear, J.W., Lewis, P.S., Platt, V., Coyle, J., Masson, M., Thanacoody, R.H., Gray, A.J., Webb, D.J., Moggs, J.G., Bateman, D.N., Goldring, C.E. and Park, B.K. (2013) Mechanistic biomarkers provide early and sensitive detection of acetaminophen-induced acute liver injury at first presentation to hospital. Hepatology, 58, 777–787.
Howell, L.S., Ireland, L., Park, B.K. and Goldring, C.E. (2018) MiR-122 and other microRNAs as potential circulating biomarkers of drug-induced liver injury. Exp. Rev. Mol. Diagn., 18, 47–54.
Sato, S., Ichihara, A., Jinnin, M., Izuno, Y., Fukushima, S. and Ihn, H. (2015) Serum miR-124 up-regulation as a disease marker of toxic epidermal necrolysis. Eur. J. Dermatol., 25, 457–462.
Ichihara, A., Wang, Z., Jinnin, M., Izuno, Y., Shimozono, N., Yamane, K., Fujisawa, A., Moriya, C., Fukushima, S., Inoue, Y., Shimozono, N., Yamane, K., Fujisawa, A., Moriya, C., Fukushima, S., Inoue, Y. and Ihn, H. (2014) Upregulation of miR-18a-5p contributes to epidermal necrolysis in severe drug eruptions. J. Allergy Clin. Immunol., 133, 1065–1074.
Williams, S.A., Murthy, A.C., DeLisle, R.K., Hyde, C., Malarstig, A., Ostroff, R., Weiss, S.J., Segal, M.R. and Ganz, P. (2018) Improving assessment of drug safety through proteomics: early detection and mechanistic characterization of the unforeseen harmful effects of torcetrapib. Circulation, 137, 999–1010.
Zhou, H., Gao, M. and Skolnick, J. (2015) Comprehensive prediction of drug-protein interactions and side effects for the human proteome. Sci. Rep., 5, 11090.
Huang, L.C., Wu, X. and Chen, J.Y. (2013) Predicting adverse drug reaction profiles by integrating protein interaction networks with drug structures. Proteomics, 13, 313–324.
Ge, F. and He, Q.Y. (2009) Genomic and proteomic approaches for predicting toxicity and adverse drug reactions. Exp. Opin. Drug Metab. Toxicol., 5, 29–37.
Wilkins, M.R. (2002) What do we want from proteomics in the detection and avoidance of adverse drug reactions. Toxicol. Lett., 127, 245–249.
Amadoz, A., Sebastian-Leon, P., Vidal, E., Salavert, F. and Dopazo, J. (2015) Using activation status of signaling pathways as mechanism-based biomarkers to predict drug sensitivity. Sci. Rep., 5, 18494.
Frueh, F.W. (2010) Real-world clinical effectiveness, regulatory transparency and payer coverage: three ingredients for translating pharmacogenomics into clinical practice. Pharmacogenomics, 11, 657–660.
Ventola, C.L. (2011) Pharmacogenomics in clinical practice: reality and expectations. P T, 36, 412–450.
Madian, A.G., Wheeler, H.E., Jones, R.B. and Dolan, M.E. (2012) Relating human genetic variation to variation in drug responses. Trends Genet., 28, 487–495.
Ventola, C.L. (2013) Role of pharmacogenomic biomarkers in predicting and improving drug response: part 1: the clinical significance of pharmacogenetic variants. P T, 38, 545–560.
Trent, R.J. (2010) Pathology practice and pharmacogenomics. Pharmacogenomics, 11, 105–111.
Ma, J.D., Lee, K.C. and Kuo, G.M. (2012) Clinical application of pharmacogenomics. J. Pharm. Pract., 25, 417–427.
Squassina, A., Manchia, M., Manolopoulos, V.G., Artac, M., Lappa-Manakou, C., Karkabouna, S., Mitropoulos, K., Del Zompo, M. and Patrinos, G.P. (2010) Realities and expectations of pharmacogenomics and personalized medicine: impact of translating genetic knowledge into clinical practice. Pharmacogenomics, 11, 1149–1167.
Vogenberg, F.R., Isaacson Barash, C. and Pursel, M. (2010) Personalized medicine: part 1: evolution and development into theranostics. P T, 35, 560–576.
Gervasini, G., Benitez, J. and Carrillo, J.A. (2010) Pharmacogenetic testing and therapeutic drug monitoring are complementary tools for optimal individualization of drug therapy. Eur. J. Clin. Pharmacol., 66, 755–774.
Link, E., Parish, S., Armitage, J., Bowman, L., Heath, S., Matsuda, F., Gut, I., Lathrop, M. and Collins, R. (2008) SLCO1B1 variants and statin-induced myopathy—a genomewide study. N. Engl. J. Med., 359, 789–799.
Flockhart, D.A., O’Kane, D., Williams, M.S., Watson, M.S., Flockhart, D.A., Gage, B., Gandolfi, R., King, R., Lyon, E., Nussbaum, R., O’Kane, D., Schulman, K., Veenstra, D., Williams, M.S., Watson, M.S.; ACMG Working Group on Pharmacogenetic Testing of CYP2C9, VKORC1 Alleles for Warfarin Use (2008) Pharmacogenetic testing of CYP2C9 and VKORC1 alleles for warfarin. Genet. Med., 10, 139–150.
Scott, S.A. (2011) Personalizing medicine with clinical pharmacogenetics. Genet. Med., 13, 987–995.
Taneja, I., Reddy, B. and Damhorst, G. (2017) Combining biomarkers with EMR data to identify patients in different phases of sepsis. Sci. Rep., 7, 10800.
Crews, K.R., Hicks, J.K., Pui, C.H., Relling, M.V. and Evans, W.E. (2012) Pharmacogenomics and individualized medicine: translating science into practice. Clin. Pharmacol. Ther., 92, 467–475.
Jiang, X.Y., Zhang, Q., Chen, P., Li, S.Y., Zhang, N.N., Chen, X.D., Wang, G.C., Wang, H.B., Zhuang, M.Q. and Lu, M. (2012) CYP7A1 polymorphism influences the LDL cholesterol-lowering response to atorvastatin. J. Clin. Pharm. Ther., 37, 719–723.
Zhu, M., Qiu, S., Zhang, X., Wang, Y., Souraka, T.D.M., Wen, X., Liang, C. and Tu, J. (2018) The associations between CYP24A1 polymorphisms and cancer susceptibility: a meta-analysis and trial sequential analysis. Pathol. Res. Pract., 214, 53–63.
Author information
Authors and Affiliations
Corresponding author
Additional information
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Rights and permissions
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Choi, Y.H., Han, C.Y., Kim, K.S. et al. Future Directions of Pharmacovigilance Studies Using Electronic Medical Recording and Human Genetic Databases. Toxicol Res. 35, 319–330 (2019). https://doi.org/10.5487/TR.2019.35.4.319
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2019.35.4.319