Abstract
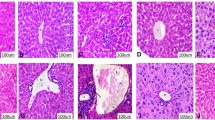
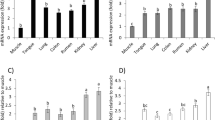
Early life exposure to aflatoxin B1 (AFB1) and low protein diet through complementary foods during weaning is common in parts of Africa and Asia. This study evaluated the effect of co-exposure to AFB1 and low protein diet on the extrahepatic tissues of rats. Twenty-four three-week old weanling male albino rats were used for this study and were randomly assigned into four groups: group 1 served as control and was fed normal protein diet (20% protein), group 2 was fed low protein diet (5% protein), group 3 was fed normal protein diet + 40 ppb AFB1 while group 4 received low protein diet + 40 ppb AFB1, all for eight weeks. Afterward, biomarkers of anemia (packed cell volume (PCV), hemoglobin) and kidney function (urea, uric acid, and creatinine) were determined in the blood while biomarkers of oxidative stress were determined in the tissues spectrophotometrically. Co-exposure to AFB1 and low protein diet significantly (p < 0.05) decreased body weight gain and PCV, increased biomarkers of kidney functions and induced oxidative stress in the tissues studied. There was significant (p < 0.05) reduction in glutathione concentration while TBARS was significantly increased in the tissues. Co-exposure to AFB1 and low protein diet had additive effects on decreasing the weight gain and potentiation effect of kidney dysfunction in the rats. The co-exposure also decreased antioxidant enzymes and increased oxidant status in the tissues. Our results demonstrate that this co-exposure has deleterious health effects on extrahepatic tissues and should be a public health concern especially in developing countries where AFB1 contamination is common.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
REFERENCES
Pitt, J., Taniwaki, M.H. and Cole, M. (2013) Mycotoxin production in major crops as influenced by growing, harvesting, storage and processing, with emphasis on the achievement of Food Safety Objectives. Food Control, 32, 205–215.
Mitchell, N.J., Hsu, H.-H., Chandyo, R.K., Shrestha, B., Bodhidatta, L., Tu, Y.-K., Gong, Y.-Y., Egner, P.A., Ulak, M. and Groopman, J.D. (2017) Aflatoxin exposure during the first 36 months of life was not associated with impaired growth in Nepalese children: an extension of the MAL-ED study. PLoS ONE, 12, e0172124.
Cho, S.Y., Kang, S.J., Jung, J., Jeong, B.O. and Jeong, C.S. (2009) Co-contamination of aflatoxins with ochratoxin A and zearalenone in Thuja orientalis Semen. Toxicol. Res., 25, 125–131.
Watson, S., Gong, Y.Y. and Routledge, M. (2017) Interventions targeting child undernutrition in developing countries may be undermined by dietary exposure to aflatoxin. Crit. Rev. Food Sci. Nutr., 57, 1963–1975.
Dohnal, V., Wu, Q. and Kuča, K. (2014) Metabolism of aflatoxins: key enzymes and interindividual as well as interspecies differences. Arch. Toxicol., 88, 1635–1644.
Marin, D.E. and Taranu, I. (2012) Overview on aflatoxins and oxidative stress. Toxin Rev., 31, 32–43.
Rotimi, O.A., Rotimi, S.O., Oluwafemi, F., Ademuyiwa, O. and Balogun, E.A. (2016) Coexistence of aflatoxicosis with protein malnutrition worsens hepatic oxidative damage in rats. J. Biochem. Mol. Toxicol., 30, 269–276.
Turner, P.C., Moore, S.E., Hall, A.J., Prentice, A.M. and Wild, C.P. (2003) Modification of immune function through exposure to dietary aflatoxin in Gambian children. Environ. Health Perspect., 111, 217.
Khlangwiset, P., Shephard, G.S. and Wu, F. (2011) Aflatoxins and growth impairment: a review. Crit. Rev. Toxicol., 41, 740–755.
Turner, P.C., Collinson, A.C., Cheung, Y.B., Gong, Y., Hall, A.J., Prentice, A.M. and Wild, C.P. (2007) Aflatoxin exposure in utero causes growth faltering in Gambian infants. Int. J. Epidemiol., 36, 1119–1125.
Gong, Y., Egal, S., Hounsa, A., Turner, P., Hall, A., Cardwell, K. and Wild, C. (2003) Determinants of aflatoxin exposure in young children from Benin and Togo, West Africa: the critical role of weaning. Int. J. Epidemiol., 32, 556–562.
Gong, Y., Hounsa, A., Egal, S., Turner, P.C., Sutcliffe, A.E., Hall, A.J., Cardwell, K. and Wild, C.P. (2004) Postweaning exposure to aflatoxin results in impaired child growth: a longitudinal study in Benin, West Africa. Environ. Health Perspect., 112, 1334.
Chen, G., Gong, Y.Y., Kimanya, M.E., Shirima, C.P. and Routledge, M.N. (2017) Comparison of urinary aflatoxin M1 and aflatoxin albumin adducts as biomarkers for assessing aflatoxin exposure in Tanzanian children. Biomarkers, 23, 131–136.
Routledge, M.N., Kimanya, M.E., Shirima, C.P., Wild, C.P. and Gong, Y.Y. (2014) Quantitative correlation of aflatoxin biomarker with dietary intake of aflatoxin in Tanzanian children. Biomarkers, 19, 430–435.
Adetunji, M.C., Atanda, O.O. and Ezekiel, C.N. (2017) Risk assessment of mycotoxins in stored maize grains consumed by infants and young children in Nigeria. Children, 4, 58.
Müller, O. and Krawinkel, M. (2005) Malnutrition and health in developing countries. Can. Med. Assoc. J., 173, 279–286.
Grover, Z. and Ee, L.C. (2009) Protein energy malnutrition. Pediatr. Clin. North Am., 56, 1055–1068.
Khare, M., Mohanty, C., Das, B., Jyoti, A., Mukhopadhyay, B. and Mishra, S. (2014) Free radicals and antioxidant status in protein energy malnutrition. Int. J. Pediatr., 2014, 254396.
Ge, J., Yu, H., Li, J., Lian, Z., Zhang, H., Fang, H. and Qian, L. (2017) Assessment of aflatoxin B1 myocardial toxicity in rats: mitochondrial damage and cellular apoptosis in cardiomyocytes induced by aflatoxin B1. J. Int. Med. Res., 45, 1015–1023.
Abdulmajeed, N.A. (2011) Therapeutic ability of some plant extracts on aflatoxin B1 induced renal and cardiac damage. Arab. J. Chem., 4, 1–10.
Linardaki, Z.I., Lamari, F.N. and Margarity, M. (2017) Saffron (Crocus sativus L.) tea intake prevents learning/memory defects and neurobiochemical alterations induced by aflatoxin B1 exposure in adult mice. Neurochem. Res., 42, 2743–2754.
Yang, X.-J., Lu, H.-Y., Li, Z.-Y., Bian, Q., Qiu, L.-L., Li, Z., Liu, Q., Li, J., Wang, X. and Wang, S.-L. (2012) Cyto-chrome P450 2A13 mediates aflatoxin B1-induced cytotoxicity and apoptosis in human bronchial epithelial cells. Toxicology, 300, 138–148.
Chen, J., Chen, K., Yuan, S., Peng, X., Fang, J., Wang, F., Cui, H., Chen, Z., Yuan, J. and Geng, Y. (2016) Effects of aflatoxin B1 on oxidative stress markers and apoptosis of spleens in broilers. Toxicol. Ind. Health, 32, 278–284.
Trebak, F., Alaoui, A., Alexandre, D., El Ouezzani, S., Anouar, Y., Chartrel, N. and Magoul, R. (2015) Impact of aflatoxin B1 on hypothalamic neuropeptides regulating feeding behavior. Neurotoxicology, 49, 165–173.
Sari, M., Pee, S.D., Martini, E., Herman, S., Bloem, M.W. and Yip, R. (2001) Estimating the prevalence of anaemia: a comparison of three methods. Bull. World Health Organ., 79, 506–511.
Lowry, O.H., Rosebrough, N.J., Farr, A.L. and Randall, R.J. (1951) Protein measurement with the Folin phenol reagent. J. Biol. Chem., 193, 265–275.
Buege, J.A. and Aust, S.D. (1978) Microsomal lipid peroxidation. Methods Enzymol., 52, 302–310.
Ellman, G.L. (1959) Tissue sulfhydryl groups. Arch. Biochem. Biophys., 82, 70–77.
Habig, W.H., Pabst, M.J. and Jakoby, W.B. (1974) Glutathione S-transferases the first enzymatic step in mercapturic acid formation. J. Biol. Chem., 249, 7130–7139.
Rotruck, J.T., Pope, A.L., Ganther, H., Swanson, A., Hafeman, D.G. and Hoekstra, W. (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science, 179, 588–590.
Marklund, S. and Marklund, G. (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. FEBS J., 47, 469–474.
Fergusson, R. and Chance, B. (1955) Substrate specificity of peroxidase. Science, 122, 466–467.
Kanbur, M., Eraslan, G., Sarıca, Z.S. and Aslan, Ö. (2011) The effects of evening primrose oil on lipid peroxidation induced by subacute aflatoxin exposure in mice. Food Chem. Toxicol., 49, 1960–1964.
De Onis, M., Onyango, A.W., Borghi, E., Garza, C. and Yang, H. (2006) Comparison of the World Health Organization (WHO) Child Growth Standards and the National Center for Health Statistics/WHO international growth reference: implications for child health programmes. Public Health Nutr., 9, 942–947.
Huang, C.-J. and Fwu, M.-L. (1993) Degree of protein deficiency affects the extent of the depression of the antioxidative enzyme activities and the enhancement of tissue lipid peroxidation in rats. J. Nutr., 123, 803–810.
Alaverdashvili, M., Hackett, M.J., Caine, S. and Paterson, P.G. (2017) Parallel changes in cortical neuron biochemistry and motor function in protein-energy malnourished adult rats. NeuroImage, 149, 275–284.
Castelino, J.M., Routledge, M.N., Wilson, S., Dunne, D.W., Mwatha, J.K., Gachuhi, K., Wild, C.P. and Gong, Y.Y. (2015) Aflatoxin exposure is inversely associated with IGF1 and IGFBP3 levels in vitro and in Kenyan schoolchildren. Mol. Nutr. Food Res., 59, 574–581.
Smith, L.E., Stoltzfus, R.J. and Prendergast, A. (2012) Food chain mycotoxin exposure, gut health, and impaired growth: a conceptual framework. Adv. Nutr., 3, 526–531.
El-Nekeety, A.A., Mohamed, S.R., Hathout, A.S., Hassan, N.S., Aly, S.E. and Abdel-Wahhab, M.A. (2011) Antioxidant properties of Thymus vulgaris oil against aflatoxininduce oxidative stress in male rats. Toxicon, 57, 984–991.
Eraslan, G., Sarıca, Z.S., Bayram, L.Ç., Tekeli, M.Y., Kanbur, M. and Karabacak, M. (2017) The effects of diosmin on aflatoxin-induced liver and kidney damage. Environ. Sci. Pollut. Res. Int., 24, 27931–27941.
Attia, Y., Allakany, H., Abd Al Hamid, A., Al Saffar, A., Hassan, R. and Mohamed, N. (2013) Capability of different non nutritive feed additives on improving productive and physiological traits of broiler chicks fed diets with or without aflatoxin during the first 3 weeks of life. J. Anim. Physiol. Anim. Nutr. (Berl.), 97, 754–772.
Yousef, M., Salem, M., Kamel, K., Hassan, G. and El Nouty, F. (2003) Influence of ascorbic acid supplementation on the haematological and clinical biochemistry parameters of male rabbits exposed to aflatoxin B1. J. Environ. Sci. Health B, 38, 193–209.
Mebius, R.E. and Kraal, G. (2005) Structure and function of the spleen. Nat. Rev. Immunol., 5, 606–616.
Peng, X., Bai, S., Ding, X., Zeng, Q., Zhang, K. and Fang, J. (2015) Pathological changes in the immune organs of broiler chickens fed on corn naturally contaminated with aflatoxins B1 and B2. Avian Pathol., 44, 192–199.
Monson, M.S., Settlage, R.E., Mendoza, K.M., Rawal, S., El-Nezami, H.S., Coulombe, R.A. and Reed, K.M. (2015) Modulation of the spleen transcriptome in domestic turkey (Meleagris gallopavo) in response to aflatoxin B1 and probiotics. Immunogenetics, 67, 163–178.
Mello, A.S., de Oliveira, D.C., Bizzarro, B., Sá-Nunes, A., Hastreiter, A.A., de Oliveira Beltran, J.S., Xavier, J.G., Borelli, P. and Fock, R.A. (2014) Protein malnutrition alters spleen cell proliferation and IL-2 and IL-10 production by affecting the STAT-1 and STAT-3 balance. Inflammation, 37, 2125–2138.
Qureshi, H., Hamid, S.S., Ali, S.S., Anwar, J., Siddiqui, A.A. and Khan, N.A. (2015) Cytotoxic effects of aflatoxin B1 on human brain microvascular endothelial cells of the blood-brain barrier. Med. Mycol., 53, 409–416.
Souto, N.S., Claudia Monteiro Braga, A., Lutchemeyer de Freitas, M., Rechia Fighera, M., Royes, L.F.F., Schneider Oliveira, M. and Furian, A.F. (2017) Aflatoxin B1 reduces non-enzymatic antioxidant defenses and increases protein kinase C activation in the cerebral cortex of young rats. Nutr. Neurosci., 21, 268–275.
Feoli, A.M., Siqueira, I.R., Almeida, L., Tramontina, A.C., Vanzella, C., Sbaraini, S., Schweigert, I.D., Netto, C.A., Perry, M.L. and Gonçalves, C.A. (2006) Effects of protein malnutrition on oxidative status in rat brain. Nutrition, 22, 160–165.
Adebayo, O.L., Adenuga, G.A. and Sandhir, R. (2016) Selenium and zinc protect brain mitochondrial antioxidants and electron transport chain enzymes following postnatal protein malnutrition. Life Sci., 152, 145–155.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Rotimi, O.A., Rotimi, S.O., Oluwafemi, F. et al. Oxidative Stress in Extrahepatic Tissues of Rats Co-Exposed to Aflatoxin B1 and Low Protein Diet. Toxicol Res. 34, 211–220 (2018). https://doi.org/10.5487/TR.2018.34.3.211
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2018.34.3.211