Abstract
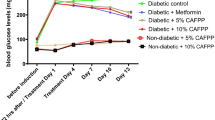
Anti-diabetes activity of Catharsius molossus (Ca, a type of dung beetle) glycosaminoglycan (G) was evaluated to reduce glucose, creatinine kinase, triglyceride and free fatty acid levels in db mice. Diabetic mice in six groups were administrated intraperitoneally: Db heterozygous (Normal), Db homozygous (CON), Heuchys sanguinea glycosaminoglycan (HEG, 5 mg/kg), dung beetle glycosaminoglycan (CaG, 5 mg/kg), bumblebee (Bombus ignitus) queen glycosaminoglycan (IQG, 5 mg/kg) and metformin (10 mg/kg), for 1 month. Biochemical analyses in the serum were evaluated to determine their anti-diabetic and anti-inflammatory actions in db mice after 1 month treatment with HEG, CaG or IQG treatments. Blood glucose level was decreased by treatment with CaG. CaG produced significant anti-diabetic actions by inhiting creatinine kinase and alkaline phosphatase levels. As diabetic parameters, serum glucose level, total cholesterol and triglyceride were significantly decreased in CaG5-treated group compared to the controls. Dung beetle glycosaminoglycan, compared to the control, could be a potential therapeutic agent with anti-diabetic activity in diabetic mice. CaG5-treated group, compared to the control, showed the up-regulation of 48 genes including mitochondrial yen coded tRNA lysine (mt-TK), cytochrome P450, family 8/2, subfamily b, polypeptide 1 (Cyp8b1), and down-regulation of 79 genes including S100 calcium binding protein A9 (S100a9) and immunoglobulin kappa chain complex (Igk), and 3-hydroxy-3-methylglutaryl-CoenzymeAsynthasel (Hmgcs1). Moreover, mitochondrial thymidine kinase (mt-TK), was up-regulated, and calgranulin A (S100a9) were down-regulated by CaG5 treatment, indicating a potential therapeutic use for anti-diabetic agent.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Change history
01 October 2018
On page 158 and 159, the authors found labeling errors in Table 4 and Table 5. Table 4 is originally result for “Upregulated genes differentially expressed in liver tissue of db mice treated with GAG over a 1-month period” but was erroneously expressed as “Upregulated genes differentially expressed in liver tissue of melanoma induced mice treated with GAG over a 1-month period”. Table 5 is originally result for “Downregulated genes differentially expressed in liver tissue of db mice treated with GAG over a 1-month period” but was erroneously expressed as “Downregulated genes differentially expressed in liver tissue of melanoma induced mice treated with GAG over a 1-month period”. Therefore author wanted to change from --melanoma induced mice-- of Table 4 and Table 5 to-- db mice--.
Table 4. Upregulated genes differentially expressed in liver tissue of db mice treated with GAG over a 1-month period
Table 5. Downregulated genes differentially expressed in liver tissue of db mice treated with GAG over a 1-month period
References
Yan, W., Zhou, B., Shen, Y. and Xu, G. (2015) Antioxidant and antithrombotic therapies for diabetic kidney disease. Iran. J. Kidney Dis., 9, 413–420.
Lepedda, A.J., Muro, P.D., Capobianco, G. and Formato, M. (2017) Significance of urinary glycosaminoglycans/proteoglycans in the evaluation of type 1 and type 2 diabetes complications. J. Diabetes Complicat., 31, 149–155.
Kubaski, F., Osago, H., Mason, R.W., Yamaguchi, S., Kobayashi, H., Tsuchiya, M., Orii, T. and Tomatsu, S. (2017) Glycosaminoglycans detection methods: application of mass spectrometry. Mol. Genet. Metab., 120, 67–77.
Li, R., Xing, J., Mu, X., Wang, H., Zhang, L., Zhao, Y. and Zhang, Y. (2015) Sulodexide therapy for the treatment of diabetic nephropathy, a meta-analysis and literature review. Drug Des. Devel. Ther., 9, 6275–6283.
Ahn, M.Y., Kim, B.J., Kim, H.J., Yoon, H.J., Jee, S.D., Hwang, J.S. and Park, K.K. (2017). Anti-obesity effect of Bombus ignitus queen glycosaminoglycans in rats on a high-fat diet. Int. J. Mol. Sci., 18, E681.
Ahn, M.Y., Kim, B.J., Kim, H.J., Hwang, J.S., Jung, Y.S. and Park, K.K. (2017) Anti-aging effect and gene expression profiling of dung beetle glycosaminoglycan in aged rats. Biomater. Res., 21, 5.
Xia, L., Bai, L., Yi, L., Liu, B., Chu, C., Liang, Z., Li, P., Jiang, Z. and Zhao, Z. (2007) Authentication of the 31 species of toxic and potent chinese materia medica (T/PCMM) by microscopic technique, part 1: three kinds of toxic and potent animal CMM. Microsc. Res. Tech., 70, 960–968.
Kim, Y.S., Jo, Y.Y., Chang, I.M., Toida, T., Park, Y. and Linhardt, R.J. (1996) A new glycosaminoglycan from the giant african snail Achatina fulica. J. Biol. Chem. 271, 11750–11755.
Fratz, E.J., Hunter, G.A. and Ferreira, G.C. (2014) Expression of murine 5-aminolevulinate synthase variants causes protoporphyrin IX accumulation and light-induced mammalian cell death. PLoS ONE, 9, e93078.
Song, J., Liu, H., Ressom, H.W., Tiwari, S. and Ecelbarger, C.M. (2008) Chronic Rosigiltazone therapy normalizes expression of ACE1, SCD1 and other genes in the kidney of obese Zucker rats as determined by microarray by microarray analysis. Exp. Clin. Endocrinol. Diabetes, 116 , 315–325.
Wang, F., Wan, A. and Rodrigues, B. (2013) The function of heparanase in diabetes and its complications. Can. J. Diabetes, 37, 332–338.
Ahn, M.Y., Hwang, J.S., Yoon, H.J. and Yun, E.Y. (2012) Anti-diabetic composition comprising glycosaminoglycans derived insect. Korean patient 10-120419.
Heishi, M., Ichihara, J., Teramoto, R., Itakura, Y., Hayashi, K., Ishikawa, H., Gomi, H., Sakai, J., Kanaoka, M., Taiji, M. and Kimura, T. (2006) Global gene expression analysis in liver of obese diabetic db/db mice treated with metformin. Diabetologia, 49, 1647–1655.
Dou, J., Ma, J., Liu J., Wang, C., Johnsson, E., Yao, H., Zhao, J. and Pan, C. (2018) Efficacy and safety of saxagliptin in combination with metformin as initial therapy in Chinese patients with type 2 diabetes: results from the START study, a multicenter, randomized, double-blind, active-controlled, phase 3 trial. Diabetes Obes. Metab., 20, 590–598.
Mata, M.D.I., Garrido-Maraver, J., Cotán, D., Cordero, M.D., Oropesa-Ávila, M., Izquierdo, L.G., Miguel, M.D., Lorite, J.B., Infante, E.R., Ybot, P., Jackson, S. and Sanchez-Alcázar, J.A. (2012) Recovery of MERRF fibroblasts and cybrids pathophysiology by Coenzyme Q10. Neurothera-peutics, 9, 446–463.
Ishida, H., Yamashita, C., Kuruta, Y., Yoshida, Y. and Noshiro, M. (2000) Insulin is a dominant suppressor of sterol 12a-hydroxylase P450 (CYP8B) expression in rat liver possible role of insulin in Circadian rhythm of CYP8B. J. Biochem., 127, 57–64.
Kaur, A., Patankar, J.V., de Haan, W., Ruddle, P., Wijesekara, N., Groen, A.K., Verchere, C.B., Singaraja, R.R. and Hayden, M.R. (2015) Loss of cyp8b1 improves glucose homeostasis by increasing GLP-1. Diabetes, 64, 1168–1179.
Mortensen, O.H., Nielsen, A.R., Erikstrup, C., Plomgaard, P., Fischer, C.P., Krogh-Madsen, R., Lindegaard, B., Petersen, A.M., Taudorf, S. and Pedersen, B.K. (2009) Calprotectin - a novel marker of obesity. PLoS ONE, 4, e7419.
Kraakman, M.J., Lee, M.K.S., Al-Sharea, A., Dragoljevic, D., Barrett, T.J., Montenont, E., Basu, D., Heywood, S., Kammoun, H.L., Flynn, M., Whillas, A., Hanssen, N.M.J., Febbraio, M.A., Westein, E., Fisher, E.A., Chin-Dusting, J., Cooper, M.E., Berger, J.S., Goldberg, I.J., Nagareddy, P.R. and Murphy, A.J. (2017) Neutrophil-derived S100 calcium-binding proteins A8/A9 promote reticulated thrombocytosis and atherogenesis in diabetes. J. Clin. Invest., 127, 2133–2147.
Giudicelli, V., Chaume, D. and Lefranc, M. (2005) IMGT/ GENE-DB: a comprehensive database for human and mouse immunoglobulin and T cell receptor genes. Nucleic Acids Res., 33, D256–D261.
Zhou, H. and Rigoutsos, I. (2014) The emerging roles of GPRC5A in diseases. Oncoscience, 1, 765–776.
Tanaka, T., Kubota, M., Shinohara, K., Yasuda, K. and Kato, J. (2003) In vivo analysis of the cyclin D1 promotor during early embryogenesis in Xenopus. Cell Struct. Funct., 28, 165–177.
Chowdhury, K.H., Montgomery, M.K., Morris, M.J., Cognard, E., Shepherd, R.R. and Smith, G.C. (2015) Glucagon phosphorylate serine 552 of beta-catenin leading to increased expression of cyclinD1 and C-Myc in isolated rat liver. Arch. Physiol. Biochem., 121, 88–96.
Lauver, D.A. and Lucchesi, B.R. (2006) Sulodexide: a renewed interest in this glycosaminoglycan. Cardiovasc. Drug Rev., 24, 214–226.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Ahn, M.Y., Kim, B.J., Yoon, H.J. et al. Anti-Diabetic Effects of Dung Beetle Glycosaminoglycan on db Mice and Gene Expression Profiling. Toxicol Res. 34, 151–162 (2018). https://doi.org/10.5487/TR.2018.34.2.151
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2018.34.2.151