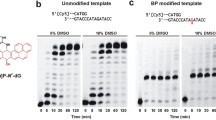
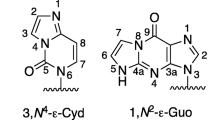
Abstract
Aryl hydrocarbons such as 3-nitrobenzanthrone (NBA), 4-aminobiphenyl (ABP), acetylaminofluorene (AAF), benzo(a)pyrene (BaP), and 1-nitropyrene (NP) form bulky DNA adducts when absorbed by mammalian cells. These chemicals are metabolically activated to reactive forms in mammalian cells and preferentially get attached covalently to the N2 or C8 positions of guanine or the N6 position of adenine. The proportion of N2 and C8 guanine adducts in DNA differs among chemicals. Although these adducts block DNA replication, cells have a mechanism allowing to continue replication by bypassing these adducts: translesion DNA synthesis (TLS). TLS is performed by translesion DNA polymerases—Pol η, Κ, ι, and ζ and Rev1—in an error-free or error-prone manner. Regarding the NBA adducts, namely, 2-(2′-deoxyguanosin-N2-yl)-3-aminobenzanthrone (dG-N2-ABA) and N-(2′ -deoxyguanosin-8-yl)-3-aminobenzanthrone (dG-C8-ABA), dG-N2-ABA is produced more often than dG-C8-ABA, whereas dG-C8-ABA blocks DNA replication more strongly than dG-N2-ABA. dG-N2-ABA allows for a less error-prone bypass than dG-C8-ABA does. Pol η and Κ are stronger contributors to TLS over dG-C8-ABA, and Pol Κ bypasses dG-C8-ABA in an error-prone manner. TLS efficiency and error-proneness are affected by the sequences surrounding the adduct, as demonstrated in our previous study on an ABP adduct, N-(2′ -deoxyguanosine-8-yl)-4-aminobiphenyl (dG-C8-ABP). Elucidation of the general mechanisms determining efficiency, error-proneness, and the polymerases involved in TLS over various adducts is the next step in the research on TLS. These TLS studies will clarify the mechanisms underlying aryl hydrocarbon mutagenesis and carcinogenesis in more detail.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- AAF:
-
acetylaminofluorene
- ABP:
-
4-aminobiphenyl
- AP:
-
apurinic/apyrimidinic
- BaP:
-
benzo(a)pyrene
- BER:
-
base excision repair
- BPDE:
-
(±)-anti-benzo[a]pyrene-7,8-diol-9,10-epoxide
- IPTG:
-
isopropyl-β-D-thiogalactopyranoside
- IQ:
-
2-amino-3-methylimidazo-[4,5-f]quinoline
- N-Aco-ABA:
-
N-acetoxy-3-aminobenzanthrone
- dG-C8-ABP:
-
N-(2′-deoxyguanosine-8-yl)-4-aminobiphenyl
- dG-N2-ABA:
-
2-(2′-deoxyguanosin-N2-yl)-3-aminobenzanthrone
- dG-C8-ABA:
-
N-(2′-deoxyguanosin-8-yl)-3-aminobenzanthrone
- NBA:
-
3-nitrobenzanthrone
- NER:
-
nucleotide excision repair
- NP:
-
1-nitropyrene
- TLS:
-
translesion DNA synthesis
- X-gal:
-
5-bromo-4-chloro-3-indolyl-β-D-galactopyranoside
- XPA:
-
xeroderma pigmentosum complementation group A
References
Choi, H., Harrison, R., Komulainen, H. and Saborit, J.M.D. (2010) Polycyclic aromatic hydrocarbons in WHO Guidelines for Indoor Air Quality: Selected Pollutants. World Health Organization, Geneva, pp. 299–346.
Baird, W.M., Hooven, L.A. and Mahadevan, B. (2005) Carcinogenic polycyclic aromatic hydrocarbon-DNA adducts and mechanism of action. Environ. Mol. Mutagen., 45, 106–114.
Alexandrov, K., Rojas, M. and Satarug, S. (2010) The critical DNA damage by benzo(a)pyrene in lung tissues of smokers and approaches to preventing its formation. Toxicol. Lett., 198, 63–68.
Shiizaki, K., Kawanishi, M. and Yagi, T. (2017) Modulation of benzo[a]pyrene-DNA adduct formation by CYP1 inducer and inhibitor. Genes Environ., 39, 14.
Takamura-Enya, T., Suzuki, H. and Hisamatsu, Y. (2006) Mutagenic activities and physicochemical properties of selected nitrobenzanthrones. Mutagenesis, 21, 399–404.
Arlt, V.M., Schmeiser, H.H., Osborne, M.R., Kawanishi, M., Kanno, T., Yagi, T., Phillips, D.H. and Takamura-Enya, T. (2006) Identification of three major DNA adducts formed by the carcinogenic air pollutant 3-nitrobenzanthrone in rat lung at the C8 and N2 position of guanine and at the N6 position of adenine. Int. J. Cancer, 118, 2139–2146.
Takamura-Enya, T., Kawanishi, M., Yagi, T. and Hisamatsu, Y. (2007) Structural identification of DNA adducts derived from 3-nitrobenzanthrone, a potent carcinogen present in the atmosphere. Chem. Asian J., 2, 1174–1185.
Kanno, T., Kawanishi, M., Takamura-Enya. T., Arlt, V.M., Phillips, D.H. and Yagi, T. (2007) DNA adduct formation in human hepatoma cells treated with 3-nitrobenzanthrone: analysis by the 32P-postlabeling method. Mutat. Res., 634, 184–191.
Kawanishi, M., Fujikawa, Y., Ishii, H., Nishida, H., Higashigaki, Y., Kanno, T., Matsuda, T., Takamura-Enya, T. and Yagi, T. (2013) Adduct formation and repair, and translesion DNA synthesis across the adducts in human cells exposed to 3-nitrobenzanthrone. Mutat. Res., 753, 93–100.
Nagy, E., Zeisig, M., Kawamura, K., Hisamatsu, Y., Sugeta, A., Adachi, S. and Möller, L. (2005) DNA adduct and tumor formations in rats after intratracheal administration of the urban air pollutant 3-nitrobenzanthrone. Carcinogenesis, 26, 1821–1828.
Cohen, S.M., Boobis, A.R., Meek, M.E., Preston, R.J. and McGregor, D.B. (2006) 4-Aminobiphenyl and DNA reactivity: case study within the context of the 2006 IPCS Human Relevance Framework for Analysis of a cancer mode of action for humans. Crit. Rev. Toxicol., 36, 803–819.
IARC (1987) 4-Aminobiphenyl in Overall Evaluations of Carcinogenicity. IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Humans (Suppl. 7). International Agency for Research on Cancer, Lyon, France, pp. 91–92.
Besaratinia, A. and Tommasi, S. (2013) Genotoxicity of tobacco smoke-derived aromatic amines and bladder cancer: current state of knowledge and future research directions. FASEB J., 27, 2090–2100.
Feng, Z., Hu, W., Rom, W.N., Beland, F.A. and Tang, M.S. (2002) 4-aminobiphenyl is a major etiological agent of human bladder cancer: evidence from its DNA binding spectrum in human p53 gene. Carcinogenesis, 23, 1721–1727.
Fuss, J.O. and Cooper, P.K. (2006) DNA repair: dynamic defenders against cancer and aging. PLoS Biol., 4, e203.
Spivak, G. (2015) Nucleotide excision repair in humans. DNA Repair (Amst.), 36, 13–18.
Bauer, N.C., Corbett, A.H. and Doetsch, P.W. (2015) The current state of eukaryotic DNA base damage and repair. Nucleic Acids Res., 43, 10083–10101.
Carter, R.J. and Parsons, J.L. (2016) Base excision repair, a pathway regulated by posttranslational modifications. Mol. Cell. Biol., 36, 1426–1437.
Sale, J.E. (2013) Translesion DNA synthesis and mutagenesis in eukaryotes. Cold Spring Harb. Perspect. Biol., 5, a012708.
Basu, A.K., Pande, P. and Bose, A. (2017) Translesion synthesis of 2′-deoxyguanosine lesions by eukaryotic DNA polymerases. Chem. Res. Toxicol., 30, 61–72.
Livneh, Z., Ziv, O. and Shachar, S. (2010) Multiple two-polymerase mechanisms in mammalian translesion DNA synthesis. Cell Cycle, 9, 729–735.
Goodman, M.F. and Woodgate, R. (2013) Translesion DNA polymerases. Cold Spring Harb. Perspect. Biol., 5, a010363.
Kirouac, K.N., Basu, A.K. and Ling, H. (2013) Replication of a carcinogenic nitropyrene DNA lesion by human Y-family DNA polymerase. Nucleic Acids Res., 41, 2060–2071.
Shibutani, S., Suzuki, N. and Grollman, A.P. (2004) Mechanism of frameshift (deletion) generated by acetylaminofluorene-derived DNA adducts in vitro. Biochemistry, 43, 15929–15935.
Schorr, S., Schneider, S., Lammens, K., Hopfner, K.P. and Carell, T. (2010) Mechanism of replication blocking and by pass of Y-family polymerase η by bulky acetylaminofluorene DNA adducts. Proc. Natl. Acad. Sci. U.S.A., 107, 20720–20725.
Suzuki, N., Ohashi, E., Kolbanovskiy, A., Geacintov, N.E., Grollman, A.P., Ohmori. H. and Shibutani, S. (2002) Translesion synthesis by human DNA polymerase K on a DNA template containing a single stereoisomer of dG-(+)- or dG-(−)-anti-N2-BPDE (7,8-dihydroxy-anti-9,10-epoxy-7,8,9,10-tetrahydrobenzo[a]pyrene). Biochemistry, 41, 6100–6106.
Choi, J.Y., Angel, K.C. and Guengerich, F.P. (2006) Translesion synthesis across bulky N2-alkyl guanine DNA adducts by human DNA polymerase K. J. Biol. Chem., 281, 21062–21072.
Frank, E.G., Sayer, J.M., Kroth, H., Ohashi, E., Ohmori, H., Jerina, D.M. and Woodgate, R. (2002) Translesion replication of benzo[a]pyrene and benzo[c]phenanthrene diol epoxide adducts of deoxyadenosine and deoxyguanosine by human DNA polymerase iota. Nucleic Acids Res., 30, 5284–5292.
Chiapperino, D., Kroth, H., Kramarczuk, I.H., Sayer, J.M., Masutani, C., Hanaoka, F., Jerina, D.M. and Cheh, A.M. (2002) Preferential misincorporation of purine nucleotides by human DNA polymerase eta opposite benzo[a]pyrene 7,8-diol 9,10-epoxide deoxyguanosine adducts. J. Biol. Chem., 277, 11765–11771.
Bose, A., Millsap, A.D., DeLeon, A., Rizzo, C.J. and Basu, A.K. (2016) Translesion synthesis of the N2-2′-deoxyguanosine adduct of the dietary mutagen IQ in human cells: Errorfree replication by DNA polymerase K and mutagenic bypass by DNA polymerases η, ζ, and Rev1. Chem. Res. Toxicol., 29, 1549–1559.
Bose, A., Pande, P., Jasti, V.P., Millsap, A.D., Hawkins, E.K., Rizzo, C.J. and Basu, A.K. (2015) DNA polymerases K and ζ cooperatively perform mutagenic translesion synthesis of the C8-2′-deoxyguanosine adduct of the dietary mutagen IQ in human cells. Nucleic Acids Res., 43, 8340–8351.
Tokarsky, E.J., Gadkari, V.V., Zahurancika, W.J., Malik, CK., Basu, A.K. and Suo, Z. (2016) Pre-steady-state kinetic investigation of bypass of a bulky guanine lesion by human Y-family DNA polymerases. DNA Repair (Amst.), 46, 20–28.
Patra, A., Politica, D.A., Chatterjee, A., Tokarsky, E.J., Suo, Z., Basu, A.K., Stone, M.P. and Egli, M. (2016) Mechanism of error-free bypass of the environmental carcinogen N-(2′-deoxyguanosin-8-yl)-3-aminobenzanthrone adduct by human DNA polymerase η. Chembiochem, 17, 2033–2037.
Sherrer, S.M., Sanman, L.E., Xia, C.X., Bolin, E.R., Malik, C.K., Efthimiopoulos, G., Basu, A.K. and Suo, Z. (2012) Kinetic analysis of the bypass of a bulky DNA lesion catalyzed by human Y-family DNA polymerases. Chem. Res. Toxicol., 25, 730–740.
Sherrer, S.M., Taggart, D.J., Pack, L.R., Malik, C.K., Basu, A.K. and Suo, Z. (2012) Quantitative analysis of the mutagenic potential of 1-aminopyrene-DNA adduct bypass catalyzed by Y-family DNA polymerases. Mutat. Res., 737, 25–33.
Maga, G., Villani, G., Crespan, E., Wimmer, U., Ferrari, E., Bertocci, B. and Hübscher, U. (2007) 8-oxo-guanine bypass by human DNA polymerases in the presence of auxiliary proteins. Nature, 447, 606–608.
Moriya, M. (1993) Single-stranded shuttle phagemid for mutagenesis studies in mammalian cells: 8-oxoguanine in DNA induces targeted G.C-->T.A transversions in simian kidney cells. Proc. Natl. Acad. Sci. U.S.A., 90, 1122–1126.
Yang, I.Y., Miller, H., Wang, Z., Frank, E.G., Ohmori, H., Hanaoka, F. and Moriya, M. (2003) Mammalian translesion DNA synthesis across an acrolein-derived deoxyguanosine adduct. Participation of DNA polymerase η in error-prone synthesis in human cells. J. Biol. Chem., 278, 13989–13994.
Hashimoto, K., Bonala, R., Johnson, F., Grollman, A.P. and Moriya, M. (2016) Y-family DNA polymerase-independent gap-filling translesion synthesis across aristolochic acidderived adenine adducts in mouse cells. DNA Repair (Amst.), 46, 55–60.
Pollack, M., Yang, I.Y., Kim, H.Y., Blair, I.A. and Moriya, M. (2006) Translesion DNA Synthesis across the heptanone--etheno-2′-deoxycytidine adduct in cells. Chem. Res. Toxicol., 19, 1074–1079.
Yasui, M., Dong, H., Bonala R.R., Suzuki, N., Ohmori, H., Hanaoka, F., Johnson, F., Grollman, A.P. and Shibutani S. (2004) Mutagenic properties of 3-(deoxyguanosin-N2-yl)-2-acetylaminofluorene, a persistent acetylaminofluorene-derived DNA adduct in mammalian cells. Biochemistry, 43, 15005–15013.
Avkin, S. and Livneh, Z. (2002) Efficiency, specificity and DNA polymerase-dependence of translesion replication across the oxidative DNA lesion 8-oxoguanine in human cells. Mutat. Res., 510, 81–90.
Avkin, S., Goldsmith, M., Velasco-Miguel, S., Geacintov, N., Friedberg, E.C. and Livneh, Z. (2004) Quantitative analysis of translesion DNA synthesis across a benzo[a]pyrene-guanine adduct in mammalian cells: the role of DNA polymerase κ. J. Biol. Chem., 279, 53298–53305.
Watt, D.L., Utzat, C.D., Hilario, P. and Basu, A.K. (2007) Mutagenicity of the 1-nitropyrene-DNA adduct N-(deoxyguanosin-8-yl)-1-aminopyrene in mammalian cells. Chem. Res. Toxicol., 20, 1658–1664.
Tolentino, J.H., Burke, T.J., Mukhopadhyay, S., McGregor, W.G. and Basu, A.K. (2008) Inhibition of DNA replication fork progression and mutagenic potential of 1, N6-ethenoadenine and 8-oxoguanine in human cell extracts. Nucleic Acids Res., 36, 1300–1308.
Pande, P., Malik, C.K., Bose, A., Jasti, V.P. and Basu, A.K. (2014) Mutational analysis of the C8-guanine adduct of the environmental carcinogen 3-nitrobenzanthrone in human cells: critical roles of DNA polymerases η and κ and Rev1 in errorprone translesion synthesis. Biochemistry, 53, 5323–5331.
Veaute, X. and Fuchs, R. (1993) Greater susceptibility to mutations in lagging strand of DNA replication in Escherichia coli than in leading strand. Science, 261, 598–600.
Sawai, T., Kawanishi, M., Takamura-Enya, T. and Yagi, T. (2009) Establishment of a method for analyzing translesion DNA synthesis across a single bulky adduct in human cells. Genes Environ., 31, 24–30.
Kawanishi, M., Kanno, T., Nishida, H., Takamura-Enya, T. and Yagi, T. (2013) Translesion DNA synthesis across various DNA adducts produced by 3-nitrobenzanthrone in Escherichia coli. Mutat. Res., 754, 32–38.
Sawai, T., Kawanishi, M., Takamura-Enya, T. and Yagi, T. (2009) Mutations mediated by translesion DNA synthesis over 4-aminobiphenyl adducts in human cells. Radiat. Biol. Commun., 44, 274–284 (in Japanese).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://doi.org/creativecommons.org/licenses/by/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Yagi, T., Fujikawa, Y., Sawai, T. et al. Error-Prone and Error-Free Translesion DNA Synthesis over Site-Specifically Created DNA Adducts of Aryl Hydrocarbons (3-Nitrobenzanthrone and 4-Aminobiphenyl). Toxicol Res. 33, 265–272 (2017). https://doi.org/10.5487/TR.2017.33.4.265
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2017.33.4.265