Abstract
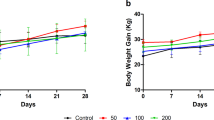
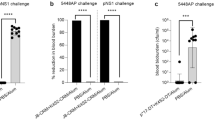
This study was performed to evaluate the toxicity of polysaccharide-based Streptococcus pneumoniae vaccine in Specific Pathogen Free (SPF), Sprague-Dawley (SD) rats. S. pneumoniae vaccine was administrated subcutaneously each dose level of high (560 μg/rat), medium (280 μg/rat) and low (140 μg/rat) on days 0, 14, 28. The rats were observed for 2 weeks or 4 weeks after the final injection. During this test, there were no significant dose-dependent changes in body weight, water and food consumption. In urinalysis and serum chemistry, dose-related changes were not detected. In hematology, the percent of neutrophils and lymphocytes in white blood cells were changed significantly. According to the measurement of organ weight, only spleen weight was significantly increased in all groups of administration compared to the control group. In the histopathological examination, an antigen-deposit, vacuolated macrophages, infiltrated inflammatory cells and a formation of granulation tissue were observed at the site of an administration. These results are considered as an outcome by immune responses through a vaccination. Consequently, the results of this study demonstrated that S. pneumoniae vaccine has no toxicity when it was administrated subcutaneously three times in 2-week interval at a high dose of 560 μg/rat.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Black, S., Shinefield, H., Fireman, B., Lewis, E., Ray, P., Hansen, J.R., Elvin, L., Ensor, K.M., Hackell, J., Siber, G., Malinoski, F., Madore, D., Chang, I., Kohberger, R., Watson, W., Austrian, R. and Edwards, K. (2000). Efficacy, safety and immunogenicity of heptavalent pneumococcal conjugate vaccine in children. Northern California Kaiser Permanente Vaccine Study Center Group. Pediatr. Infect. Dis. J., 19, 187–195.
Charles, A.J., Paul, T., Mark, W. and Mark, S. (2005). ImmunoBiology-the immune system in health and disease (6th edition), Garland Science, NewYork, pp. 12–23.
Daoust, V., Carlo, D.J., Zelther, J.Y. and Perry, M.B. (1981). Specific Capsular polysaccharide of Type 45 Streptococcus pneumonia (American Type 72). Infect. Immun., 32, 1028–1033.
Fenske, D.B., Chonn, A. and Cullis, P.R. (2008). Liposomal Nanomedicines: An Emerging Field. Toxicol. Pathol., 36, 21–29.
Hausdorff, W.P., Feikin, D.R. and Klugman, K.P. (2005). Epidemiological differences among pneumococcal serotypes. Lancet. Infect. Dis., 5, 83–93.
Henriksen-Lacey, M., Bramwell, V.W., Christensen, D., Agger, E.M., Andersen, P. and Perrie, Y. (2010). Liposome based on dimethyldioctadecylammonium promote a depot effect and enhance immungenicity of soluble antigen. J. Control. Release., 142, 180–186.
Korea Food & Drug administration (2008): Guideline on Nonclinical Evaluation of Biopharmaceuticals, 11-147000 0-001713-14.
Korea Food & Drug administration (2008). Food & Drug statistical yearbook, 11-1470000-000023-10.
Meng, C., Lin, H., Huang, J., Wang, H., Cai, Q., Fang, L. and Guo, Y. (2009). Development of 5-valent conjugate pneumococcal protein A-capsular polysaccharide pneumococcal vaccine against invasive pneumococcal disease. Microb. Pathog., 47, 151–156.
Pletz, M.W., Maus, U., Krug, N., Welte, T. and Lode, H. (2008). Pneumococcal vaccines: mechanism of action, impact on epidemiology and adaption of the species. Int. J. Antimicrob. Agents., 32, 199–206.
Rao, M. and Alving, C.R. (2000). Delivery of lipids and liposomal proteins to the cytoplasm and Golgi of antigen-presenting cells. mangala.rao@na.amedd.army.mil. Adv. Drug. Deliy. Rev., 41, 171–188.
Richard, A.G., Thomas, J.K., Barbara, A.O. and Janis, K. (2003). Immunology (5th edition), W.H.Freeman, NewYork, pp. 335–336.
Ring, A., Weiser, J.N. and Tuomanen, E.I. (1998). Pneumococcal Trafficking across the Blood-Brain Barrier. Molecular Analysis of a Novel Bidirectional Pathway. J. Clin. Invest., 102, 347–360.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://doi.org/creativecommons.org/licenses/by/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Park, S.J., Seo, K.H. & Han, S.I. Toxicity Study of Streptococcus pneumoniae Vaccine Administrated Subcutaneously in Rats. Toxicol Res. 27, 111–118 (2011). https://doi.org/10.5487/TR.2011.27.2.111
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2011.27.2.111