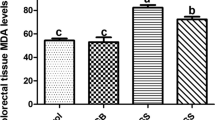
Abstract
This study investigated the antioxidative action of Corni Fructus aqueous extract on kidneys of diabetic mice. The electron donating abilities of Corni Fructus aqueous extract and its antioxidant activities (XO, SOD, CAT, GST, eNOS) in kidneys of C57BL/6 or db/db mice were evaluated. For in vivo study, seven week-old male mice were divided into normal control group (NC, C57BL/6 mice), diabetic control group (DC, db/db mice) and Corni Fructus (500 mg/kg/day for 8 weeks) treated diabetic group (DCF, db/db mice). The electron donating abilities of Corni Fructus aqueous extract exhibited 7%, 24.4%, and 42.7% at concentrations of 100, 500, and 1000 μg/ml, respectively. The activity of XO in the DCF group was significantly lower than the DC group by 35% (p < 0.05). The SOD activity was significantly higher in the DCF group than the DC group by 26% (p < 0.05). The activities of CAT and GST were lowered in the DCF group than the DC group by 26% (p < 0.05) and 7.6%, respectively. The mRNA expression of eNOS in kidneys was lower in the DCF group than the DC group by 24%. These results indicate that Corni Fructus reduced oxidation stress as evidenced by the restoration of the enzymatic antioxidative defense system in renal tissues of db/db mice. It is suggested that these antioxidative actions of Corni Fructus on renal tissues in db/db mice could contribute to its renoprotective effects on diabetic nephropathy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aebi, H. (1984). Catalase in vitro. Methods Enzymol, 105, 121–126.
Baynes, J.W. (1991). Role of oxidative stress in development of complications in diabetes. Diabetes, 40, 405–412.
Blois, M.S. (1958). Antioxidant determination by the use of a stable free radical. Nature, 181, 1199–1200.
Butler, R., Morris, A.D., Belch, H., Hill, A. and Struthers, A.D. (2000). Allopurinol normalizes endothelial dysfunction in type 2 diabetics with mild hypertension. Hypertension, 35, 746–751.
Del Maestro, R.E. (1980). An approach to free radicals in medicine and biology. Acta Physiol. Scand. Suppl, 492, 153–168.
Habig, W.H., Pabst, M.J. and Jakoby, W.B. (1974). Glutathione S-transferase. The first enzymatic step in mercapturic acid formation. J. Biol. Chem., 249, 7130–7139.
Hunt, J.V., Dean, R.T. and Wolff, S.P. (1988). Hydroxyl radical production and autoxidative glycosylation. Glucose autoxidation as the cause of protein damage in the experimental glycation model of diabetes and aging. Biochem. J., 256, 205–212.
Jin, U.H., Kim, D.I., Lee, T.K., Lee, D.N., Kim, J.K., Lee, I.S. and Kim, C.H. (2006). Herbal formulation, Yukmi-jihang-tang-Jahage, regulates bone resorption by inhibition of phosphorylation mediated by tyrosine kinase Src and cyclooxygenase expression. J. Ethnopharmacol., 106, 333–343.
Karasu, C. (2010). Glycoxidative stress and cardiovascular complications in experimentally-induced diabetes: effects of antioxidant treatment. Open Cardiovasc. Med. J., 4, 240–256.
Kim, H.J. and Kim, Y.C. (2010). Antidiabetic and renoprotective effects of Corni Fructus extract in db/db mice. Mol. Cell Toxicol, 6, 135–142.
Kim, H.J., Kim, K.S., Lee, T.J. and Kim, Y.C. (2009). Antidiabetic effects of Corni Fructus extract on blood glucose and insulin resistance in db/db mice. Toxicol. Res., 25, 93–99.
Larkins, R.G. and Dunlop, M.E. (1992). The link between hyperglycemia and diabetic nephropathy. Diabetologia, 35, 499–504.
Latha, M. and Pari, L. (2003). Preventive effects of Cassia auricu-lata L. flowers on brain lipid peroxidation in rats treated with streptozotocin. Mol. Cell. Biochem., 243, 23–28.
Lawrence, J.C., Jill, S.G., Eric, P.D., Joyce, A.D., Donald, D.L. and Mark, A.Y (2001). Effect of antioxidant treatment of strep-tozotocin-induced diabetic rats on endoneurial blood flow, motor nerve conduction velocity and vascular reactivity of epineurial arterioles of the sciatic nerve. Diabetes, 50, 1927–1937.
Li, K.M., Yang, X.J., Yu, M.Q., Xie, C. and Xu, L.Z. (1994). Determination of loganin in Cornus officinalis Sieb. et Zucc. by TLC scanner. Zhongguo Zhong Yao Za Zhi, 19, 738–763.
Lowenstein, C.J. and Snyder, S.J. (1992). Nitric oxide, a novel biologic messenger. Cell, 70, 705–707.
Lowry, O.H., Rosenbrough, N.J., Far, A.L. and Randall, R.J. (1951). Protein measurement with the folin phenol reagent. J. Biol. Chem., 193, 265–275.
Maritim, A.C., Sanders, R.A. and Watkins, J.B. (2003). Effect of alpha lipoic acid on biomarkers of oxidative stress in streptozo-tocin-induced diabetic rats. J. Nutr. Biochem., 14, 288–294.
Martin, J.P., Dailey, M. and Sugarman, E. (1987). Negative and positive assays of superoxide dismutase based on hematoxylin autoxidation. Arch. Biochem. Biophys., 255, 329–336.
Morel, D.W. and Chisolm, G.M. (1989). Antioxidative treatment of diabetic rats inhibits lipoprotein oxidation and cytotoxicity. J. Lipid Res., 30, 1827–1834.
Rong, Y., Li, L., Shah, V. and Lau, B.H.S. (1995). Pycnogenol protects vascular endothelial cells from t-butyl hydroperoxide-induced oxidant injury. Biotechnol. Ther., 5, 117–126.
Stirpe, F. and Delia, C.E. (1969). The regulation of rat liver xanthine oxidase. Conversion in vitro of the enzyme activity from dehydrogenase (type D) to oxidase (type O). J. Biol. Chem., 244, 3855–3863.
Taghizadeh Afshari, A., Shirpoor, A., Farshid, A., Saadatian, R., Rasmi, Y., Saboory, E., Ilkhanizadeh, B. and Allameh, A. (2007). The effect of ginger on diabetic nephropathy, plasma antioxidant capacity and lipid peroxidation in rats. Food Chem., 101, 148–153.
Venkateswaran, S. and Pari, L. (2002). Antioxidant effect of Phaseolus vulgaris in streptozotocin-induced diabetic rats. Asia Pac. J. Clin. Nutr., 11, 206–209.
Xia., Y., Dawson, V.L., Dawson, T.M., Snyder, S.H. and Zweier, J.L. (1996). Nitric oxide synthase generates superoxide and nitric oxide in arginine-depleted cells leading to peroxynitrite-mediated cellular injury. Proc. Natl. Acad. Sci. USA, 93, 6770–6774.
Yamabe, N., Kang, K.S., Goto, E., Tanaka, T. and Yokozawa, T. (2007). Beneficial effect of Corni Fructus, a constituent of Hachimi-jio-gan, on advanced glycation end-product-mediated renal injury in streptozotocin-treated diabetic rats. Biol. Pharm. Bull., 30, 520–526.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, HJ., Kim, BH. & Kim, YC. Antioxidative Action of Corni Fructus Aqueous Extract on Kidneys of Diabetic Mice. Toxicol Res. 27, 37–41 (2011). https://doi.org/10.5487/TR.2011.27.1.037
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2011.27.1.037