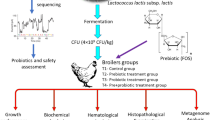
Abstract
Growth promoters including hormonal substances and antibiotics are used legally and illegally in food producing animals for the growth promotion of livestock animals. Hormonal substances still under debate in terms of their human health impacts are estradiol-17β, progesterone, testosterone, zeranol, trenbolone, and melengestrol acetate (MGA). Many of the risk assessment results of natural steroid hormones have presented negligible impacts when they are used under good veterinary practices. For synthetic hormonelike substances, ADIs and MRLs have been established for food safety along with the approval of animal treatment. Small amounts of antibiotics added to feedstuff present growth promotion effects via the prevention of infectious diseases at doses lower than therapeutic dose. The induction of antimicrobial resistant bacteria and the disruption of normal human intestinal flora are major concerns in terms of human health impact. Regulatory guidance such as ADIs and MRLs fully reflect the impact on human gastrointestinal microflora. However, before deciding on any risk management options, risk assessments of antimicrobial resistance require large-scale evidence regarding the relationship between antimicrobial use in food-producing animals and the occurrence of antimicrobial resistance in human pathogens. In this article, the risk profiles of hormonal and antibacterial growth promoters are provided based on recent toxicity and human exposure information, and recommendations for risk management to prevent human health impacts by the use of growth promoters are also presented.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Angsusingha, K., Kenny, F.M., Nankin, H.R. and Taylor, F.H. (1974). Unconjugated estrone, estradiol and FSH and LH in prepubertal and pubertal males and females. J. Endocrinol. Metab., 39, 63–68.
Bartholomew, R.M. and Ryan, D.S. (1980). Lack of mutagenicity of some phytoestrogens in the Salmonella/mammalian microsome assay. Muta. Res., 78, 317–321.
Bauer, E.R.S., Daxenberger, A., Petri, T., Sauerwein, H. and Meyer, H.H.D. (2000). Characterisation of the affinity of different anabolics and synthetic hormones to the human androgen receptor, human sex hormone binding globulin, and bovine gestagen receptor. APMIS, 108, 838–846.
Butaye, P., Devriese, L.A. and Haesebrouck, F. (2003). Antimicrobial growth promoters used in animal feed: Effects of less well known antibiotics on gram-positive bacteria. Clin. Microbiol. Rev., 16, 175–188.
Butler, G.E., Sellar, R.E., Walker, R.F., Kelnar, C.J.H. and Wu, F.C.W. (1992). Oral testosterone undecanoate in the management of delayed puberty in boys: Pharmacokinetics and effect on sexual maturation. J. Clin. Endocrinol. Metab., 75, 37–44.
Corpet, D.E. (2000). Oral testosterone undecanoate in the management of delayed puberty in boys. Rev. Med. Vet., 151, 99–104.
Cox, L.A. and Popken, D.A. (2004). Quantifying human health risks from virginiamycin used in chickens. Risk Anal., 24, 271–288.
Cox, L.A., Popken, D.A. and Mathers, J.J. (2009). Human health risk assessment of penicillin/aminopenicillin resistance in Enterococci due to penicillin use in food animals. Risk Anal., 29, 796–805.
Davis, G.I., McLachlan, J.A. and Lucier, G.W. (1977). Fetotoxicity and teratogenicity of zeranol in mice. Tox. Appl. Pharm., 41, 138–139.
Dibner, J.J. and Richards, J.D. (2005). Antibiotic growth promoters in agriculture: History and mode of action. Poult. Sci., 84, 634–643.
Durhan, E.J., Lambright, C.S., Makynen, E.A., Lazorchak, J., Hartig, P.C., Wilson, V.S., Gray, L.E. and Ankley, G.T. (2006). Identification of metabolites of trenbolone acetate in androgenic runoff from a beef feedlot. Environ. Health Perspect., 114, 65–68.
European Food Safety Authority (EFSA) (2007). Opinion of the scientific panel on contaminations in the food chain on a request from the European Commission related to hormone residues in bovine meat and meat products. The EFSA Journal, 510, 1–62.
European Medicine Agency (EMA) (2003). Veterinary medicines-Maximum residue limits (MRL): Summary Reports and EPMARs-Bacitrcin (3). http://www.ema.europa.eu/pdfs/vet/mrls/085702en.pdf.
European Medicine Agency (EMA) (2008). Veterinary medicines-Maximum residue limits (MRL): Summary Reports and EPMARs-Tiamulin (3). http://www.ema.europa.eu/pdfs/vet/mrls/074700en.pdf.
Everett, D.J., Perry, C.J., Scott, K.A., Martin, B.W. and Terry, M.K. (1987). Estrogenic potencies of resorcyclic acid lactones and 17-β-estradiol in female rats. J. Toxicol. Environ. Health, 20, 435–443.
FAO/WHO (2004). FAO/WHO technical workshop on residues of veterinary drugs without ADI/MRL. 8–17.
Foss, G.L. and Camb, M.B. (1939). Clinical administration of androgens. Lancet., 1, 502–504.
Fotherby, K. (1996). Bioavailability of orally administered sex steroids used in oral contraception and hormone replacement therapy. Contraception, 54, 59–69.
Galbraith, H. (2002). Hormones in international meat production: biological, sociological and consumer issues. Nutr. Res. Rev., 15, 293–314.
Gardner, W.U. (1941). The effect of estrogen on the incidence of mammary and pituitary tumors in hybrid mice. Cancer Res., 1, 345–358.
Han, X., Liehr, J.G. and Bosland, M.C. (1995). Induction of a DNA adduct detectable by 32P-postlabeling in the dorsolateral prostate of NBL/Cr rats treated with estradiol-17b and testosterone. Carcinogenesis, 16, 951–954.
Health Canada (2002). Uses of antimicrobials in food animals in Canada: Impact on resistance and human health. Report of the advisory committee on animal uses of antimicrobials and impact on resistance and human health.
Heitzman, R.J. (1983). The absorption, distribution and excretion of anabiloc agents. J. Anim. Sci., 57, 233–238.
Heitzman, R.J. and Hardwood, D.J. (1977). Residue levels of trenbolone and oestradiol-17-β in plasma and tissues of steers implanted with anabolic steroid preparations, Br.Vet. J., 133, 564–571.
Ho, S.M. and Roy, D. (1994). Sex hormone-induced nuclear DNA damage and lipid peroxidation in the dorsolateral prostates of noble rats. Cancer Lett., 84, 155–162.
Hoffman, B., Schopper, D. and Karg, H. (1984). Investigations on the occurrence of non-extractable residues of trenbolone acetate in cattle tissues in respect to their bioavailability and immunological reactivity. Food Addit. Contam., 1, 253–259.
Hurd, H.S. and Malladi, S. (2008). A stochastic assessment of the public health risks of the use of macrolide antibiotics in food animals. Risk Anal., 28, 695–710.
IARC (1979). IARC monographs on the evaluation of the carcinogenic risk of chemicals to humans. Vol. 21, Sex Hormones (II), Lyon, IARC Press. pp. 35-82, 139–547.
IARC (1987). IARC monographs on the evaluation of the carcinogenic risk of chemicals to humans. Suppl. 7, Overall evaluations of carcinogenicity: An updating of IARC monographs Volumes 1 to 42. Lyon, IARC Press. pp. 272–400.
IARC (1999). IARC monographs on the evaluation of carcinogenic risks to humans. Vol. 72, Hormonal contraception and postmenopausal hormone therapy. Lyon, IARC Press.
Ingerowski, G.H., Scheutwinkel-Reich, M. and Stan, H.J. (1981). Mutagenicity studies on veterinary anabolic drugs with the Salmonella/microsome test. Mutat. Res., 91, 93–98.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (1988). Evaluation of certain veterinary drug residues in food. WHO Technical Report Series, 763, 16–33.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (1998a). Toxicological evaluation of certain veterinary drug residues in food: Tetracyclines. WHO Food Additives Series, 41.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (1998b). Toxicological evaluation of certain veterinary drug residues in food: Benzylpenicillin. http://www.inchem.org/documents/jecfa/jecmono/v27je05.html
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (1998c). Toxicological evaluation of certain veterinary drug residues in food: Dihydrostreptomycin and streptomycin. WHO Food Additives Series, 39.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (2000a.) Procedures for recommending maximum residue limits-residues of veterinary drugs in food. pp. 43–51.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (2000b). Toxicological evaluation of certain veterinary drug residues in food: Estradiol-17â, progesterone, and testosterone. WHO Food Additives Series, 43.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (2000c). Toxicological evaluation of certain veterinary drug residues in food: Melengestrol acetate. WHO Food Additives Series, 45.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (2000d). Toxicological evaluation of certain veterinary drug residues in food: Lincomycin. WHO Food Additives Series, 45.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (2006a). Toxicological evaluation of certain veterinary drug residues in food: Colistin sulfate. WHO Food Additives Series, 57.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (2006b). Toxicological evaluation of certain veterinary drug residues in food: Erythromycin. WHO Food Additives Series, 57.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (2006c). Evaluation of certain veterinary drug residues in food. Melengestrol acetate. WHO Technical Report Series, 939.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (2009a). Toxicological evaluation of certain veterinary drug residues in food: Tilmicosin. WHO Food Additives Series, 61.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (2009b). Toxicological evaluation of certain veterinary drug residues in food: Avilamycin. WHO Food Additives Series, 61.
Joint FAO/WHO Expert Committee on Food Additives (JECFA). (2009c). Toxicological evaluation of certain veterinary drug residues in food: Tylosin. WHO Food Additives Series, 61.
Jeong, S. H., Song, Y. K., Cho, J. H. (2009). Risk assessment of ciprofloxacin, flavomycin, olaquindox and Colistin sulfate based on microbiological impact on human gut biota. Regl. Toxicol. Pharmacol., 53, 209–216.
Johnsen, S.G., Bennett, E.P. and Jensen, V.G. (1974). Therapeutic effectiveness of oral testosterone. Lancet, 2, 1473–1475.
Johnsen, S.V., Kampmann, J.P., Bennett, E.P., Jorgensen, F.S. (1976). Enzyme induction by oral testosterone. Clin. Ther., 20, 233–237.
Jones, L.A. and Bern, H.A. (1977). Long-term effects of neonatal treatment with progesterone alone and in combination with estrogen, on the mammary gland and reproductive tract of female BALB/cfc3H mice. Cancer Res., 37, 67–75.
Karr, J.P., Kim, U., Resko, J.A., Schneider, S., Chai, L.S., Murphy, G.P. and Sandburg, A.A. (1984). Induction of benign prostatic hypertrophy in baboons. Urology, 3, 276–289.
Katzenellenbogen, B.S., Katzenellenbogen, J.A. and Mordecai, D. (1979). Zearalenones: Characterization of the estrogenic potencies and receptor interactions of a series of fungal β-resorcylic acid lactones. Endocrinology, 105, 33–40.
Kim, S., Korhonen, M., Wilborn, W., Foldesy, R., Snipes, W., Hodgen, G.D. and Anderson, F.D. (1996). Antiproliferative effects of low-dose micronized progesterone. Fertil. Sterility, 65, 323–331.
Kuhnz, W., Gansau, C. and Mahler, M. (1993). Pharmacokinetics of estradiol, free and total estrone, in young women following single intravenous and oral administration of 17beta-estradiol. Arzeimittelforschung, 43, 966–973.
Lasne, C., Lu, Y.P., Orfila, L., Ventura, L. and Chouroulinkov, I. (1990). Study of various transforming effects of the anabolic agents trenbolone and testosterone on Syrian hamster embryo cells. Carcinogenesis, 11, 541–547.
Lauderdale, J.W. (1977). Studies of a progestogen (MGA) as related to residues and human consumption. J. Toxicol. Environ. Health, 3, 5–33.
van Leeuwen, F.X.R. (1993). Endocrine Toxicology: A review of the application of endocrinology in experimental toxicology. Comp. Haematol. Int., 3, 8–13.
Lutz, W.K., Deuber, R., Caviezel, M., Sagelsdorff, P., Friederich, U. and Schlatter, C. (1988). Trenbolone growth promotant: covalent DNA binding in rat liver and in Salmonella typhimurium, and mutagenicity in the Ames test. Arch. Toxicol., 62, 103–109.
Metzler, M. and Pfeiffer, E. (2001). Genotoxic potential of xenobiotic growth promoters and their metabolites. APMIS, 109, 89–95.
Miyamoto, H., Yeh, S., Lardy, H., Messing, E. and Chang, C. (1998). Delta5-androstenediol is a natural hormone with androgenic activity in human prostate cells. Proc. Natl Acad. Sci. U.S.A., 15, 11083–11088.
Moore, A.B., Bottoms, G.D., Coppoc, R.C., Pohland, R.C. and Roesel, O.F. (1982). Metabolism of estrogens in the gastrointestinal tract of swine. 1. Instilled estradiol. J. Anim. Sci., 55, 124–134.
Moyer, D.L., de Lingnieres, B., Driguez, P. and Pez, J.P. (1993). Prevention of endometrial hyperplasia by progesterone during long-term estradiol replacement: Influence of bleeding pattern and secretory changes. Fertil. Sterility, 59, 992–997.
Nahoul, K., Dehennin, L., Jondet, M. and Roger, M. (1993). Profiles of plasma estrogens, progesterone and their metabolites after oral or vaginal administration of estradiol or progesterone. Maturitas., 16, 185–202.
Neidert, E.E., Gedir, R.G., Milward, L.J., Salisbury, C.D., Gurprasad, N.P. and Saschenbreckert, P.W. (1990). Determination and qualitative confirmation of melengestrol acetate residues in beef fat by electron capture gas chromatography and gas chromatographic/chemical ionization mass spectrometry. J. Agric. Food Chem., 38, 979–981.
Paris, A. (coordinateur), Andre, F., Antignac, J.-P., Le Bizec, B., Bonneau, M., Briant, C., Caraty, A., Chilliard, Y., Cognie, Y., Combarnous, Y., Cravedi, J.-P., Fabre-Nys, C., Fernandez-Suarez, A., Fostier, A., Humblot, P., Laudet, V., Leboeuf, B., Louveau, I., Malpaux, B., Martinat-Bottle, F., Maurel, M.-C., Pelicer-Rubio, M.-T., Picard-Hagen, N., Pinault, L., Pinel, G., Ponsard, C., Popot, M.-A., Schmidely, P., Toutain, P.-L. and Zalko, D. (2006). Hormones et promoteurs de croissance en productions animales: de la physiologie à l’évaluation du risque. INRA Prod. Anim., 19, 149–240.
Perry, G.A., Welshonsb, W.V., Botta, R.C. and Smith, M.F. (2005). Basis of melengestrol acetate action as a progestin. Dornest. Anim. Endocrinol., 28, 147–161.
Phillips, I., Casewell, M., Cox, T., De Groot, B., Friis, C., Jones, R., Nightingale, C., Preston, R. and Waddell, J. (2004). Does the use of antibiotics in food animals pose a risk to human health? A critical review of published data. J. Antimicrob. Chemother, 53, 28–52.
Portier, J., Busigny, M. and Gradadam, J.A. (1973). Tissue residues in cows after implantation of trenbolone acetate. J. Anim. Sci., 37, 256 (Abstract No. 110F).
Presi, P., Stärk, K.D.C., Stephan, R., Breidenbach, E., Frey, J. and Regula, G. (2009). Risk scoring for setting priorities in a monitoring of antimicrobial resistance in meat and meat products. Int. J. Food Microbiol, 130, 94–100.
Revuelta, M.P., Cantabrana, B. and Hidalgo, A. (1997). Transcriptional Mechanisms involved in the relaxant effect of zeranol on isolated rat uterus. Gen. Pharmacol., 28, 561–565.
Robinson, P.A., Hawkey, C. and Hammond, G.L. (1985). A phylogenetic study of the structural and functional characteristics of corticosteroid binding globulin in primates. J. Endocrinol., 104, 251–257.
Rothman, K.J. and Louik, C. (1978). Oral contraceptives and birth defects. N. Engl. J. Med., 210, 522–524.
Sands, R. and Studd, J. (1995). Exogenous androgens in postmenopausal women. Am. J. Med., 98, 76S–79S.
Sarkar, K., Kinson, G.A. and Rowsell, H.C. (1986). Embryo resorption following administration of steroidal compounds to rats in mid-pregnancy. Can. J. Vet. Res., 50, 433–437.
Scheutwinkel, M., Hude, W.v.d. and Basier, A. (1986). Studies on the genotoxicity of the anabolic drugs trenbolone and zeranol. Arch. Toxicol, 59, 4–6.
Schiffmann, D., Hieber, L., Schmuck, G., Pechan, R., Metzel, M. and Henschler, D. (1988). Trenbolone induces micronucleus formation and neoplastic transformation in Syrian hamster embryo fribroblasts but not in mouse C3H10T1/2 cells. Arch. Toxicol., 63, 49–53.
Schiffman, D., Metzler, M., Neudecker, T. and Henschler, D. (1985). Morphological transformation of Syrian hamster embryo fibroblasts by the anabolic agent trenbolone. Arch. Toxicol., 58, 59–63.
Seraj, M.J., Umemoto, A., Tanaka, M., Kajikawa, A., Hamada, K. and Monden, Y. (1996). DNA adduct formation by hormonal steroids in vitro. Mutat. Res., 370, 49–59.
Shangold, M.M., Tomai, T.P., Cook, J.D., Jacobs, S.L., Zinaman, M.J., Chin, S.Y. and Simon, J.A. (1991). Factors associated with withdrawal bleeding after administration of oral micronized progesterone in women with secondary amenorrhea. Fertil. Steril., 56, 1040–1047.
Shenderov, B.A. (1998). Some results and problems of host microbial ecology investigations. The 7th International Symposium on lactic acid bacteria and human health. pp. 322–330.
Sillence, M.N. and Rodway, R.G. (1990). Effects of trenbolone acetate and testosterone on growth and on plasma concentrations of corticosterone and ACTH in rats. J. Endocrinol., 126, 461–466.
Simon, J.A., Robinson, D.E., Andrews, M.C., Hildebrand, J.R., III, Rocci, M.L., Jr, Blake, R.E. and Hodgen, G.D. (1993). The absorption of oral micronized progesterone: The effect of food, dose proportionality, and comparison with intramuscular progesterone. Fertil. Steril., 60, 26–33.
Sitruk-Ware, R., Bricaire, C., de Lignieres, B., Yaneva, H. and Mauvais-Jarvis, P. (1987). Oral micronized progesterone. Contraception, 36, 373–402.
Snary, E.L., Kelly, L.A., Davison, H.C., Teale, C.J. and Wooldridge, M. (2004). Antimicrobial resistance: A microbial risk assessment perspective. J. Antimicrob. Chemother, 53, 906–917.
Tauber, U., Schroder, K., Dusterberg, B. and Matthes, H. (1986). Absolute bioavailability of testosterone after oral administration of testosterone-undecanoate and testosterone. Eur. J. Drug. Metab. Pharmacokinet., 11, 145–149.
Tsutsui, T., Konine, A., Huff, J. and Barrett, J.C. (1995). Effects of testosterone, testosterone propionate, 17beta-trenbolone and progesterone on cell transformation and mutagenesis in Syrian hamster embryo cells. Carcinogenesis, 16, 1329–1333.
Wagner, J.F. (1983). Estradiol controlled release implants-efficacy and drug delivery. In: Meissonnier, E. ed. Anabolics in animal production. France, Office International des Epizooties. pp. 129–142.
Wharton, L.R. and Scott, R.B. (1964). Experimental production of genital lesions with norethindrone. Am. J. Obstet. Gynecol., 89, 701–715.
World Health Organization (WHO) (2002). Antimicrobial resistance. http://www.who.int/mediacentre/factsheets/fs194/en/
World Health Organization (WHO) (2004). Evaluation of certain veterinary drug residues in food. WHO Technical Report Series, 925, 22–25.
Williams, G.M. (1984). DNA damage and repair tests for the detection of genotoxic material. Food Addit. Contam., 1, 173–178.
Wilson, V.S., Lambright, C., Ostby, J. and Gray, Jr. L.E. (2002). In Vitro and in Vivo Effects of 17β-Trenbolone: A Feedlot Effluent Contaminant. Toxicol. Sci., 70, 202–211.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jeong, SH., Kang, D., Lim, MW. et al. Risk Assessment of Growth Hormones and Antimicrobial Residues in Meat. Toxicol Res. 26, 301–313 (2010). https://doi.org/10.5487/TR.2010.26.4.301
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2010.26.4.301