Abstract
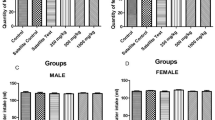
This study was conducted to obtain information of the oral dose toxicity of low molecular fucoidan (LMF) in male and female mice. In order to calculate 50% lethal dose (LD50) and approximate lethal dose (LD), test material was once orally administered to male and female ICR mice at dose levels of 2000, 1000, 500, 250, 125 and 0 (vehicle control) mg/kg (body wt.). The mortality and the changes on body weight, clinical signs, gross observation and organ weight and histopathology of principle organs were monitored 14 days after LMF treatment. We could not find any mortalities, clinical signs, body weight changes and gross findings. In addition, significant changes in the organ weight and histopathology of principal organs were not observed except for some sporadic findings. The results obtained in this study suggest that LMF may not be toxic in mice and may be therefore safe for clinical use. The LD50 and approximate LD in mice after single oral dose of LMF were considered over 2000 mg/kg in both female and male mice.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alkhatib, B., Freguin-Bouilland, C., Lallemand, F., Henry, J.P., Litzler, P.Y., Marie, J.P., Richard, V., Thuillez, C. and Plissonnier, D. (2006). Low molecular weight fucan prevents transplant coronaropathy in rat cardiac allograft model. Transpl. Immunol., 16, 14–19.
Banks, W.J. (1986). Female reproductive system in Applied veterinary histology (Banks, W.J. Ed.). Williams & Wilkins, Baltimore, pp. 506–526.
Béress, A., Wassermann, O., Tahhan, S., Bruhn, T., Beress, L. and Kraiselburd, E.N. (1993). A new procedure for the isolation of anti-HIV compounds (polysaccharide and polyphenols) from the marine algae Fucus vesiculosus. J. Nat. Prod., 56, 478–488.
Berteau, O. and Mulloy, B. (2003). Sulfated fucans, fresh perspectives: structures, functions, and biological properties of sulfated fucans and an overview of enzymes active toward this class of polysaccharide. Glycobiology, 13, 29R–40R.
Blondin, C., Chaubet, F., Nardella, A., Sinquin, C. and Jozefonvicz, J. (1996). Relationships between chemical characteristics and anticomplementary activity of fucans. Biomaterials, 17, 597–603.
Bojakowski, K., Abramczyk, P., Bojakowska, M., Zwolinska, A., Przybylski, J. and Gaciong, Z. (2001). Fucoidan improves the renal blood flow in the early stage of renal ischemia/reperfusion injury in the rat. J. Physiol. Pharmacol., 52, 137–143.
Colliec, S., Fischer, A.M., Tapon-Bretaudiere, J., Boisson, C., Durand, P. and Jozefonvicz, J. (1991). Anticoagulant properties of a fucoidan fraction. Thromb. Res., 64, 143–154.
Dourish, C.T. (1987). Effects of drugs on spontaneous motor activity in Experimental Psychopharmacology (Greenshaw, A.J. and Dourish, C.T. Eds.). Humana Press, Clifton, pp. 325–334.
Dubois, M., Gilles, K.A., Hamilton, J.K., Rebers, P.A. and Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Anal. Chem., 28, 350–354.
Farndale, R.W., Buttle, D.J. and Barret, A.J. (1986). Improved quantification and discrimination of sulphated glycosaminoglycans by use of dimethylmethylene blue. Biochim. Biophys. Acta, 883, 173–177.
Fréguin-Bouilland, C., Alkhatib, B., David, N., Lallemand, F., Henry, J.P., Godin, M., Thuillez, C. and Plissonnier, D. (2007). Low molecular weight fucoidan prevents neointimal hyperplasia after aortic allografting. Transplantation, 83, 1234–1241.
Haroun-Bouhedja, F., Ellouali, M., Sinquin, C. and Boisson-Vidal, C. (2000). Relationship between sulfate groups and biological activities of fucans. Thromb. Res., 100, 453–459.
Hodge, H.C. and Sterner, J.H. (1949). Tabulation of toxicity classes, Am. Ind. Hyg. Q., 10, 93.
Irwin, S. (1968). Comprehensive observational assessment: Ia. A systemic, quantitative procedure for assessing the behavioral and physiological state of the mouse. Psychopharmacology, 13, 222–257.
Jung, Y.M., Kim, T.K., Park, D.C. and Lee, Y.H. (2007). Bioavailable Fucoidan and Methods for Preparing the Same, Korean Patent 0705975.
Korea Food and Drug Administration. (2005). Testing Guidelines for Safety Evaluation of Drugs (Notification No. 2005-60, issued by the Korea Food and Drug Administration on October 21, 2005).
Lake, A.C., Vassy, R., Di Benedetto, M., Lavigne, D., Le Visage, C., Perret, G.Y. and Letourneur, D. (2006). Low molecular weight fucoidan increases VEGF165-induced endothelial cell migration by enhancing VEGF165 binding to VEGFR-2 and NRP1. J. Biol. Chem., 281, 37844–37852.
Lee, H.S., Lee, I.G. and Ku, S.K. (2006). Single oral dose toxicity study of water extracts of Picrorrhiza Rhizoma in mice. J. Toxicol. Pub. Health, 22, 117–126.
Lee, H.S., Yang, K.J., Shin, H.D., Park, B.R., Son, C.W., Jang, H.J., Park, D.C., Jung, Y.M. and Ku, S.K. (2005). Single oral dose toxicity study of polycan, β-glucan originated from Aureobasidium in mice. J. Toxicol. Pub. Health, 21, 361–365.
Lee, Y.H., Son, M.K., Jung, Y.M., Kim, T.K., Park, D.C., Lee, H.S., Kim, P.S. and Ku, S.K. (2007). Mouse single oral dose toxicity studies of PGB-1, a novel polyglucosamine polymer produce from Enterobacter sp. BL-2. J. Toxicol. Pub. Health, 23, 373–382.
Li, N., Zhang, Q. and Song, J. (2005): Toxicological evaluation of fucoidan extracted from Laminaria japonica in Wistar rats. Food Chem. Toxicol., 43, 421–426.
Marais, M.F. and Joseleau, J.P. (2001). A fucoidan fraction from Ascophyllum nodosum. Carbohydr. Res., 336, 155–159.
Matsubara, K., Xue, C., Zhao, X., Mori, M., Sugawara, T. and Hirata, T. (2005). Effects of middle molecular weight fucoidans on in vitro and ex vivo angiogenesis of endothelial cells. Int. J. Mol. Med., 15, 695–699.
Nardella, A., Chaubet, F., Sinquin, C., Colliec Jouault, S., Boisson-Vidal, C., Durand, P. and Jozefonvicz, J. (2000). Method for Obtaining Sulfated Polysaccharides, United States Patent 6,028,191.
Organization for Economic Co-Operation and Development (Ed.). (2001): OECD guideline (423) for testing of chemicals-acute oral toxicity-acute toxic class method.
Patankar, M.S., Oehninger, S., Barnett, T., Williams, R.L. and Clark, G.F. (1993). A revised structure for fucoidan may explain some of its biological activities. J. Biol. Chem., 268, 21770–21776.
Plata, E.J. and Murphy, W.H. (1972). Growth and haematologic properties of the BALB/wm strain of inbred mice. Lab. Anim. Sci., 22, 712–720.
Senni, K., Gueniche, F., Foucault-Bertaud, A., Igondjo-Tchen, S., Fioretti, F., Colliec-Jouault, S., Durand, P., Guezennec, J., Godeau, G. and Letourneur, D. (2006). Fucoidan a sulfated polysaccharide from brown algae is a potent modulator of connective tissue proteolysis. Arch. Biochem. Biophys. 445, 56–64.
Shimizu, J., Wada-Funada, U., Mano, H. Matahira, Y. Kawaguchi, M. and Wada, M. (2005). Proportion of murine cytotoxic T cells is increased by high molecular-weight fucoidan extracted from Okinawa mozuku (Cladosiphon okamuranus). J. Health Sci., 51, 394–397.
US Environmental Protection Agency. (1998). Health Effects Test Guidelines OPPTS 870.100, Acute Toxicity Testing Background, US EPA August, Washington, USA.
Yamaguchi, C., Fujita, S., Obara, T. and Ueda, T. (1983). Effects of room temperature on reproduction, body weight and organ weights, food and water intakes, and hematology in mice. Exp. Anim., 32, 1–11.
Zemani, F., Benisvy, D., Galy-Fauroux, I., Lokajczyk, A., Colliec-Jouault, S., Uzan, G., Fischer, A.M. and Boisson-Vidal, C. (2005). Low-molecular-weight fucoidan enhances the proangiogenic phenotype of endothelial progenitor cells. Biochem. Pharmacol., 70, 1167–1175.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jung, YM., Yoo, K.M., Park, DC. et al. Oral Single Dose Toxicity Study of Low Molecular Fucoidan in Mice. Toxicol Res. 24, 79–86 (2008). https://doi.org/10.5487/TR.2008.24.1.079
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2008.24.1.079