Abstract
Background: Herniation of nuclear or disc material along with, inflammatory chemokines such as prostaglandin E2, interleukin-6, matrix metalloproteinase and nitric oxide has definite correlation, possibly they are over produced. CX3CL1 and its receptor (CX3CR1) are part of chemokine system involved in leukocyte recruitment and adhesion in chronic inflammatory disease, but its role in spinal herniated nucleus pulposus (HNP) is unknown. We evaluated the expression of CX3CL1 and CX3CR1 in patients with disc herniation to clarify the role of CX3CL1 and CX3CR1 in the disc degeneration and to compare between cervical and lumbar HNP
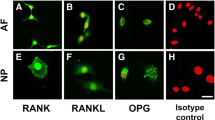
Materials and Methods: The mRNA concentrations of CX3CL1/CX3CR1 chemokine were analyzed in the surgically obtained disc specimens from C-HNP (n = 13) and L-HNP (n = 13) by real-time polymerase chain reaction (PCR). The localization of CX3CL1/CX3CR1 chemokine in the disc of C-HNP and L-HNP patients was determined using immunohistochemical study. Blood samples from patients with C-HNP and L-HNP patients were stained for CX3CR1 with flow cytometric analysis.
Results: The CX3CL1 positive cell ratio in the discs was observed in both groups by immunohistochemical study. CX3CR1 was strongly expressed on endothelial cells in C-spine disc, but sparely expressed in L-spine disc. There was greater CX3CR1 mRNA expression in C-HNP patients than in L-HNP patients as quantified by reversal transcription-PCR (P = 0.010). CX3CR1 positive cell frequencies and CX3CR1 expression levels were increased in CD4 (+) T-cells and natural killer (NK) cells from patients with C-HNP (P = 0.210 and P = 0.040).
Conclusions: This study identified that increases in CX3CL1 and CX3CR1-expressing cells are significantly related to pathomechanism of HNP for the first time. Especially, CD4(+) T-cells and NK cells expressing CX3CR1 may play an important role in developing C-HNP.
Similar content being viewed by others
References
Kalichman L, Hunter DJ. The genetics of intervertebral disc degeneration. Associated genes. Joint Bone Spine 2008;75:388–96.
Bazan JF, Bacon KB, Hcardiman G, Wang W, Soo K, Rossi D, et al. A new class of membrane-bound chemokine with a CX3C motif. Nature 1997;385:640–4.
Fraticelli P, Sironi M, Bianchi G, DAmbrosio D, Albanesi C, Stoppacciaro A, et al. Fractalkine (CX3CL1) as an amplification circuit of polarized Thl responses. J Clin Invest 2001;107:1173–81.
Imai T, Hieshima K, Haskell C, Baba M, Nagira M, Nishimura M, et al. Identification and molecular characterization of fractalkine receptor CX3CR1, which mediates both leukocyte migration and adhesion. Cell 1997;91:521–30.
Fong AM, Robinson LA, Steeber DA, Tedder TF, Yoshie O, Imai T, et al. Fractalkine and CX3CR1 mediate a novel mechanism of leukocyte capture, firm adhesion, and activation under physiologic flow. J Exp Med 1998;188:1413–9.
Haskell CA, Cleary MD, Charo IF. Molecular uncoupling of fractalkine-mediated cell adhesion and signal transduction. Rapid flow arrest of CX3CRl-expressing cells is independent of G-protein activation. J Biol Chem 1999;274:10053–8.
Nishimura M, Umehara H, Nakayama T, Yoneda O, Hieshima K, Kakizaki M, et al. Dual functions of fractalkine/ CX3Cligand 1 in trafficking of perforin+/granzymeB+cytotoxic effector lymphocytes that are defined by CX3CR1 expression. J Immunol 2002;168:6173–80.
Braunersreuther V, Mach F, Steffens S. The specific role of chemokines in atherosclerosis. Thromb Haemost 2007;97:714–21.
Shimoda S, Harada K, Niiro H, Taketomi A, Maehara Y, Tsuneyama K, et al. CX3CL1 (fractalkine): A signpost for biliary inflammation in primary biliary cirrhosis. Hepatology 2010;51:567–75.
Suzuki F, Kubota T, Miyazaki Y, Ishikawa K, Ebisawa M, Hirohata S, et al. Serum level of soluble CX3CL1/fractalkine is elevated in patients with polymyositis and dermatomyositis, which is correlated with disease activity. Arthritis Res Ther 2012;14:R48.
Xuan W, Liao Y, Chen B, Huang Q, Xu D, Liu Y, et al. Detrimental effect of fractalkine on myocardial ischaemia and heart failure. Cardiovasc Res 2011;92:385–93.
Available from http://fokker.wi.mit.edu/cgi-bin/primer3 [Last accessed on 2015 Feb 4]
Cassinelli EH, Hall RA, KangJD. Biochemistry of intervertebral disc degeneration and the potential for gene therapy applications. Spine J 2001;1:205–14.
Vernon-Roberts B, Pirie CJ. Degenerative changes in the intervertebral discs of the lumbar spine and their sequelae. Rheumatol Rehabil 1977;16:13–21.
Lawrence JS. Disc degeneration. Its frequency and relationship to symptoms. Ann Rheum Dis 1969;28:121–38.
Miller JA, Schmatz C, Schultz AB. Lumbar disc degeneration: Correlation with age, sex, and spine level in 600 autopsy specimens. Spine (Phila Pa 1976) 1988;13:173–8.
Haefeli M, Kalberer F, Saegesser D, NerlichAG, Boos N, Paesold G. The course of macroscopic degeneration in the human lumbar intervertebral disc. Spine (Phila Pa 1976) 2006;31:1522–31.
Moneta GB, Videman T, Kaivanto K, Aprill C, Spivey M, Vanharanta H, et al. Reported pain during lumbar discography as a function of anular ruptures and disc degeneration. A re analysis of 833 discograms. Spine (Phila Pa 1976) 1994;19:1968–74.
Leonov A, Trofimov S, Ermakov S, Livshits G. Quantitative genetic study of amphiregulin and fractalkine circulating levels-Potential markers of arthropathies. Osteoarthritis Cartilage 2011;19:737–42.
Nanki T, Imai T, Nagasaka K, Urasaki Y, Nonomura Y, Taniguchi K, et al. Migration of CX3CRl-positive T cells producing type 1 cytokines and cytotoxic molecules into the synovium of patients with rheumatoid arthritis. Arthritis Rheum 2002;46:2878–83.
Doita M, Kanatani T, Harada T, Mizuno K. Immunohistologic study of the ruptured intervertebral disc of the lumbar spine. Spine (Phila Pa 1976) 1996;21:235–41.
Haro H, Shinomiya K, Komori H, Okawa A, Saito I, Miyasaka N, et al. Upregulated expression ofchemokines in herniated nucleus pulposus resorption. Spine (Phila Pa 1976) 1996;21:1647–52.
Ikeda T, Nakamura T, Kikuchi T, Umeda S, Senda H, Takagi K. Pathomechanism of spontaneous regression of the herniated lumbar disc: Histologic and immunohistochemical study. J Spinal Disord 1996;9:136–40.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oh, IS., Suh, DW., Park, SR. et al. Fractalkine receptor chemokine (CX3CR1) influences on cervical and lumbar disc herniation. IJOO 49, 239–244 (2015). https://doi.org/10.4103/0019-5413.152505
Published:
Issue Date:
DOI: https://doi.org/10.4103/0019-5413.152505