Abstract
Actinoplanes missouriensis Couch 1963 is a well-characterized member of the genus Actinoplanes, which is of morphological interest because its members typically produce sporangia containing motile spores. The sporangiospores are motile by means of flagella and exhibit chemotactic properties. It is of further interest that members of Actinoplanes are prolific sources of novel antibiotics, enzymes, and other bioactive compounds. Here, we describe the features of A. missouriensis 431T, together with the complete genome sequence and annotation. The 8,773,466 bp genome contains 8,125 protein-coding and 79 RNA genes.
Similar content being viewed by others
Introduction
Strain 431T [= NBRC 102363T = DSM 43046T = ATCC 14538T and other culture collections] is the type strain of the species Actinoplanes missouriensis [1,2], which is a well-characterized member of the genus Actinoplanes [3]. The genus is of morphological interest because its members typically produce spherical, subspherical, cylindrical, or very irregular sporangia arising from vegetative mycelia [1,4]. Sporangiospores, which are released following the immersion of sporangia in water, are motile by means of polar or peritrichous flagella [5] and display a positive chemotactic response to a number of amino acids, aromatic compounds, sugars and inorganic ions [6,7]. Recently, the flagellin gene of various Actinoplanes strains was successfully amplified and sequenced to reveal the evolutionary relationships between flagellar genes [8]. As members of the genus Actinoplanes, including A. missouriensis, produce a variety of antibiotics, enzymes and other bioactive compounds [9–12], the genomes of Actinoplanes species are potentially useful genetic resources for discovering secondary metabolites and enzymes. The genome analysis of the acarbose producer Actinoplanes sp. SE50/110 was most recently reported [13].
Here, we present a summary classification and a set of features for A. missouriensis strain 431T, together with a description of the complete genome sequencing and annotation.
Classification and features
A. missouriensis strain 431T was originally isolated from barnyard soil near Hamilton, Missouri, USA using the pollen-baiting technique and was first described by Couch in 1963 [1]. Couch also isolated additional strains of A. missouriensis from ten soil collections obtained from the Mississippi Valley to the West Coast. The range of 16S rRNA gene sequence similarities between strain 431T (= NBRC 102363T) (AB711914) and valid members of the genus Actinoplanes was 95.7–97.9%. The highest sequence similarities were to Actinoplanes utahensis NBRC 13244T.
Figure 1 shows the phylogenetic neighborhood of A. missouriensis 431T in a 16S rRNA gene-based tree. The sequences of five of the six 16S rRNA gene copies in the genome of A. missouriensis 431T are identical (one sequence differed by 4 nucleotides), but differ by 55 nucleotides (3.8%) from the previously published 16S rRNA gene sequence of NBRC 13243T (AB037008). The differences between the genome data and the reported 16S rRNA gene sequence are likely due to sequencing errors in the previously reported sequence data. The 16S rRNA sequence of strain 431T did not match significantly with any 16S rRNA gene sequences from environmental genomic samples and surveys available at the NCBI BLAST server (March 2012).
Phylogenetic tree highlighting the position of A. missouriensis 431T relative to Actinoplanes sp. strain SE50/110 and other type strains within the genus Actinoplanes. The tree was inferred from 1,351 aligned characters [14,15] of the 16S rRNA gene sequence under the maximum likelihood criterion [16] and rooted with the type strain of the neighboring genus Dactylosporangium. Only bootstrap values above 50% are shown (1,000 resamplings) at branching points. Lineages with type strain genome sequencing projects registered in GOLD [17] are labeled with an asterisk, those also listed as ‘Complete and Published’ with two asterisks.
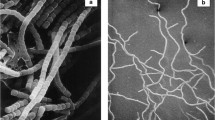
The color of the substrate mycelia of strain 431T is ochraceous salmon on Czapek agar. The strain does not form aerial mycelium. On Czapek agar, the strain produces a soluble pale lavender pigment [1]. Optimum growth occurs at 28°C. Strain 431T produces globose spores (1–1.2 µm) arranged in irregular coils within a terminal sporangium. Sporangia are globose to subglobose in shape and 6 to 14 µm in diameter [Figure 2] [1].
Sporangia release motile spores, called zoospores, after unwrapping of the membranous sheath covering sporangia (Table 1) [5]. Released zoospores have chemotactic properties for several substrate types, including sugars, amino acids, aromatic compounds and mineral ions [6,7]. Flagella of the zoospores consist of a 44-kDa flagellar protein (FliC) [5] encoded by the fliC gene, which has been applied as a molecular marker in a taxonomic study of the genus Actinoplanes [8]. Strain 431T utilizes D-xylose, L-arabinose, D-glucose, D-fructose, D-mannose, L-rhamnose, D-mannitol, sucrose, mannose, dextrin, D-galactose, D-lactose, methyl β-D-glucoside and L-rhamnose (1%, w/v), but not myo-inositol, raffinose, cellulose, adonitol, D-arabinose, D-melezitose, methyl α-D-glucoside, D-ribose or D-sorbitol (1%, w/v) [4,29]. Strain 431T also utilizes p-hydroxybenzoic acid, sodium acetate and sodium fumarate (0.1%, w/v), but not m-hydroxybenzoic acid, sodium succinate, syringic acid, or vanillin (0.1%, w/v) [4,29]. Strain 431T degrades chitin, DNA, elastin, lecithin, RNA and tyrosine (26), and also actively decomposes natural rubber [32] and flavonoids, such as quercetin, rutin and hesperidin [33]. Strain 431T produces glucose isomerase (xylose isomerase) with a molecular weight of approximately 8,000 daltons [34] and also produces 6-alkyl-4-O-dihydrogeranyl-2-methoxyhydroquinones as novel phenolic lipids [35]. Antimicrobial activity against Streptomyces murinus ISP 5091 is positive, but is negative against Aspergillus niger LIV 131, Bacillus subtilis NCIB 3610 and Staphylococcus aureus NCTC 8532 [29]. Additional physiological and drug susceptibility data are described in detail elsewhere [29].
Chemotaxonomy
The diagnostic amino acids of strain 431T are meso- and hydroxydiaminopimelic acids, and the characteristic whole-cell sugars are xylose and arabinose [29,36]. The peptidoglycan of strain 431T contains glycosylated muramic acid residues [28,37], and the major phospholipids are phosphatidylethanolamine, diphosphatidylglycerol, phosphatidylglycerol, phosphatidylinositol and phosphatidylinositol mannoside [28]. Major fatty acids (relative ratio %) are iso-C16:0 (18.3) and C19:0 (26.4) [28,29]. The predominant menaquinone is MK-9(H4) (>50%), and the minor menaquinones are MK-9(H6) and MK-10(H4) [28,29].
Genome sequencing information
Genome project history
A consortium consisting of universities, research institutions and private companies in Japan was organized to accelerate cooperative research and development utilizing genome sequences of actinomycetes. The consortium successfully accomplished the genome projects of Kitasatospora setae NBRC 14216T [38] and A. missouriensis 431T (= NBRC 102363T), as reported here. The complete genome sequences of these strains have been deposited in the INSDC database and are also available from the DOGAN genome database [39]. A summary of the genome sequencing project information of A. missouriensis is shown in Table 2.
Growth conditions and DNA isolation
A. missouriensis strain 431T (= NBRC 102363T) was grown in NBRC 231 medium (Maltose-Bennett’s Agar) at 28°C. DNA was isolated from 3.76 g of mycelial paste using the CTAB method and fragmented with the HydroShear device (Genomic Solutions), as recommended by the manufacturer.
Genome sequencing and assembly
The genome sequence of A. missouriensis 431T was determined using a whole-genome shotgun sequencing approach together with the Sanger method. DNA shotgun libraries with average insert sizes of 1.6 and 6 kbp were constructed in pUC118, and a library with an average insert size of 36 kbp was constructed in pCC1FOS (Epicentre). A total of 96,384 reads were trimmed at a threshold quality value of 20 and assembled using the Phrap and CONSED assembly tools [40,41]. A total of 141 gaps (18,532 bp in total) were closed by sequencing PCR products, and 236 low-quality regions were re-sequenced by primer walking. In the final assembly step, we confirmed that each base of the genome was sequenced from multiple clones in either both or a single direction and had Phrap quality scores of >70 and >40, respectively. For validation of the contig alignment, the Argus Optical Mapping System (OpGen, Inc.) was used.
Genome annotation
Gene prediction was performed using Glimmer3 [42] and tRNA-scanSE [43], followed by manual inspection of each translation start site using the Frameplot program [44]. Similarity search results against the NCBI nr and Pfam databases were used for functional prediction. Manual functional annotation was performed within an in-house platform developed by J.I. (Unpublished).
Genome properties
The genome of strain 431T consists of one circular chromosome with a length of 8,773,466 bp and a G+C content of 70.8% (Table 3 and Figure 3). A. missouriensis DSM 43046T was reported to contain a linear plasmid, pAM1 [33]; however, the corresponding plasmid was not found in strain 431T (= NBRC 102363T) by CHEF electrophoresis analysis. It is possible that the linear plasmid was cured from strain 431T by repeated subculturing in the laboratory. In comparison to other members of the family Micromonosporaceae, such as Salinispora and Micromonospora, A. missouriensis has a larger genome size. Of the total of 8,204 predicted genes, 8,125 were protein-coding genes and 79 were RNA genes. More than half of the protein-coding genes (4,539, 55.9%) were assigned a putative function, while the remaining predicted genes were annotated as hypothetical proteins. The distribution of genes into COGs functional categories is presented in Table 4.
References
Couch JN. Some new genera and species of the Actinoplanaceae. J Elisha Mitchell Sci Soc 1963; 79:53–70.
Skerman VBD, McGowan V, Sneath PHA. Approved lists of bacterial names. Int J Syst Bacteriol 1980; 30:225–420. http://dx.doi.org/10.1099/00207713-30-1-225
Couch JN. Actinoplanes, a new genus of the Actinomycetales. J Elisha Mitchell Sci Soc 1950; 66:87–92.
Vobis G. Actinoplanetes. In: ST Williams (eds) Bergey’s Manual of Systematic Bacteriology, Volume 4, Williams & Winkins. Baltimore, 1989, p. 2418–2450.
Uchida K, Jang MS, Ohnishi Y, Horinouchi S, Hayakawa M, Fujita N, Aizawa S. Characterization of Actinoplanes missouriensis spore flagella. Appl Environ Microbiol 2011; 77:2559–2562. PubMed http://dx.doi.org/10.1128/AEM.02061-10
Arora DK. Chemotaxis of Actinoplanes missouriensis zoospores to fungal conidia, chlamydospores and sclerotia. J Gen Microbiol 1986; 132:1657–1663.
Hayakawa M, Tamura T, Nonomura H. Selective isolation of Actinoplanes and Dactylosporangium from soil by using y-collidine as the chemoattractant. J Ferment Bioeng 1991; 72:426–432. http://dx.doi.org/10.1016/0922-338X(91)90049-M
Yamamura H, Hanawa K, Kusunoki M, Nakamura K, Nakagawa Y, Ishida Y, Hamada M, Otoguro M, Tamura T, Fujita N, et al. Variation and predicted structure of the flagellin gene in Actinoplanes species. FEMS Microbiol Lett 2011; 324:21–27. PubMed http://dx.doi.org/10.1111/j.1574-6968.2011.02380.x
Amore R, Hollenberg CP. Xylose isomerase from Actinoplanes missouriensis: primary structure of the gene and protein. Nucleic Acids Res 1989; 17:7515. PubMed http://dx.doi.org/10.1093/nar/17.18.7515
Jarling M, Cauvet T, Grundmeire M, Kuhnert K, Pape H. Isolation of mak1 from Actinoplanes missouriensis and evidence that Pep2 from Streptomyces coelicolor is a maltokinase. J Basic Microbiol 2004; 44:360–373. PubMed http://dx.doi.org/10.1002/jobm.200410403
Debono M, Merkel KE, Molloy RM, Barnhart M, Presti E, Hunt AH, Hamill RL. Actaplanin, new glycopeptide antibiotics produced by Actinoplanes missouriensis. The isolation and preliminary chemical characterization of actaplanin. J Antibiot (Tokyo) 1984; 37:85–95. PubMed http://dx.doi.org/10.7164/antibiotics.37.85
Ensign JC. Formation, properties, and germination of actinomycete spores. Annu Rev Microbiol 1978; 32:185–219. PubMed http://dx.doi.org/10.1146/annurev.mi.32.100178. 001153
Schwientek P, Szczepanowski R, Rückert C, Kalinowski J, Klein A, Selber K, Wehmeier UF, Stoye J, Pühler A. The complete genome sequence of the acarbose producer Actinoplanes sp. SE50/110. BMC Genomics 2012; 13:112. PubMed http://dx.doi.org/10.1186/1471-2164-13-112
Castresana J. Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol Biol Evol 2000; 17:540–552. PubMed http://dx.doi.org/10.1093/oxfordjournals.molbev.a026334
Lee C, Grasso C, Sharlow MF. Multiple sequence alignment using partial order graphs. Bioinformatics 2002; 18:452–464. PubMed http://dx.doi.org/10.1093/bioinformatics/18.3.452
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 2011; 28:2731–2739. PubMed http://dx.doi.org/10.1093/molbev/msr121
Liolios K, Chen IM, Mavromatis K, Tavernarakis N, Hugenholtz P, Markowitz VM, Kyrpides NC. The Genomes On Line Database (GOLD) in 2009: status of genomic and metagenomic projects and their associated metadata. Nucleic Acids Res 2010; 38:D346–D354. PubMed http://dx.doi.org/10.1093/nar/gkp848
Field D, Garrity G, Gray T, Morrison N, Selengut J, Sterk P, Tatusova T, Thomson N, Allen MJ, Angiuoli SV, et al. The minimum information about a genome sequence (MIGS) specification. Nat Biotechnol 2008; 26:541–547. PubMed http://dx.doi.org/10.1038/nbt1360
Woese CR, Kandler O, Wheelis ML. Towards a natural system of organisms: proposal for the domains Archaea,Bacteria, and Eucarya. Proc Natl Acad Sci USA 1990; 87:4576–4579. PubMed http://dx.doi.org/10.1073/pnas.87.12.4576
Garrity GM, Holt JG. The Road Map to the Manual. In: Garrity GM, Boone DR, Castenholz RW (eds), Bergey’s Manual of Systematic Bacteriology, Second Edition, Springer, New York, 2001, p. 119–169.
Stackebrandt E, Rainey F, Ward-Rainey N. Proposal for a new hierarchic classification system, Actinobacteria classis nov. Int J Syst Bacteriol 1997; 47:479–491. http://dx.doi.org/10.1099/00207713-47-2-479
Buchanan RE. Studies in the Nomenclature and classification of the Bacteria: VIII. The subgroups and genera of the Actinomycetales. J Bacteriol 1918; 3:403–406. PubMed
Buchanan RE. Studies in the nomenclature and classification of bacteria. II. The primary subdivisions of the Schizomycetes. J Bacteriol 1917; 2:155–164. PubMed
Zhi XY, Li WJ, Stackebrandt E. An update of the structure and 16S rRNA gene sequence-based definition of higher ranks of the class Actinobacteria, with the proposal of two new suborders and four new families and emended descriptions of the existing higher taxa. Int J Syst Evol Microbiol 2009; 59:589–608. PubMed http://dx.doi.org/10.1099/ijs.0.65780-0
Krasil’nikov NA. Ray fungi and Related Organisms. Actinomycetales, Akademii Nauk, Moscow, 1938, p. 272.
Koch C, Kroppenstedt RM, Rainey FA, Stackebrandt E. 16S ribosomal DNA analysis of the genera Micromonospora,Actinoplanes, Catellatospora, Catenuloplanes, Couchioplanes, Dactylosporangium, and Pilimelia and emendation of the family Micromonosporaceae. Int J Syst Bacteriol 1996; 46:765–768. PubMed http://dx.doi.org/10.1099/00207713-46-3-765
Couch JN, Bland CE. Genus I. Actinoplanes Couch 1950, 89; emend. mut. char. 1955, 153. In: Buchanan RE, Gibbons NE (eds), Bergey’s Manual of Determinative Bacteriology, Eighth Edition, The Williams and Wilkins Co., Baltimore, 1974, p. 708–710.
Stackebrandt E, Kroppenstedt RM. Union of the genera Actinoplanes Couch, Ampullariella Couch, and Amorphosporangium Couch in a redefined genus Actinoplanes. Syst Appl Microbiol 1987; 9:110–114. http://dx.doi.org/10.1016/S0723-2020(87)80063-2
Goodfellow M, Stanton LJ, Simpson KE, Minnikin DE. Numerical and chemical classification of Actinoplanes and some related actinomycetes. J Gen Microbiol 1990; 136:19–36. http://dx.doi.org/10.1099/00221287-136-1-19
Wink JM. Compendium of Actinobacteria. http://www.dsmz.de/microorganisms/wink_pdf/DSM43247.pdf
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, et al. Gene Ontology: tool for the unification of biology. Nat Genet 2000; 25:25–29. PubMed http://dx.doi.org/10.1038/75556
Jendrossek D, Tomasi G, Kroppenstedt RM. Bacterial degradation of natural rubber: a privilege of actinomycetes? FEMS Microbiol Lett 1997; 150:179–188. PubMed http://dx.doi.org/10.1016/S0378-1097(97)00072-4
Rose K, Fetzner S. Identification of linear plasmid pAM1 in the flavonoid degrading strain Actinoplanes missouriensis1 (DSM 43046). Plasmid 2006; 55:249–254. PubMed http://dx.doi.org/10.1016/j.plasmid.2005.10.003
Gong CS, Chen LF, Tsao GT. Purification and properties of glucose isomerase of Actinoplanes missouriensis. Biotechnol Bioeng 1980; 22:833–845. http://dx.doi.org/10.1002/bit.260220408
Awakawa T, Fujita N, Hayakawa M, Ohnishi Y, Horinouchi S. Characterization of the biosynthesis gene cluster for alkyl-O-dihydrogeranyl-methoxyhydroquinones in Actinoplanes missouriensis. ChemBioChem 2011; 12:439–448. PubMed http://dx.doi.org/10.1002/cbic.201000628
Horan AC, Brodsky B. Actinoplanes caeruleus sp. nov., a blue-pigmented species of the genus Actinoplanes. Int J Syst Bacteriol 1986; 36:187–191. http://dx.doi.org/10.1099/00207713-36-2-187
Kawamoto I, Oka T, Nara T. Cell wall composition of Micromonospora olivoasterospora, Micromonospora sagamiensis, and related organisms. J Bacteriol 1981; 146:527–534. PubMed
Ichikawa N, Oguchi A, Ikeda H, Ishikawa J, Kitani S, Watanabe Y, Nakamura S, Katano Y, Kishi E, Sasagawa M, et al. Genome sequence of Kitasatospora setae NBRC 14216T: an evolutionary snapshot of the family Streptomycetaceae. DNA Res 2010; 17:393–406. PubMed http://dx.doi.org/10.1093/dnares/dsq026
DOGAN genome database. http://www.bio.nite.go.jp/dogan.
Ewing B, Hillier L, Wendl MC, Green P. Base-calling of automated sequencer traces using phred. I. Accuracy assessment. Genome Res 1998; 8:175–185. PubMed
Ewing B, Green P. Base-calling of automated sequencer traces using phred. II. Error probabilities. Genome Res 1998; 8:186–194. PubMed
Delcher AL, Bratke KA, Powers EC, Salzberg SL. Identifying bacterial genes and endosymbiont DNA with Glimmer. Bioinformatics 2007; 23:673–679. PubMed http://dx.doi.org/10.1093/bioinformatics/btm009
Lowe TM, Eddy SR. tRNAscan-SE: a program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res 1997; 25:955–964. PubMed
Ishikawa J, Hotta K. FramePlot: a new implementation of the frame analysis for predicting protein-coding regions in bacterial DNA with a high G + C content. FEMS Microbiol Lett 1999; 174:251–253. PubMed http://dx.doi.org/10.1111/J.1574-6968.1999.tb13576.x
Author information
Authors and Affiliations
Corresponding author
Additional information
Deceased July 12th, 2009
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Yamamura, H., Ohnishi, Y., Ishikawa, J. et al. Complete genome sequence of the motile actinomycete Actinoplanes missouriensis 431T (= NBRC 102363T). Stand in Genomic Sci 7, 294–303 (2012). https://doi.org/10.4056/sigs.3196539
Published:
Issue Date:
DOI: https://doi.org/10.4056/sigs.3196539