Abstract
The present study identified subgroups based on inhibitory and reward activation, two key neural functions involved in risk-taking behavior, and then tested the extent to which subgroup differences varied by age, sex, behavioral and familial risk, and substance use. Participants were 145 young adults (18–21 years old; 40.0% female) from the Michigan Longitudinal Study. Latent profile analysis (LPA) was used to establish subgroups using task-based brain activations. Demographic and substance use differences between subgroups were then examined in logistic regression analyses. Whole-brain task activations during a functional magnetic resonance imaging go/no-go task and monetary incentive delay task were used to identify beta weights as input for LPA modeling. A four-class model showed the best fit with the data. Subgroups were categorized as: (1) low inhibitory activation/moderate reward activation (39.7%), (2) moderate inhibitory activation/low reward activation (22.7%), (3) moderate inhibitory activation/high reward activation (25.2%), and (4) high inhibitory activation/high reward activation (12.4%). Compared with the other subgroups, Class 2 was older, less likely to have parental alcohol use disorder, and had less alcohol use. Class 4 was the youngest and had greater marijuana use. Classes 1 and 3 did not differ significantly from the other subgroups. These findings demonstrate that LPA applied to brain activations can be used to identify distinct neural profiles that may explain heterogeneity in substance use outcomes and may inform more targeted substance use prevention and intervention efforts.
Similar content being viewed by others
Substance use increases during adolescence and peaks in young adulthood (Miech et al., 2019; Schulenberg et al., 2019). Experimental substance use is normative among youth, due in part to greater opportunities and social incentives for use (Schulenberg et al., 2016). However, heavy use contributes to negative outcomes, such as risky sexual behavior (Ritchwood et al., 2015) and higher rates of mental health problems (Conway et al., 2018). Early substance use may confer long-term risk, as the majority of adults who meet criteria for a substance use disorder began using substances as adolescents (Grant & Dawson, 1997; Richmond-Rakerd et al., 2016). A better understanding of factors differentiating youth who do versus do not display problematic substance use is crucial for improving public health.
A substantial body of research has demonstrated that differences in brain function, specifically inhibitory control and reward responsivity, may underlie individual differences in substance use behaviors. Throughout adolescence and young adulthood, the brain undergoes dramatic changes to its structure, connectivity, and physiology (Casey & Jones, 2010; Gogtay et al., 2004; Spear, 2004) that are thought to make these age groups particularly susceptible to risk-taking behaviors. Heightened risk-taking often is explained through dual-systems models of brain development (Shulman et al., 2016; Steinberg et al., 2008), which attribute risk behaviors observed in youth to two complimentary systems: 1) a subcortical socioemotional-reward system—including the striatum and amygdala—characterized by responsivity to reward and novelty; and 2) a cortical cognitive system—including the prefrontal cortex—characterized by impulse control and emotion regulation. The subcortical reward system matures much earlier compared with the prefrontal cognitive control system, which is unable to fully dampen subcortical impulses during this time period.
Increased impulsivity and reward-seeking are thought to contribute to a higher risk for substance use (Dick et al., 2010; Quinn & Harden, 2013). For example, individuals with substance use disorders have been found to display poorer behavioral performance on measures of inhibitory control relative to healthy individuals (Wright et al., 2014). Prospective neuroimaging studies have used the go/no-go paradigm, in which individuals must respond quickly to stimuli but inhibit their response on a subset of trials, to probe whether abnormalities in the neural correlates of inhibitory control are associated with substance use in youth. Youth who initiate heavy substance use during adolescence have generally been found to display reduced activation in prefrontal and parietal brain regions during no-go (inhibition) trials earlier in life (Heitzeg, Nigg, et al., 2014a; Mahmood et al., 2013). Differences in inhibitory brain activation during the go/no-go task, specifically in frontoparietal regions, have been documented in youth who report heavy alcohol use compared with nondrinkers (Norman et al., 2011; Wetherill, Castro, et al., 2013a; Wetherill, Squeglia, et al., 2013b). Linkages between nicotine use and deficits in inhibitory control also have been observed (Luijten et al., 2014; Smith et al., 2014, for review), including associations between heavy nicotine use and lower activation in regions involved in inhibitory control (e.g., bilateral middle frontal gyrus (MFG), supplemental motor area (SMA); Galván et al., 2011). In terms of marijuana use, studies have found greater activation related to inhibitory processing in frontoparietal and cerebellar regions in youth who use marijuana compared with nonusers (Smith et al., 2011; Tapert et al., 2007). Although the aforementioned studies included youth who predominately used alcohol or marijuana respectively, other substance use was not exclusionary, and polysubstance use was relatively common among the samples. Thus, specific effects of substance type are difficult to parse.
Studies attempting to characterize reward system functioning in the context of substance use risk have relied heavily on the monetary incentive delay task (MID; Knutson et al., 2000) during functional magnetic resonance imaging (fMRI), which categorizes reward processing into an anticipatory phase and a consummatory phase. The anticipatory component of the task robustly activates the ventral striatum (VS), which is a central component of the brain’s dopaminergic reward system. With regard to substance use risk, there is an ongoing question as to the direction of effect. Some argue for a “reward deficiency” model, wherein activation to reward anticipation is reduced in those who are at risk for problem substance use (Blum et al., 2000; Whelan et al., 2014). Others instead have found heightened activation in the VS to be associated with later alcohol problems (Heitzeg, Villafuerte, et al., 2014b) and early substance use initiation in substance-naïve children (Cope et al., 2019). Furthermore, nicotine has been found to increase activation in reward-related areas, such as the VS and anterior insula (Moran et al., 2018). Nicotine administration creates an influx of dopamine to the nucleus accumbens (NAcc), and in turn, an effect on anticipatory reward responding (Wang et al., 2020). There also are questions in the literature pertaining to substance-specific effects on reward responding. A cross-sectional study compared groups of alcohol-, tobacco-, and marijuana-only using, nonusing, and polysubstance using adolescents, finding that the tobacco-only group uniquely showed blunted anticipatory reward activation (Karoly et al., 2015). More recently, work using the Michigan Longitudinal Study (MLS) sample found a significant longitudinal association between greater marijuana use and reduced anticipatory reward activation in the NAcc over time when controlling for concomitant tobacco and alcohol use (Martz et al., 2016). Important methodological differences may account for these seemingly conflicting findings. Specifically, while Martz et al. (2016) looked at longitudinal associations between substance use and anticipatory reward responding within-subjects, Karoly et al. (2015) conducted a cross-sectional study comparing groups of participants based on their substance use behavior in relation to reward activation. Additional research is needed to better understand group-level variation and substance-specific associations with inhibitory and reward activation.
Although the current literature suggests that inhibitory control and reward responsivity each play a role in substance use vulnerability, it remains unclear how the interplay between these two systems—as posited by dual-systems models of brain development—is related to varying levels of drug and alcohol use displayed by youth. Examining individual differences in brain function associated with both inhibitory control and reward responsivity may help identify youth at risk for heavy substance use. Latent profile analysis (LPA), a person-centered approach used to identify latent subgroups derived from multiple observations across different dimensions (Lanza & Rhoades, 2013), may be useful to identify neural profiles based on both inhibitory control and reward activation.
Unlike confirmatory factor analysis (CFA), a method that uses measurement items to determine latent constructs, LPA uses individual data points to identify groupings of people (Ferguson et al., 2020). The approximate equivalent of a factor loading in LPA is probability of class membership. Whereas CFA uses multiple variables to find homogeneity (i.e., a common factor), LPA uses indicator variables to find heterogeneity (i.e., different classes) among participants. LPA also differs from other grouping analyses, such as cluster analysis, by using a model-based, probabilistic approach. Compared with variable-centered approaches, LPA does not assume that the study sample shows a similar pattern of behavior (or neural activation). Group-based means provide useful information, but only if observed data display minimal variance across the study sample. Individual differences often are viewed as statistical noise rather than variables of interest (Foulkes & Blakemore, 2018). Thus, LPA is a useful tool to uncover subgroups that group averages may fail to capture. Indeed, LPA has been identified as a robust tool to characterize heterogeneity not only in children with ADHD (Fair et al., 2012; van Hulst et al., 2015) but also in relation to other behaviors, such as substance use (Evans-Polce et al., 2016).
Furthermore, LPA is relatively agnostic in terms of data entered into the model to determine class groupings; data can include parameter estimates, means, or even beta weights from brain activation during fMRI tasks. Identifying subgroups based on brain function has important implications for research aimed at better understanding correlates of psychological phenomena or behaviors of interest and related clinical efforts. A strength of LPA over other approaches to examining brain-behavior associations, such as regression, is that it can provide a data-driven approach to test for latent subgroupings across multiple tasks. Specifically related to inhibitory control and reward responsivity, LPA has been used to create neuropsychological profiles based on behavioral performance during go/no-go and MID tasks in individuals with attention deficit hyperactivity disorder (ADHD) (van Hulst et al., 2015). As an exploratory follow-up to the van Hulst et al. (2015) study, Lecei et al. (2019) identified latent subgroups based on functional imaging data within a relatively small sample of children reporting ADHD symptoms (N = 56). However, few studies to-date have used LPA with neuroimaging data. This gap in the literature is concerning given findings suggesting individual differences in brain function are likely linked to various behavioral outcomes (Anandakumar et al., 2018; Foulkes & Blakemore, 2018; Hariri, 2009).
The present study extends the application of LPA to imaging data by using an emerging adult sample and focusing specifically on substance use in participants from the MLS, a longitudinal, developmental study of the emergence of risk for alcohol and other substance use disorder (AUD; SUD). Two key objectives of this study were to: (1) conduct LPA with mean beta weights from regions showing significant task activation in go/no-go and MID tasks, respectively; and (2) examine demographic and substance use variation in adolescence as predictors of latent subgroup membership. Substance use measures examined in the present study were alcohol, marijuana, and cigarette use, due to these substances being the most commonly used drugs of abuse in adolescents and young adults (Schulenberg et al., 2019) and also the most prevalent substances among the study sample. Youth are typically opportunistic rather than specialists in terms of their substance use behavior (Maggs et al., 2011; Schulenberg et al., 2016), and yet there is some evidence for specific effects of substance type on brain activation (Coronado et al., 2020; Karoly et al., 2015). Thus, associations between profiles of brain activation and substance use were examined first in models including all substance use types together to determine whether there were effects of each substance over and above the others, and then in substance-specific models to account for the potential for collinearity to mask findings for a particular substance.
Methods
Participants
Participants were 145 right-handed 18- to 21-year-olds (mean age 19.80; SD = 1.22; 40.0% female) from the MLS, a community-based, longitudinal study of families with parental AUD (FH+) and a contrast sample of FH− families (Zucker et al., 2000). Families who had a child with symptoms of fetal alcohol syndrome were excluded. Exclusion criteria for the neuroimaging protocol were neurologic, acute, uncorrected, or chronic medical illness; treatment with centrally active medications either currently or within the last 6 months; history of psychosis or schizophrenia in first-degree relatives; or the presence of Axis I psychiatric or developmental disorders. Conduct disorder, ADHD, substance use disorder, and history of, or current unmedicated depression or anxiety were not exclusionary due to their association with AUD risk. Diagnosis was determined using the Diagnostic Interview Schedule (Version IV; Robins et al., 2000). Participants who were taking medication for ADHD were required to stop taking their medication at least 48 hours before fMRI scanning. A five-panel multidrug urine screen occurred before each scan. Participants who tested positive for marijuana but self-reported abstinence within 48 hours before the scan were not excluded due to Δ9-tetrahydrocannabinol metabolites being detectable in urine for a week or longer. Self-reported drug use within the past 48 hours and/or a positive test for drugs other than marijuana were exclusionary. All participants provided written consent, which was approved by the University of Michigan Medical School Institutional Review Board.
Participant Measures
Substance use
The self-report Drinking and Drug History Form (DDH; Zucker et al., 1990; Zucker et al., 1994) was conducted annually beginning at age 11 years to assess quantity, frequency, and variability of alcohol consumption, frequency of drug use, and consequences associated with the use of these substances. Previous work using the DDH supports its validity (Noll et al., 1992). To measure substance use before the fMRI scan session, cumulative alcohol, marijuana, and cigarette use from age 11 years until the date of the scan were computed. Cumulative alcohol use was computed by taking the sum of drink volume (number of drinking days per month and drinks per day across different types of alcohol) at each assessment age (Weiland et al., 2014; Weiland et al., 2017). The MLS originated as a study with a predominant focus on alcohol use, and as a result, there is greater precision for alcohol-related questions compared to other types of substance use. Marijuana and cigarette use were assessed via frequency of use report, rather than precise number of use occasions. For cumulative marijuana use, participants were asked to report the number of occasions they used marijuana over the past 12 months. Response options ranged from 0 = no occasions to 8 = 1,000+ occasions. For cumulative cigarette use, participants were asked to report their regularity of using cigarettes over the past 12 months. Response options ranged from 0 = never to 4 = regularly now. Due to the high variance of substance use among study participants, these variables were log transformed to improve the normality of the response distribution. See Supplemental Materials for detailed information on calculations for cumulative substance use variables.
Externalizing behavior
Due to the association between early externalizing behavior and later substance use outcomes (Zucker et al., 2011), mean externalizing behavior from ages 9 to 14 years was assessed using T-scores from the aggression and delinquency subscales of the Youth Self Report (YSR; Achenbach, 1991).
Imaging Tasks
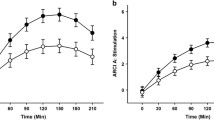
An event-related fMRI go/no-go task (Durston et al., 2002; Hardee et al., 2014; Heitzeg, Nigg, et al., 2014a) was used to measure blood oxygen level-dependent (BOLD) response associated with inhibitory control. Participants were instructed to respond as quickly and accurately as possible to target stimuli (letters other than “X”) during go trials via button press, but to withhold their response to infrequent non-target stimuli (“X”) during no-go trials. Stimulus duration was 500 ms, followed by 3,500 ms of a fixation cross. There were a total of 5 runs of 49 trials, each lasting 3.5 min. To focus on successful inhibitory control, analyses in the present study were conducted using the contrast for correct rejection (correct no-go trials) versus go trials. Hit accuracy, reaction times to hits, and correction rejection rates were calculated as task performance measures. Observed hit rates indicated that performance was well-above chance (>63%) for all individuals in the study.
To assess BOLD response during reward anticipation, participants performed a modified version of the MID task (Knutson et al., 2001; Martz et al., 2016; Yau et al., 2012). An incentive cue was displayed for 2,000 ms during each trial. Cues consisted of a large reward (win $5.00), large loss (lose $5.00), small reward (win $0.20), small loss (lose $0.20), or neutral condition (no money at stake). A fixation cross was then shown for 2,000 ms, followed by a variable-duration target, during which participants were instructed to press a button as quickly as possible. Pressing the button while the target was on the screen signified a correct response. Participants were then shown feedback indicating whether they won money, failed to win money, lost money, avoided losing money, or no money was at stake. Trials were presented in pseudorandom order in two 5 min runs, each of which included 20 gain trials (10 large reward, 10 small reward), 20 loss trials (10 large loss, 10 small loss), and 10 neutral trials. Analyses were conducted using the contrast for combined large and small reward cue anticipation versus neutral cue anticipation trials. Hit rates and reaction times for reward anticipation and neutral trials were calculated as task performance measures.
fMRI Acquisition
Whole-brain BOLD functional images were acquired on a 3.0-T GE Signa scanner (GE Healthcare, Milwaukee, WI) using a T2*-weighted single-shot combined spiral in–out sequence (Glover & Law, 2001) with the following parameters: repetition time (TR) = 2,000 ms, echo time (TE) = 30 ms, flip angle = 90°; field-of-view (FOV) = 200 mm; matrix size = 64 x 64; in-plane resolution 3.12 x 3.12 mm; and slice thickness = 4 mm. A high-resolution, anatomic, T1-weighted scan was obtained for spatial normalization (three-dimensional spoiled gradient-recalled echo, TR = 25 ms, min TE, FOV = 25 cm, 256 x 256 matrix, slice thickness = 1.4 mm).
fMRI Preprocessing
Functional images were reconstructed using an iterative algorithm (Noll et al., 2005; Sutton et al., 2003). Subject head motion and slice-acquisition timing were corrected using FSL 4.0 (Analysis Class, FMRIB, Oxford, United Kingdom; Jenkinson et al., 2002). Analysis of estimated motion parameters confirmed that overall head motion within each run did not exceed 3-mm translation or 3° rotation in any direction. All remaining image processing and statistical analysis were completed using statistical parametric mapping (SPM8; Wellcome Trust Center for Neuroimaging, London, United Kingdom). Functional images were spatially normalized to a standard stereotactic space as defined by the Montreal Neurological Institute. A 6-mm, full-width, half-maximum Gaussian spatial smoothing kernel was applied to improve signal-to-noise ratio and to account for individual differences in anatomy. One-sample t-tests were completed in SPM to examine whole-brain task activation (go/no-go: correct rejection>go; MID: reward anticipation>neutral). For both go/no-go and MID tasks, motion parameters and white matter signal intensity were modeled as nuisance regressors to remove residual motion artifacts and capture nontask-related noise.
Analytic Plan
Three steps comprised the analytic plan of the present study. First, ROIs were selected based on significant whole-brain task activation during a single session of the go/no-go task and the MID task. Beta weights for regions showing significant task activation (Figure 1) were extracted using MarsBaR (Brett et al., 2002). Because ROIs from two separate tasks were input into LPA, a level of thresholding was selected that was equal across both tasks. Thresholding also was determined by ROIs that were comprised of discreet brain regions showing adequate cluster size and that were corrected for multiple comparisons to control for false positives.
Whole-brain task activations during go/no-go and monetary incentive delay tasks. Significant whole-brain task activations during the (A) correct rejection vs. go contrast from the go/no-go task, shown by coordinates x = 40, y = 42, z = 22 and the (B) combined large and small reward anticipation vs. neutral contrast from the monetary incentive delay task, shown by coordinates x = 0, y = −4, z = −4. Regions are significant at a family-wise corrected threshold of p < 0.0000005 and voxel extent >10. Coordinates are in Montreal Neurological Institute (MNI) space, and the color bar represents t-values
Second, LPA was performed in Mplus version 7.4 (Muthén & Muthén, 2015) using extracted ROIs. LPA involves iteratively running a succession of models, beginning with a one-class model, then two-class model, then three-class model, and so on. Both theoretical and statistical considerations are used to determine the appropriate number of classes (Collins & Lanza, 2010). Lower values for Bayesian Information Criteria (BIC) and Akaike Information Criteria (AIC) typically suggest better model fit. However, this strategy for model selection is somewhat exploratory, in that substantive or interpretation concerns also guide decision-making. These concerns also led to the use of a threshold of 5% as the smallest class size in determining the number of classes (Ferguson et al., 2020 ). As is done in factor analysis, scree plots provide a helpful visualization of where BIC values begin to display diminishing value for each additional class. The class number at the “elbow” of the plot where BIC values level out provides another indicator of best fitting number of classes (Nylund-Gibson & Choi, 2018). Regarding interpretability, it is often best to select a model that shows greater differentiation in data patterns (Nylund et al., 2007).
Third, estimated class assignments were exported into SPSS v24 to examine descriptive data and predictors of each latent class. Output of LPA provides a posterior probability assigned to each individual, with values closer to 1 indicating greater confidence in individual class assignments. Predictors of each class were examined using multivariable logistic regression analyses with latent class as the dependent variable. Analyses were conducted first with all substances (i.e., alcohol, marijuana, cigarettes) modeled together to test for potential effects of certain substances over and above other types. Second, each substance type was included in separate models to test for influences of particular substances on class membership.
Results
Latent Profile Analysis
LPA was conducted using extracted beta weights from eight clusters from the go/no-go task and seven clusters from the MID task (Table 1), resulting in fifteen variables corresponding to the mean extracted beta weights entered into LPA models. A threshold of p < 0.0000005, family-wise error (FWE) corrected for multiple comparisons, >10 voxel extent, was used for extractions. Model fit indices showed that the 4-class model was superior (Figure 2), as indicated by the best fit to the data when comparing models with increasing numbers of classes (Supplemental Table 4) and examination of the scree plot of BIC values (Supplemental Figure 1). Class 1 was comprised of 39.68% of the total sample and was qualitatively characterized by low BOLD activation during inhibitory control and moderate activation during reward anticipation compared to the other three classes. Class 2 (22.72%) showed moderate inhibitory control and low reward activation, whereas Class 3 (25.20%) had moderate inhibitory control and high reward activation. Class 4 (12.40%) had the highest inhibitory control and reward anticipation compared to the other classes. Labels to describe levels of activation across classes (e.g., low, moderate, high) are strictly descriptive rather than quantitative in order to aid in interpreting characterizations of each profile.
Latent profiles of neural activations during go/no-go and monetary incentive delay tasks. Four-class model fit indices: AIC = 5164.53, BIC = 5396.71, Entropy = 0.92; Percentage of total sample (N = 145): Class 1: 39.68%; Class 2: 22.72%; Class 3: 25.20%; Class 4: 12.40%; CR = correct rejection; MID = monetary incentive delay; MFG = middle frontal gyrus; SMA = supplemental motor area; OCC = occipital cortex
Class Comparisons
Descriptive data across the four classes are shown in Table 2. Behavioral performance for go/no-go and MID tasks in each class is reported in Table 3. The MID task is designed so that success rate is individually calibrated in order to achieve comparable behavioral performance across subjects. For reaction time, however, results from Tukey’s HSD test to examine class differences showed that Class 4 was significantly faster during reward trials compared to Class 1 (p = 0.013). For the go/no-go task, Class 3 had a significantly higher correct rejection rate compared to Class 1 (p = 0.049) during the go/no-go task. There were no other significant differences between classes in relation to behavioral task performance.
Each class was then compared to the remaining three groups together in four separate multivariable logistic regression models (e.g., Class 1 vs. 2, 3, 4; Table 4). Class was the dependent variable, and scan age, sex, parental AUD, externalizing behavior, cumulative alcohol use, cumulative marijuana use, and cumulative cigarette use were independent variables. Class 2 was significantly older and had a lower likelihood of parental AUD. Class 4 was younger and more likely to have greater marijuana use. Results from regression analyses showed that there were no significant predictors differentiating Classes 1 and 3 from all other classes.
Alcohol use was highly correlated with both marijuana use (r = 0.58, p < 0.001) and cigarette use (r = 0.57, p < 0.001). Marijuana and cigarette use were also highly correlated (r = 0.58, p < 0.001). To account for potential effects of multicollinearity, we conducted separate multivariable logistic regression models for alcohol, marijuana, and cigarette use (Table 5). In the alcohol-only model, Class 2 was older, less likely to be FH+, and less likely to use alcohol compared with the other classes. Class 4 was younger and had higher alcohol use. In the marijuana-only model, Class 2 was older and less likely to be FH+. Class 4 was younger and more likely to have greater marijuana use. In the cigarette-only model, there were no class differences in cigarette use, but Class 2 was less likely to be FH+. For all substance use specific models, there were no significant differences in any predictors between Class 1 versus all other classes and Class 3 versus all other classes.
Discussion
The present study identified heterogeneous subgroups based on brain activation associated with inhibitory control and reward anticipation—two neural functions integral to dual-systems models of brain development and underlying substance use behavior—and then examined the extent to which age, sex, behavioral and familial risk, and substance use varied among latent classes. Importantly, findings from the present study demonstrate the potential utility of using LPA with brain imaging data to differentiate subgroups linked to addiction-related behaviors. Among the four distinct classes identified, Classes 2 and 4 showed the most striking differences between the other classes in regression analyses. Class 2 was older, less likely to be FH+, and had lower cumulative alcohol use, whereas Class 4 was younger and had greater alcohol and marijuana use. The lack of significant subgroup differences in regression models that included cigarette use was likely due to the relatively high incidence rate of cigarette use in the MLS sample. Class 4 was arguably the highest risk group and Class 2 the lowest risk group compared with the other classes; as such, these two classes are the focus of the discussion.
Despite displaying greater substance use, Class 4 showed the greatest inhibitory activation compared to the other classes during the go/no-go task, particularly in frontoparietal regions (e.g., bilateral MFG, precentral gyrus, insula, SMA). This finding is consistent with prior work showing an association between marijuana use and greater inhibitory activation in these regions (Smith et al., 2011; Tapert et al., 2007), especially considering that Class 4 had higher rates of marijuana use compared to the other classes. However, a great extent of inhibitory control activation may not necessarily equate to stronger impulse control, which is generally believed to be protective against substance use. Instead, inhibitory activation displayed by Class 4 may indicate a compensatory neural effort to perform the go/no-go task successfully rather than an elevated extent of top-down cognitive control. Prior research using MLS data has found significant differences between low- and high-risk substance use groups in brain activation despite equivocal behavioral task performance (Hardee et al., 2014; Heitzeg et al., 2010), suggesting that different brain regions may be recruited—and to varying extents—in order to perform the task successfully. For example, Hardee et al. (2014) found that FH+ youth showed a developmental increase in brain activation within regions involved in response inhibition from childhood to adolescence, whereas FH− youth had decreased activation in these regions over the same time span. Except for one difference in go/no-go task performance between Classes 1 and 3 and in MID task performance between Classes 1 and 4, no other significant differences in task performance between classes were not detected in the present study. However, Class 4 showed the fastest mean reaction times for both reward and neutral targets during the MID task and for hits during the go/no-go task, indicating a tendency toward impulsive responding that is consistent with a behavioral pattern of higher substance use.
In the same vein, it also is possible that the relatively high level of inhibitory control activation in Class 4 may be a result of heavy substance use, reflecting increased engagement of inhibitory circuitry in order to successfully withhold a prepotent response. Wetherill, Squeglia, et al. (2013b) assessed a sample of adolescents before and after substance use initiation and found that future heavy substance users had blunted activation during no-go trials of a go/no-go task at the baseline scan in frontoparietal regions, which were also areas that showed inhibitory activation in the present study. These results also mirror findings by Heitzeg, Nigg, et al. (2014a) that found lower inhibitory activation in the left MFG during late childhood predicted problem substance use in adolescence. However, when youth from the Wetherill, Squeglia, et al. (2013b) study were assessed three years later and had transitioned into heavy substance use, they showed greater activation compared to nondrinking controls, supporting the possibility of compensatory neural function to perform the inhibitory control task successfully. Also related to compensatory neural function, Mahmood et al. (2013) described unexpected activation in the supramarginal gyri specifically in the highest frequency substance users as a possible indicator of a “verbally mediated calculation strategy” used during the go/no-go task. Coinciding with findings reported by Mahmood et al. (2013), the present study found greater activation in bilateral supramarginal regions among participants in Class 4. In sum, evidence from previous studies and the present study highlight the importance of considering both maturational changes and potential effects of cumulative use that may impact brain function underlying inhibitory control.
Class 4 not only had greater brain activation involved in inhibitory control compared with the other classes, but also had heightened activation in anticipation of monetary rewards. Given the greater likelihood of Class 4 to engage in marijuana use and alcohol use compared with the other classes, this level of reward responsivity, especially in the VS, may reflect increased dopaminergic response to rewarding stimuli. Class 3 showed a similarly high level of VS activation. Of note, Class 3 had the next highest mean rates of marijuana use after Class 4. Nestor et al. (2010) found that young adults who used marijuana showed heightened activation in the VS during the reward anticipation condition of the MID task. Considering that approximately 22% of individuals in Class 4 tested positive for marijuana during their urine drug screen (but were still eligible for the scan because they self-reported use at least 48 hours beforehand) it is possible that recent marijuana use contributed to this level of reward activation. Indeed, marijuana has been found to increase neural blood flow for up to a month after last use (Herning et al., 2005).
Reward sensitivity may even exist prior to the onset of substance use, as evident by findings from recent studies using MLS data. Cope et al. (2019) reported that greater activation in the NAcc during monetary reward anticipation measured in childhood predicted substance use initiation in early adolescence. Thus, continued substance use may disrupt the brain’s natural ability to produce dopamine and respond to non-drug rewards. In a cross-lagged analysis spanning ages 20 to 24, marijuana use was associated with blunted NAcc activation during the anticipation of monetary rewards, suggesting a decrease in reward responsivity to non-drug rewards (Martz et al., 2016). Although results from the present study showed an association between greater substance use—particularly marijuana use—and elevated anticipatory reward responding in the VS, it is important to note that LPA was conducted with cross-sectional brain imaging data. It is possible that with age and continued substance use, reward-related brain activation in Class 4 may decrease over time.
Class 2 was the only subgroup identified in the present study to show lower reward activation compared to inhibitory control activation. Furthermore, compared to the other classes, Class 2 displayed the lowest activation during reward anticipation. This pattern may be associated with lower substance use, which is consistent with findings here. In addition to being older, Class 2 also was less likely to have parental AUD compared with the other classes. In relation to anticipatory reward activation, FH+ individuals tend to show either no significant differences or blunted activation compared to FH− individuals. Consistent with the reward deficiency hypothesis, Andrews et al. (2011) found that FH+ individuals showed lower activation in the VS, orbitofrontal cortex, and insula during reward anticipation compared to FH- individuals. In contrast, Müller et al. (2015) found no differences between FH+ and FH− individuals both in analyses focused specifically on the VS and in exploratory whole-brain analyses. Similarly, Bjork et al. (2008) reported no FH differences in anticipatory reward responding in the VS. Our results, however, showed that Class 2 was less likely to be FH+ and had lower activation across reward circuitry compared to the other classes. A possible explanation for this discrepancy with prior literature is that the present study did not directly compare FH+ and FH− individuals. While Class 2 did indeed have lower odds of being FH+, more than half of Class 2 were classified as FH+ due to the entire study sample being enriched for parental AUD. Results from the present study suggest that even within a predominately FH+ sample, there appears to be heterogeneity in both inhibitory control and anticipatory reward activation, as well as substance use behavior.
Strengths and Limitations
A key strength of the present study is its application of LPA to functional brain imaging data measured through go/no-go and MID tasks. This work has important implications for research on dual-systems models of brain development by identifying subgroups based on inhibitory and reward activation. Linkages between substance use behavior and brain function based on profiles of activation across multiple tasks may help researchers better characterize individuals based on heterogeneity across dimensions of risk (e.g., low levels of inhibitory control coupled with elevated reward responsivity). This analytic approach differs from studies examining differences in brain function between predefined subgroups classified by their substance use (e.g., Karoly et al., 2015), although both approaches have merit.
The only previous studies to include inhibitory control and reward responsivity in the same latent modeling approach were focused on childhood samples. Van Hulst et al. (2015) and Lecei et al. (2019) focused on ADHD symptoms. Although potentially limited in generalizability, Lecei et al. (2019) demonstrated the feasibility of identifying latent subgroups based on brain activation during go/no-go and MID tasks. Thus, other strengths of the present study are its use of brain activation during emerging adulthood, when substance use is most prevalent, and its inclusion of prospective externalizing and substance use data from late childhood through adolescence to test for potential influences of these earlier indicators on brain function. However, additional work in larger, longitudinal samples with a wider range of substance use behavior is needed to better assess substance-specific effects on the brain.
One limitation of the present study is generalizability. MLS is relatively homogenous in race/ethnicity, sex, and parental AUD, so results may be limited in their generalizability to other populations. It is important to note that the MLS was originally designed to focus predominately on alcohol use. As such, questions pertaining to alcohol use are much more detailed regarding type, quantity, and frequency of use compared with marijuana and cigarette use. This may impact the extent to which comparisons can be made across substances. Furthermore, ROI selection in the present study was based on whole-brain task activation, which was then examined through LPA. This approach was chosen to extract an optimal amount of information given the relatively small sample. However, as a data-driven, model-estimation approach, class estimates found through LPA are dependent upon the sample from which they are derived. Using LPA to examine latent subgroups based on neuroimaging data should be replicated in larger and more diverse samples. Even though LPA modeling is not strictly limited to large sample sizes, a greater N allows for improved model stability and accuracy in detecting small classes (Nylund-Gibson & Choi, 2018). It also is possible that individual variation in brain activation may impact which ROIs show significant activation in group-level main effects maps. However, an advantage of LPA applied to fMRI data is that it allows for individual variation to be identified through latent classes based on group-level activation.
Another limitation of the current work is that imaging data was only available for this sample beginning approximately at age 18 years and assessed at a single time point. Additional work, such as the Adolescent Brain Cognitive Development (ABCD) study that begins at ages 9 and 10 years and then follows participants through the onset and progression of substance use, will be useful to disentangle aspects of brain function impacting substance use vulnerability and later impacts of substance use on the brain. In the present study, substance use data were collected over time, but brain imaging data were conducted cross-sectionally. Thus, causation cannot be inferred from our results.
There are limitations of conducting analyses of independent variables predicting class membership based on posterior probabilities. The BCH method in Mplus is often used to conduct regression analyses predicting class membership, because this method accounts for classification error, and latent class measurement parameters are held fixed (Nylund-Gibson & Choi, 2018). However, using extracted class assignments from posterior probabilities as a categorical grouping variable is appropriate if entropy of the model is adequate (i.e., >0.80; Ferguson et al., 2020). The entropy of the final model used in the present study was 0.92, indicating confidence in classification certainty.
Conclusions
Using a novel analytical approach, the present study applied LPA to brain imaging data in a sample of emerging adults. Based on the theoretical perspective of the dual-systems model of brain development and subsequent critiques that this perspective may not capture potential heterogeneity within a given age group, we sought to identify subgroups based on brain activation during inhibitory control and reward tasks. Indeed, we found four distinct classes with varying activation patterns and also differences in age and substance use behaviors across adolescence. Examining how inhibitory control and reward circuitry are associated with substance use behavior has the potential to help uncover individual differences in neural function associated with differential risk-outcomes, which may be beneficial to better understand at-risk groups and create more individualized intervention and prevention efforts.
References
Achenbach, T. M. (1991). Manual for the Youth Behavior Self-Report and 1991 Profile. University of Vermont Burlington.
Anandakumar, J., Mills, K. L., Earl, E. A., Irwin, L., Miranda-Dominguez, O., Demeter, D. V., ... & Fair, D. A. (2018). Individual differences in functional brain connectivity predict temporal discounting preference in the transition to adolescence. Developmental Cognitive Neuroscience, 34, 101–113.
Andrews, M. M., Meda, S. A., Thomas, A. D., Potenza, M. N., Krystal, J. H., Worhunsky, P., et al. (2011). Individuals family history positive for alcoholism show functional magnetic resonance imaging differences in reward sensitivity that are related to impulsivity factors. Biological Psychiatry, 69, 675–683.
Bjork, J. M., Knutson, B., & Hommer, D.W. (2008). Incentive-elicited striatal activation in adolescent children of alcoholics. Addiction, 103, 1308–1319.
Blum, K., Braverman, E. R., Holder, J. M., Lubar, J. F., Monastra, V. J., Miller, D., et al. (2000). The reward deficiency syndrome: A biogenetic model for the diagnosis and treatment of impulsive, addictive and compulsive behaviors. Journal of Psychoactive Drugs, 32, 1–112.
Brett, M., Anton, J-L., Valabregue, R., & Poline, J. B. (2002). Region of interest analysis using an SPM toolbox. Presented at the 8th International Conference on Functional Mapping of the Human Brain; June 2–6, 2002; , Japan.
Casey, B. J., & Jones, R. M. (2010). Neurobiology of the adolescent brain and behavior: implications for substance use disorders. Journal of the American Academy of Child & Adolescent Psychiatry, 49, 1189–1201.
Collins, L. M., & Lanza, S. T. (2010). Latent class and latent transition analysis: With applications in the social, behavioral, and health sciences. New York, NY: Wiley.
Conway, K. P., Green, V. R., Kasza, K. A., Silveira, M. L., Borek, N., Kimmel, H. L., et al. (2018). Co-occurrence of tobacco product use, substance use, and mental health problems among youth: findings from wave 1 (2013–2014) of the population assessment of tobacco and health (PATH) study. Addictive Behaviors, 76, 208–217.
Cope, L. M., Martz, M. E., Hardee, J. E., Zucker, R. A., & Heitzeg, M. M. (2019). Reward activation in childhood predicts adolescent substance use initiation in a high-risk sample. Drug & Alcohol Dependence, 194, 318–325.
Coronado, C., Wade, N. E., Aguinaldo, L. D., Mejia, M. H., & Jacobus, J. (2020). Neurocognitive correlates of adolescent cannabis use: An overview of neural activation patterns in task-based functional MRI studies. Journal of Pediatric Neuropsychology, 6(1), 1–13.
Dick, D. M., Smith, G., Olausson, P., Mitchell, S. H., Leeman, R. F., O'Malley, S. S., et al. (2010). Understanding the construct of impulsivity and its relationship to alcohol use disorders. Addictive Biology, 15, 217–226.
Durston, S., Thomas, K. M., Yang, Y., Uluğ, A. M., Zimmerman, R. D., & Casey, B. J. (2002). A neural basis for the development of inhibitory control. Developmental Science, 5, F9–F16.
Evans-Polce, R., Lanza, S., & Maggs, J. (2016). Heterogeneity of alcohol, tobacco, and other substance use behaviors in US college students: A latent class analysis. Addictive Behaviors, 53, 80–85.
Fair, D. A., Bathula, D., Nikolas, M. A., & Nigg, J. T. (2012). Distinct neuropsychological subgroups in typically developing youth inform heterogeneity in children with ADHD. Proceedings of the National Academy of Sciences, 109, 6769–6774.
Ferguson, S. L., G. Moore, E. W., & Hull, D. M. (2020). Finding latent groups in observed data: A primer on latent profile analysis in Mplus for applied researchers. International Journal of Behavioral Development, 44(5), 458–468.
Foulkes, L., & Blakemore, S. J. (2018). Studying individual differences in human adolescent brain development. Nature Neuroscience, 21(3), 315-323.
Galván, A., Poldrack, R. A., Baker, C. M., et al. (2011). Neural correlates of response inhibition and cigarette smoking in late adolescence. Neuropsychopharmacology, 36, 970–978.
Glover, G. H., & Law, C. S. (2001). Spiral-in/out BOLD fMRI for increased SNR and reduced susceptibility artifacts. Magnetic Resonance in Medicine, 46, 515–522.
Gogtay, N., Giedd, J. N., Lusk, L., Hayashi, K. M., Greenstein, D., Vaituzis, A. C., et al. (2004). Dynamic mapping of human cortical development during childhood through early adulthood. Proceedings of the National Academy of Sciences, 101, 8174–8179.
Grant, B. F., & Dawson, A. (1997). Age at onset of alcohol use and its association with DSM-IV alcohol abuse and dependence: Results from the National Longitudinal Alcohol Epidemiologic Survey. Journal of Substance Abuse, 9, 103–110.
Hardee, J. E., Weiland, B. J., Nichols, T. E., Welsh, R. C., Soules, M. E., Steinberg, D. B., et al. (2014). Development of impulse control circuitry in children of alcoholics. Biological Psychiatry, 76, 708–716.
Hariri, A. R. (2009). The neurobiology of individual differences in complex behavioral traits. Annual Review of Neuroscience, 32, 225–247.
Heitzeg, M. M., Nigg, J. T., Hardee, J. E., Soules, M., Steinberg, D., Zubieta, J. K., et al. (2014a). Left middle frontal gyrus response to inhibitory errors in children prospectively predicts early problem substance use. Drug & Alcohol Dependence, 141, 51–57.
Heitzeg, M. M., Nigg, J. T., Yau, W. Y. W., Zucker, R. A., & Zubieta, J. K. (2010). Striatal dysfunction marks preexisting risk and medial prefrontal dysfunction is related to problem drinking in children of alcoholics. Biological Psychiatry, 68, 287–295.
Heitzeg, M. M., Villafuerte, S., Weiland, B. J., Enoch, M. A., Burmeister, M., Zubieta, J. K., et al. (2014b). Effect of GABRA2 genotype on development of incentive-motivation circuitry in a sample enriched for alcoholism risk. Neuropsychopharmacology, 39, 3077–3086.
Herning, R. I., Better, W. E., Tate, K., & Cadet, J. L. (2005). Cerebrovascular perfusion in marijuana users during a month of monitored abstinence. Neurology, 64(3), 488–493.
Jenkinson, M., Bannister, P., Brady, M., & Smith, S. (2002). Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage, 17, 825–841.
Karoly, H. C., Bryan, A. D., Weiland, B. J., Mayer, A., Dodd, A., & Feldstein Ewing, S. W. (2015). Does incentive-elicited nucleus accumbens activation differ by substance of abuse? An examination with adolescents. Developmental Cognitive Neuroscience, 16, 5–15.
Knutson, B., Adams, C. M., Fong, G. W., & Hommer, D. (2001). Anticipation of increasing monetary reward selectively recruits nucleus accumbens. Journal of Neuroscience, 21, RC159–RC159.
Knutson, B., Westdorp, A., Kaiser, E., & Hommer, D. (2000). fMRI visualization of brain activity during a monetary incentive delay task. Neuroimage, 12, 20–27.
Lanza, S. T., & Rhoades, B. L. (2013). Latent class analysis: An alternative perspective on subgroup analysis in prevention and treatment. Prevention Science, 14, 157–168.
Lecei, A., van Hulst, B. M., de Zeeuw, P., van der Pluijm, M., Rijks, Y., & Durston, S. (2019). Can we use neuroimaging data to differentiate between subgroups of children with ADHD symptoms: A proof of concept study using latent class analysis of brain activity. Neuroimage: Clinical, 21, 101601.
Luijten, M., Machielsen, M. W., Veltman, D. J., Hester, R., de Haan, L., & Franken, I. H. (2014). Systematic review of ERP and fMRI studies investigating inhibitory control and error processing in people with substance dependence and behavioural addictions. Journal of Psychiatry & Neuroscience, 39(3), 149–169.
Mahmood, O. M., Goldenberg, D., Thayer, R., Migliorini, R., Simmons, A. N., & Tapert, S. F. (2013). Adolescents' fMRI activation to a response inhibition task predicts future substance use. Addictive Behaviors, 38(1), 1435–1441.
Maggs, J. L., Williams, L. R., & Lee, C. M. (2011). Ups and downs of alcohol use among first-year college students: Number of drinks, heavy drinking, and stumble and pass out drinking days. Addictive Behaviors, 36, 197–202.
Martz, M. E., Trucco, E. M., Cope, L. M., Hardee, J. E., Jester, J. M., Zucker, R. A., et al. (2016). Association of marijuana use with blunted nucleus accumbens response to reward anticipation. JAMA Psychiatry, 73, 838–844.
Miech, R. A., Johnston, L. D., O'Malley P. M., Bachman, J. G., Schulenberg, J. E., & Patrick, M. E. (2019). Monitoring the Future national survey results on drug use, 1975-2018: Volume I, Secondary school students. Ann Arbor: Institute for Social Research, The University of Michigan
Moran, L. V., Stoeckel, L. E., Wang, K., Caine, C. E., Villafuerte, R., Calderon, V., ... & Eden Evins, A. (2018). Nicotine increases activation to anticipatory valence cues in anterior insula and striatum. Nicotine and Tobacco Research, 20(7), 851–858.
Müller, K. U., Gan, G., Banaschewski, T., Barker, G. J., Bokde, A. L., Büchel, C., et al. (2015). No differences in ventral striatum responsivity between adolescents with a positive family history of alcoholism and controls. Addictive Biology, 20, 534–545.
Muthén, L. K., & Muthén, B. O. (2015). Mplus version 7.4 software. Los Angeles: Statmodel.com
Nestor, L., Hester, R., & Garavan, H. (2010). Increased ventral striatal BOLD activity during non-drug reward anticipation in cannabis users. Neuroimage, 49,1133–1143.
Noll, D. C., Fessler, J. A., & Sutton, B. P. (2005). Conjugate phase MRI reconstruction with spatially variant sample density correction. IEEE Transactions on Medical Imaging, 24, 325–336.
Noll, R. B., Zucker, R. A., Fitzgerald, H. E., & Curtis, W. J. (1992). Cognitive and motoric functioning of sons of alcoholic fathers and controls: The early childhood years. Developmental Psychology, 28, 665–675.
Norman, A. L., Pulido, C., Squeglia, L. M., Spadoni, A. D., Paulus, M. P., & Tapert, S. F. (2011). Neural activation during inhibition predicts initiation of substance use in adolescence. Drug & Alcohol Dependence, 119, 216–223.
Nylund, K. L., Asparouhov, T., & Muthén, B. O. (2007). Deciding on the number of classes in latent class analysis and growth mixture modeling: A Monte Carlo simulation study. Structural Equation Modeling, 14, 535–569
Nylund-Gibson, K., & Choi, A. Y. (2018). Ten frequently asked questions about latent class analysis. Translational Issues in Psychological Science, 4(4), 440–461.
Quinn, P. D., & Harden, K. P. (2013). Differential changes in impulsivity and sensation seeking and the escalation of substance use from adolescence to early adulthood. Developmental Psychopathology, 25, 223–239.
Richmond-Rakerd, L. S., Slutske, W. S., Lynskey, M. T., Agrawal, A., Madden, P. A., Bucholz, K. K., et al. (2016). Age at first use and later substance use disorder: Shared genetic and environmental pathways for nicotine, alcohol, and cannabis. Journal of Abnormal Psychology, 125, 946.
Ritchwood, T. D., Ford, H., DeCoster J., Sutton, M., & Lochman, J. E. (2015). Risky sexual behavior and substance use among adolescents: A meta-analysis. Child & Youth Services Review, 52, 74–88.
Robins, L. N., Cottler, L. B., Bucholz, K. K., Compton, W. M., North, C. S., & Rourke, K. M. (2000). Diagnostic Interview Schedule for the DSM-IV (DIS-IV). Washington University School of Medicine St. Louis, MO.
Schulenberg, J. E., Johnston, L. D., O'Malley, P. M., Bachman, J. G., Miech, R. A., & Patrick, M. E. (2019). Monitoring the Future national survey results on drug use, 1975-2018: Volume II, college students and adults ages 19-60. Ann Arbor: Institute for Social Research, The University of Michigan.
Schulenberg, J., Maslowsky, J., Patrick, M. E., & Martz, M. (2016). Substance use in the context of adolescent development. In The Oxford handbook of adolescent substance abuse (pp. 1-34). Oxford University Press Oxford.
Shulman, E. P., Smith, A. R., Silva, K., Icenogle, G., Duell, N., Chein, J., et al. (2016). The dual systems model: Review, reappraisal, and reaffirmation. Developmental Cognitive Neuroscience, 17, 103–117.
Smith, A. M., Zunini, R. A. L., Anderson, C. D., Longo, C. A., Cameron, I., Hogan, M. J., & Fried, P. A. (2011). Impact of marijuana on response inhibition: An fMRI study in young adults. Journal of Behavioral and Brain Science, 1(03), 124.
Smith, J. L., Mattick, R. P., Jamadar, S. D., & Iredale, J. M. (2014). Deficits in behavioural inhibition in substance abuse and addiction: a meta-analysis. Drug and Alcohol Dependence, 145, 1–33.
Spear, L. P. (2004). Adolescent brain development and animal models. Annals of the New York Academy of Sciences, 1021, 23-26.
Steinberg, L., Albert, D., Cauffman, E., Banich, M., Graham, S., & Woolard, J. (2008). Age differences in sensation seeking and impulsivity as indexed by behavior and self-report: Evidence for a dual systems model. Developmental Psychology, 44, 1764–1778.
Sutton, B. P., Noll, D. C., & Fessler, J. A. (2003). Fast, iterative image reconstruction for MRI in the presence of field inhomogeneities. IEEE Transactions on Medical Imaging, 22, 178–188.
Tapert, S. F., Schweinsburg, A. D., Drummond, S. P. A., Paulus, M. P., Brown, S. A., Yang, T. T., & Frank, L. R. (2007). Functional MRI of inhibitory processing in abstinent adolescent marijuana users. Psychopharmacology, 194(2), 173–183
van Hulst, B. M., De Zeeuw, P., & Durston, S. (2015). Distinct neuropsychological profiles within ADHD: A latent class analysis of cognitive control, reward sensitivity and timing. Psychological Medicine, 45, 735–745.
Wang, K. S., Zegel, M., Molokotos, E. et al. (2020). The acute effects of nicotine on corticostriatal responses to distinct phases of reward processing. Neuropsychopharmacology, 45, 1207–1214.
Weiland, B. J., Korycinski, S. T., Soules, M., Zubieta, J.-K., Zucker, R. a, & Heitzeg, M. M. (2014). Substance abuse risk in emerging adults associated with smaller frontal gray matter volumes and higher externalizing behaviors. Drug and Alcohol Dependence, 137, 68–75.
Weiland, B. J., Zucker, R. A., Zubieta, J. K., & Heitzeg, M. M. (2017). Striatal dopaminergic reward response relates to age of first drunkenness and feedback response in at-risk youth. Addiction Biology, 22(2), 502–512.
Wetherill, R. R., Castro, N., Squeglia, L. M., & Tapert, S. F. (2013a). Atypical neural activity during inhibitory processing in substance-naive youth who later experience alcohol-induced blackouts. Drug and Alcohol Dependence, 128(3), 243–249.
Wetherill, R. R., Squeglia, L. M., Yang, T. T., & Tapert, S. F. (2013b). A longitudinal examination of adolescent response inhibition: Neural differences before and after the initiation of heavy drinking. Psychopharmacology, 230, 663–671.
Whelan, R., Watts, R., Orr, C. A., Althoff, R. R., Artiges, E., Banaschewski, T., et al. (2014). Neuropsychosocial profiles of current and future adolescent alcohol misusers. Nature, 512, 185–189.
Wright, L., Lipszyc, J., Dupuis, A., Thayapararajah, S. W., & Schachar, R. (2014). Response inhibition and psychopathology: A meta-analysis of go/no-go task performance. Journal of Abnormal Psychology, 123, 429–439.
Yau, W. Y. W., Zubieta, J. K., Weiland, B. J., Samudra, P. G., Zucker, R. A., & Heitzeg, M. M. (2012). Nucleus accumbens response to incentive stimuli anticipation in children of alcoholics: relationships with precursive behavioral risk and lifetime alcohol use. Journal of Neuroscience, 32, 2544–2551.
Zucker, R. A., Fitzgerald, H. E., & Noll, R. (1990). Drinking and Drug History (Rev. Ed, Version 4), University of Michigan Department of Psychiatry, Addiction Research Center
Zucker, R. A., Fitzgerald, H. E., Noll, R. B. (1994) Drinking and Drug History Form for Children. University of Michigan Department of Psychiatry, Addiction Research Center.
Zucker, R. A., Fitzgerald, H. E., Refior, S. K., Puttler, L. I., Pallas, D. M., & Ellis, D. A. (2000). The clinical and social ecology of childhood for children of alcoholics: Description of a study and implications for a differentiated social policy. In Fitzgerald, H. E., Lester, B. M., & Zuckerman, B. M. (Eds.), Children of addiction: Research, health, and policy issues. Routledge Falmer, pp. 109–141
Zucker, R. A., Heitzeg, M. M., & Nigg, J. T. (2011). Parsing the undercontrol–disinhibition pathway to substance use disorders: A multilevel developmental problem. Child Development Perspectives, 5, 248–255.
Acknowledgments
Research reported in this publication was supported by the National Institute on Alcohol Abuse and Alcoholism of the National Institutes of Health under Award Numbers K01 AA027558 to MEM, K01 AA024804 to JEH, T32 AA007477 to F. Blow, R37 AA07065 to RAZ, and R01 AA12217 to MMH and RAZ. This research was also supported by the National Institute on Drug Abuse of the National Institutes of Health under Award Numbers K01 DA044270 to LMC and R01 DA027261 to MMH and RAZ. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest.
Additional information
Open Practices Statement
None of the data or materials for the analyses reported here are available, and the study was not preregistered.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 289 kb)
Rights and permissions
About this article
Cite this article
Martz, M.E., Cope, L.M., Hardee, J.E. et al. Subtypes of inhibitory and reward activation associated with substance use variation in adolescence: A latent profile analysis of brain imaging data. Cogn Affect Behav Neurosci 21, 1101–1114 (2021). https://doi.org/10.3758/s13415-021-00907-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13415-021-00907-8