Abstract
The inferior frontal gyrus/anterior insula (IFG/AI) and anterior cingulate cortex (ACC) are key regions involved in risk appraisal during decision making, but accounts of how these regions contribute to decision making under risk remain contested. To help clarify the roles of these and other related regions, we used a modified version of the Balloon Analogue Risk Task (Lejuez et al., Journal of Experimental Psychology: Applied, 8, 75–84, 2002) to distinguish between decision-making and feedback-related processes when participants decided to pursue a gain as the probability of loss increased parametrically. Specifically, we set out to test whether the ACC and IFG/AI regions correspond to loss aversion at the time of decision making in a way that is not confounded with either reward-seeking or infrequency effects. When participants chose to discontinue inflating the balloon (win option), we observed greater ACC and mainly bilateral IFG/AI activity at the time of decision as the probability of explosion increased, consistent with increased loss aversion but inconsistent with an infrequency effect. In contrast, we found robust vmPFC activity when participants chose to continue inflating the balloon (risky option), consistent with reward seeking. However, in the cingulate and in mainly bilateral IFG regions, blood-oxygenation-level-dependent activation decreased when participants chose to inflate the balloon as the probability of explosion increased, findings that are consistent with a reduced loss aversion signal. Our results highlight the existence of distinct reward-seeking and loss-averse signals during decision making, as well as the importance of distinguishing between decision and feedback signals.
Similar content being viewed by others
The avoidance of risky behaviors, particularly related to the experience of loss (Rothman & Salovey, 1997; Saaty, 1987; Yates, 1992), is a central feature of decision making (Tversky & Kahneman, 1981), and one that varies significantly across a population (Weber, Blais, & Betz, 2002). Some amount of risk-taking behavior is considered beneficial to human growth and survival, whereas having a reduced cognitive capacity to avoid excessive risk taking can contribute to the development of a variety of subclinical and clinical problems, including bipolar disorder, pathological gambling, substance abuse, and depression (Holmes et al., 2009; Martins, Tavares, & da Silva Lobo, 2004; Swann, Lijffijt, & Lane, 2009). In recent years, a host of neuroscience approaches have been used to help identify and model the neural mechanisms of risky decision making.
In spite of this increased focus, the growing neuroimaging literature on the neural correlates of loss aversion in risky decision making continues to yield equivocal findings. As a consequence of experimental confounds related to decision processes, alternative hypotheses and models have emerged as potential explanations for how brain regions, especially the inferior frontal gyrus/anterior insula (IFG/AI) and anterior cingulate cortex (ACC), are involved in risky decision making. By using a task design that avoids confounding distinct components of risky decision making, the present study addresses several key issues that may help clarify the roles of the IFG/AI and ACC regions. The existing literature suggests the IFG/AI and ACC as likely loci of loss avoidance effects (Brown & Braver, 2007; Krawitz, Fukunaga, & Brown, 2010; Magno, Foxe, & Molholm, 2006), so the analysis below focuses on these regions specifically.
Distinguishing between decision-making and feedback signals in choice tasks
Neuroimaging research has established that decision making under risk or uncertainty involves a distributed neural network of cortical and subcortical regions showing distinct activation patterns at different stages of the decision process (Cohen, Heller, & Ranganath, 2005; Ernst & Paulus, 2005; Krain, Wilson, & Arbuckle, 2006). During the decision phase, the ACC has been associated with error monitoring (Holroyd & Coles, 2002), learning the likelihood of an error (Brown & Braver, 2005), conflict detection (van Veen, Holroyd, & Cohen, 2004), and performance-monitoring processes (Gehring & Knight, 2000). Whereas in the outcome phase, good outcomes (when compared to bad outcomes) generate increased blood-oxygenation-level-dependent (BOLD) activity in the posterior orbitomedial cortex, bilaterally in the ventral medial prefrontal cortex, and in the subcallosal ACC (Rogers et al., 2004). Brain regions implicated in cognitive–affective processes, such as the ventral striatum, also are critically involved in outcome and feedback processes (Knutson, Fong, & Bennett, 2003; Rogers et al., 2004; Schoenbaum & Setlow, 2003).
In spite of this evidence, inferences about brain activity during the time of decision making have been confounded by brain activity at the time that the outcome is presented. The ACC and AI typically show a strong error effect (Kiehl, Liddle, & Hopfinger, 2000; Magno et al., 2006) such that risky conditions, which on average lead to more errors, will show greater average activity based solely on making the errors rather than on the anticipation of errors during the decision period. This issue has affected some fMRI studies of loss avoidance that did not include sufficient temporal delay variability, or jitter, between the decision and feedback periods of a trial (Fukui, Murai, & Fukuyama, 2005; Rao, Korczykowski, & Pluta, 2008).
Loss aversion signals in the ACC, AI, and IFG
Converging evidence has suggested that brain regions critically involved in cognitive control processes, such as the ACC, play an important role in decision making (Gehring & Willoughby, 2002; Kuhnen & Knutson, 2005; Platt & Huettel, 2008), particularly in making bad decisions as compared to good ones (Fukui et al., 2005; Lawrence, Jollant, & O’Daly, 2009). Some studies have shown that ACC regions may signal anticipated risks, and especially potential loss (Krawitz et al., 2010). Here, loss aversion signals are defined where brain regions show increased activity for choices that entail greater losses and where such activity correlates positively with improvements in behavioral performance. The premise here is that loss aversion signals, if they exist, may become active when an option with a potential loss is contemplated, but they are not sufficiently strong to always prevent the option from being selected. Instead, they reduce the overall rate of choices involving potential losses.
Similarly, others have argued that AI activity signals risk (Preuschoff, Quartz, & Bossaerts, 2008) or risk avoidance during a risky decision (Kuhnen & Knutson, 2005; Paulus, Rogalsky, & Simmons, 2003), where greater risk is defined as a greater variance in the likely outcomes. Other reports have argued that while AI is sensitive to errors (Magno et al., 2006) and pain (Singer et al., 2004), its role is primarily constrained to avoidance behaviors and to signaling harm or loss avoidance, rather than to being involved in reward seeking (Krawitz et al. 2010; Paulus et al., 2003). Moreover, additional reports have shown that interoceptive signals within the insula (Craig, 2002; Gray & Critchley, 2007) are associated with cue-induced urges that result in experiences of “craving” (Naqvi & Bechara, 2009; Naqvi, Rudrauf, Damasio, & Bechara, 2007; Verdejo-García & Bechara, 2009). Similarly, IFG BOLD responses may reflect risk aversion signals (Christopoulos, Tobler, & Bossaerts, 2009), particularly as measured by subjective versions of risk and risk prediction error (Bossaerts, 2010).
This activation in the IFG apparently signaling subjective risk may reflect a carryover effect from the AI (Christopoulos et al., 2009), which is believed to be critical in the formation of subjective feelings (Craig, 2009). Similar activation patterns between the IFG and AI also have been reported for emotional responses (Jabbi & Keysers, 2008). A recent review suggested that while it remains unclear which brain region is responsible for the origination of the risk-induced signals, the IFG, insula, and anterior cingulate are strongly implicated in such processes (Bossaerts, 2010).
Distinguishing between infrequency effects and loss aversion signals
The ACC is known to respond more strongly to infrequent events than to more common events (Braver, Barch, & Gray, 2001; Hayden, Heilbronner, & Pearson, 2011; Jessup, Busemeyer, & Brown, 2010; Nee, Kastner, & Brown, 2011; Oliveira, McDonald, & Goodman, 2007). We therefore define infrequency effects here as an inverse relationship between event-related brain activity and the prior probability of that event. Infrequency effects could be found for any type of event, such as a safe decision with a known outcome, a risky decision with an uncertain outcome, or feedback from the environment. The unifying principle is that the less often an event occurs—whether it be an infrequent choice or outcome—the more brain activity will be found when it does occur. For example, recent research showed that a rare monetary win following a gamble with a low payoff probability corresponded to more ACC activity when the win occurs (Jessup et al., 2010).
It should be emphasized that the presence of infrequency effects does not necessarily imply causation. In the present study, if ACC and IFG/AI are more active during risky choices in participants who tend to avoid risk, as has been shown previously (Fukui et al., 2005; Krawitz et al., 2010), there are at least two possible explanations of this increased activity. First, it is possible that the ACC and IFG/AI have no influence on the decision, but merely reflect, by their greater activity, that some options are chosen less frequently—that is, an infrequency effect. Alternatively, it is possible that the IFG/AI and/or the ACC play causal roles by biasing decisions to avoid loss in more risk-averse individuals (Magno et al., 2006).
Testing loss aversion signals in the Balloon Analogue Risk Task (BART)
The central aim of the present study is to investigate the extent and manner by which the ACC and IFG/AI regions signal loss avoidance, in a way that is not confounded with either reward-seeking signals or infrequency effects, especially at the time of decision making. To assess this, we used a variant of the Balloon Analogue Risk Task (BART; Lejuez et al., 2002), a popular laboratory task for testing risk taking across both clinical and nonclinical populations.
To date, we are unaware of any BART neuroimaging study that has examined these confounds based on the implementation of two key manipulations (for details, see the Method section): First, we provided sufficient jitter between distinct decision and outcome stages of each trial to distinguish BOLD signals between the decision-making and feedback-related processes. Second, we paired an increasing risk of explosion (whose exact probabilities are unknown to the participant) with an increasing wager amount for each successive inflation, allowing for a parametric measure of the BOLD signal at the time of decision making. This allowed us to examine how each brain region responds to parametric manipulations of reward and loss probability.
To test the central question, we focused on two parametrically modulated regressors [indicated by the label *P(explode)] that denoted that the event-related regressors were modulated by the probability of explosion on the corresponding trial (for details, see the Method section): (1) ChooseInflate*P(explode), which is a decision event when the participant chooses to continue inflating the balloon, and (2) ChooseWin*P(explode), which is a decision event for when the participant chooses to discontinue inflating the balloon. We used ChooseInflate*P(explode) and ChooseWin*P(explode) to test competing hypotheses of loss avoidance against the confound of either reward seeking or the infrequency of risky choices. In general, the observed effects for these two parametrically modulated regressors could be consistent with one or more possible cognitive processes, but taken together, they constrain the solution space so that a single account remains the most defensible one. Even so, we do not claim that such observed effects are necessary to serve as a loss aversion signal, a question that can only be addressed with a lesion study. Rather, we argue for the possibility that a loss aversion signal exists by virtue of such effects. If a loss aversion does exist, then we predict that it would be marked by lesser activity with reduced loss avoidance during decisions in the behavioral task. The logic behind these parametrically modulated regressors and their respective hypotheses are described below:
Hypothesis 1: Loss aversion versus reward seeking (ChooseInflate condition)
If ACC and AI drive loss aversion, then ChooseInflate activity in those regions should decrease as the probability of explosion increases (see Fig. 1A). This is because choosing to continue inflating reflects a reduced sensitivity to the risk of explosion despite an increasing risk of losing the entire (and growing) amount at stake. Conversely, if ACC and/or IFG/AI signal reward seeking, ChooseInflate activity in these regions should increase rather than decrease, as choosing to inflate increases the amount of monetary reward that can be gained (see Fig. 1B).
Competing hypotheses regarding decision-making signals. Parametric modulators were used to identify brain regions where activation was positively or negatively correlated with the probability of explosion. We focused on two main parametrically modulated regressors [indicated by the label *P(explode)]. In all panels, the vertical axis represents a neural “risk signal”—that is, a magnetic resonance signal effect in which activation increases as inflation [ChooseInflate*P(explode)] is chosen with increasing probability of explosion. The horizontal axis represents the predetermined probabilities of balloon explosion. (A, B, C) These three panels depict competing hypotheses about the role of the inferior frontal gyrus/anterior insula (IFG/AI) and anterior cingulate cortex (ACC) in choice behavior. The bold lines represent the selection of the risky option [ChooseInflate*P(explode)], in which the participant chooses to inflate the balloon. The dotted lines represent the selection of the safe option [ChooseWin*P(explode)], in which the participant chooses to stop inflating the balloon and take the sure win. If the IFG/AI activity reflects loss aversion signals, as we hypothesize, the correlation between the probability of explosion and the neural risk signal should look like panel A, showing a negative relationship for the risky option and a positive relationship for the safe option. Alternatively, if the IFG/AI generates reward-seeking signals, the correlation for the risky option should be positive (panel B), and if ACC signals infrequency effects, the correlation for the sure option should be negative (panel C)
Hypothesis 2: Loss aversion versus infrequency effects
It must be noted that participants are very likely to inflate the balloon at the beginning of a trial, when the balloon has not yet been inflated at all. As the inflation proceeds, participants are less likely to continue inflating, so that at late inflation choices, participants are unlikely to continue inflating (the ChooseInflate event), and instead will opt to stop inflating and bank their winnings (the ChooseWin event). Thus, any effect of infrequency in the ChooseInflate condition should be greatest at high inflation numbers (where explosion probability is high and the probability of ChooseInflate events occurring is low). Should this infrequency explanation hold, we also would predict the ChooseInflate regressor that is parametrically modulated by explosion probability to be positive. Of note, this also could be consistent with a reward-seeking effect.
The follow-up test to discriminate between reward-seeking and infrequency effects involves the ChooseWin regressor: This regressor, which is parametrically modulated by explosion probability, should be negative if an infrequency explanation holds, owing to the rarity of the instances when participants choose to win/stop when the probability of explosion is low (see Fig. 1C). In contrast, if the ChooseWin regressor that is parametrically modulated by explosion probability is positive instead, this would be consistent with the region showing loss aversion or reward seeking (where reward seeking as an explanation can be ruled out if these regions signal loss avoidance, as described in Hypothesis 1 above).
Method
The Indiana University Bloomington Institutional Review Board approved the study procedures reported here.
Participants
A group of 16 participants (8 female, 8 male), ages 18–23 years (mean age = 20.19 years) were recruited from the student body of Indiana University, Bloomington. All of the participants provided written informed consent and met standard health and safety requirements for entry into the magnetic resonance imaging scanner. They were paid $25 for participation, plus performance bonuses based on points earned during the task.
Design and procedure
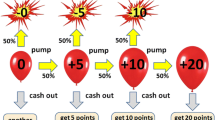
The implementation of the BART was guided, in part, by prior imaging work using the task (cf. Rao et al., 2008). Here, the task was modified to include a longer variable delay between choice- and feedback-related brain processes when participants chose to pursue a gain while the probability of loss increased parametrically. In the earlier fMRI study of the BART (Rao et al., 2008), jittering was included from the time of feedback until the onset of the next trial, but there was no jittering between the buttonpress response and the subsequent feedback of inflation versus explosion, which made it difficult to dissociate decision versus feedback-related neural activity as we do here. In the present design, participants completed one 8-min block of the task outside of the scanner using a laptop computer in order to become familiar with the response options and task procedure. The participants were instructed to “inflate the balloon as much as possible without popping it” and to “maximize the number of points earned.” Additionally, participants were told that at the end of the session, a portion of the cumulative winnings shown on the screen would be converted into bonus money and added to the standard compensation for taking part in the study. Twelve inflation responses were possible for a given balloon trial, with a parametric increase in the probability of explosion over successive inflation responses (i.e., 0 % for $0.0; 2.1 % for $0.05; 4.2 % for $0.15; 6.3 % for $0.25; 14.6 % for $0.55; 23.9 % for $0.95; 31.3 % for $1.45; 43.8 % for $2.05; 56.3 % for $2.75; 68.8 % for $3.45; 79.2 % for $4.25; 89.6 % for $5.15).
During fMRI data collection, the participants completed two 8-min blocks of the BART. At the beginning and end of each block, the screen showed a fixation cross (“+”) in the center for approximately 30 s to establish baseline activity. Participants were instructed to simply look at the fixation point and rest. At the start of each trial (from top to bottom), an image of a balloon, a square green decision cue, the current wager amount, and the banked winnings across all balloon trials through the most recently completed trial were displayed on the screen (see Fig. 2). The participants had unlimited time to respond (to inflate the balloon or to take the accumulated wager amount for a given trial). After a response, an exponentially distributed delay of 0, 2, 4, or 6 s occurred (with a 60 % stopping probability after each 2-s interval up to 6 s). During the delay, no feedback was given, to allow for separate estimation of the BOLD response during (inflation) choice and (explosion or successful inflation) outcome (feedback) periods (Dale, 1999). Following the delay, the outcome feedback was presented, showing an image of a balloon either exploding or successfully inflating, or the balloon was replaced by the words “You Win!” in red letters centered on the screen for 1,000 ms. If the balloon inflated, the decision cue turned red for an equiprobable 1,500, 2,000, or 2,500 ms, during which time further responses were disallowed. After that, the decision cue turned green again to indicate that the next choice could be made. Following a win or loss, the screen was blank for an equiprobable 2,000, 3,000, or 4,000 ms, after which a new balloon appeared and the inflation process repeated. Following an explosion, the exploded balloon remained visible for 500 ms, followed by the words “You Lose!” in red letters centered on the screen for 1,000 ms. Total winnings were updated after the outcome presentation.
fMRI acquisition and data preprocessing
Imaging data were collected on a Siemens Magnetom Trio 3.0-Tesla MRI scanner and a 32-channel head coil. For each participant, functional BOLD data were collected using a gradient echo T2*-weighted echo planar imaging sequence with free induction decay for two blocks of 240 whole-brain volumes (echo time [TE] = 25 ms, repetition time [TR] = 2,000 ms, flip angle = 70º) with 35 axial slices (64 × 64 grid, 3.4 × 3.4 × 3.8 mm voxels, interleaved order, 3.8-mm thickness, 0-mm spacing). A structural scan was collected at the end of each session using three-dimensional MP-RAGE imaging using a high-resolution T1-weighted imaging sequence, (TE = 2.67 ms, TR = 1,800 ms, flip angle = 9º) with nonselective excitation consisting of 192 sagittal slices (512 × 448 grid, 1.0 × 1.0 × 1.0 mm voxels, 1-mm thickness).
Preprocessing was done using SPM5 (Wellcome Trust Centre for Neuroimaging, 2005), except where otherwise specified. The structural scan was skull-stripped using FSL’s BET2 with default parameters (Péchaud, Jenkinson, & Smith, 2006). Functional data were spike-corrected on a voxel-by-voxel basis to reduce the impact of artifacts using AFNI’s 3dDespike. The functional images were slice-timing corrected using sync interpolation (Oppenheim, Schafer, & Buck, 1999), motion corrected by means of a least-squares six-parameter rigid body, and coregistered with the structural scan. Once the structural scan was normalized to the SPM MNI template and the warps were applied to the functional data, the resulting normalized images were smoothed with an 8-mm3 full-width-at-half-maximum isotropic Gaussian kernel.
fMRI analysis
Event-related responses were estimated using a general linear model (GLM) with random effects for each participant with a canonical hemodynamic response function with no derivatives, a microtime resolution of 16 time bins per scan, a high-pass filter cutoff of 128 s using a residual forming matrix, autoregressive AR(1) to account for serial correlations, and restricted maximum likelihood (ReML) for model estimation. The model included a total of 17 regressors: two constant terms, six motion regressors, and nine regressors for experimental conditions during the decision and outcome periods. Decision-making events, which were aligned to the time of response, were modeled by two regressors, one for choosing to continue inflating the balloon (ChooseInflate), and another for choosing to discontinue inflating the balloon (ChooseWin). Outcome events were modeled by three regressors, one for a loss event in which the balloon explodes (ExplodeOutcome) and the text “You Lose!” appears, another for a successful balloon inflation event (SuccessfulInflate), in which a larger balloon with an increased wager is displayed, and finally one for choosing to discontinue inflations—an immediate win event (WinOutcome) in which the text “You Win!” is presented. In the event-related analysis, the probabilities of explosions were included as a parametric modulator with each event-type regressor, except for the win event. This provided four parametric modulators to identify brain regions where activation was positively or negatively correlated with the probability of explosion: ChooseInflate*P(explode), ChooseWin*P(explode), SuccessfulInflate*P(explode), and ExplodeOutcome*P(explode).
Analyses for main effects, parametric modulators, and planned contrasts, which included additional tests for error effects [ExplodeOutcome*P(explode) – SuccessfulInflate*P(explode)] and loss aversion effects [ChooseWin*P(explode) – ChooseInflate*P(explode)] scaled with explosion probability were performed at the second level using whole-brain analysis. The second-level analyses used linear regression on the per-participant measures with ReML estimation in SPM5. The statistical threshold for significance was p < .05, with false-discovery rate (FDR) correction. In keeping with the central hypotheses, we focused on the two parametrically modulated regressors at the time of decision, namely ChooseInflate*P(explode) and ChooseWin*P(explode). The additional parametric modulators included in the full model, as well as the planned contrasts for error effects and loss aversion effects are reported in the supplementary materials. It should also be noted that higher levels of inflation were associated with both greater reward and greater explosion probability. We dissociated the reward-seeking and loss aversion influences by separately analyzing the choices to stop inflating (ChooseWin, presumably involving loss avoidance) versus choices to continue inflating (ChooseInflate, presumably involving reward seeking).
Region-of-interest (ROI) analysis
Follow-up ROI analyses were performed using SPM5 and MarsBaR (Brett, Anton,& Valabrègue, 2002) within significant regions from the group analyses described above. Specifically, the contrasts computed within a given ROI were the same contrasts used to define the ROI, which necessitated appropriate correction for multiple comparisons. The ROI analyses were used to illustrate and provide further details about the significant relationships found using the whole-brain voxel-by-voxel tests described above. Regions were defined as contiguous voxels that each passed the p < .05, FDR-corrected, threshold. However, where noted, some additional analyses were selected by the peak areas of activation for clusters of activation that passed both an uncorrected threshold of p < .001 for each voxel and a corrected cluster-level p value of less than .01. Mean parameter estimates within ROIs are reported as percentages of magnetic resonance (MR) signal change, calculated as the mean magnitude of the event regressor relative to the mean magnitude of the constant term regressor within the region. The WFU Pick Atlas (Maldjian, Laurienti, & Kraft, 2003) and Talairach Daemon (Lancaster et al., 2000) were used for anatomical labeling. MRIcron software (www.sph.sc.edu/comd/rorden/mricron/) was used to display the ROIs.
Results
Behavioral results
The participants completed on average 36.2 (SD = 5.2) balloons, and each balloon was inflated a mean of 5.2 times. The average number of inflations per balloon for a given participant ranged from 2.5 to 7.3. Across participants, the average of the standard deviations of inflations per balloon was 0.8. Across all participants, the minimum number of inflations per balloon was 0, and the maximum was 9.
If the analysis is restricted to only the balloons for which the participants chose to stop so that no explosion occurred, these balloons end with a ChooseWin trial and reveal the limit of the participants’ risk preferences. Participants completed on average 21.8 (SD = 7.6) balloons that did not explode, and for each balloon inflated 5.3 times. For balloons that did not explode, the average number of inflations per balloon for a given participant ranged from 3.6 to 6.8. Across participants, the average of the standard deviations of inflations per (nonexploding) balloon was 0.9.
Some participants inflated the balloons as many as nine times. This suggests the possibility that some of the highest inflation numbers might be outliers. To address this, we carried out an analysis restricted to only ChooseWin trials. Assuming a normal distribution of ChooseWin trials as a function of inflation number, only four single trials across two participants had inflation numbers three standard deviations above the mean for the corresponding participant. Overall, the behavioral results suggest that participants showed a range of frequent and less frequent choices, but the infrequent choices were not so infrequent as to constitute outlier data points.
The number of balloons on which participants won (21.8 ± 7.6) was greater than the number of balloons on which they lost (13.8 ± 4.5), and the difference was significant (p = .01). The average numbers of pumps, compared across the first, middle, and last ten trials across participants, did not significantly differ, F(2, 45) = 0.18, p = .84, with no differences between the first and second sets of ten trials, t(15) = –0.2, p > .8, the first and third sets, t(15) = 0.7, p > .5, or the second and third sets, t(15) = 1.2, p > .2. No significant difference was found for mean reaction times between the two response options (725 ± 298 ms in the inflate choice vs. 775 ± 332 ms in the sure win choice, p > .1), and reaction times across trials also did not change between blocks (732 ± 287 ms in Block 1 vs. 796 ± 362 ms in Block 2, p > .1). Table 1 shows that individuals with more balloon trials tended to have fewer inflations per trial, more win choices, and less winnings per trial than did individuals who had fewer total trials.
fMRI results
Decision phase
We used parametric modulation analysis to test the competing hypotheses of loss avoidance (see Fig. 1) against either reward seeking (Hypothesis 1) or the infrequency of risky choices (Hypothesis 2) in the brain ROIs. This approach allowed us to examine the within-subjects correlations between regional BOLD activity and predetermined probabilities of explosion during the decision phase. We did this by focusing on two main parametrically modulated regressors [indicated by the label *P(explode)]: (1) ChooseInflate*P(explode), which is a decision event in which the participant chooses to continue inflating the balloon, and (2) ChooseWin*P(explode), which is a decision event in which the participant chooses to discontinue inflating the balloon.
Loss aversion versus reward seeking: ChooseInflate parametric modulator
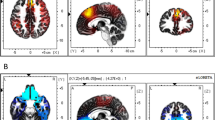
Consistent with loss aversion signals, the ChooseInflate*P(explode) regressor showed decreasing activity as inflation was chosen despite increasing probability of explosion (negative correlation, βs < 0) in the right anterior cingulate cortex (rACC) (cluster size k = 769, peak voxel = MNI 6, 26, 24), t(15) = 6.73, p < .05 FDR corrected (see Fig. 3A) and bilateral inferior frontal gyrus (biIFG) [(left IFG, cluster size k = 83, peak voxel = MNI –44, 16, –8), t(15) = 4.26, p < .05 FDR; (right IFG, cluster size k = 353, peak voxel = MNI 48, 20, –6), t(15) = 6.39, p < .05 FDR (see Fig. 3B)]. This is consistent with a loss aversion signal that is attenuated when participants choose to continue inflating despite increased risk, and it is inconsistent with both greater reward-seeking effects and infrequency effects.
Decision-phase parametric modulators. We focused on two main parametrically modulated regressors [indicated by the label *P(explode)]. Panels A and B show region-of-interest (ROI) analyses of brain regions showing a negative correlation between the neural risk effect and the increasing probability of explosion during the decision phase. a Shown in red is a sagittal slice of ACC (peak voxel = MNI 6, 26, 24). b Shown in yellow and blue is a transverse slice of bilateral IFG/AI (peak voxels = MNI –44, 16, –8 and 48, 20, –6) for the ChooseInflate*P(explode) (risky option) regressor. Panels C and D show ROI analyses of brain regions showing a positive correlation between the neural risk effect and the increasing probability of explosion during the decision phase. c Shown in green is a sagittal slice of left vmPFC (peak voxel = MNI –12, 36, –18) for the ChooseInflate*P(explode) (risky option) regressor. d Shown in violet and cyan is a transverse slice of bilateral IFG/INS for the ChooseWin*P(explode) (sure option) regressor
Other regions showed effects consistent with reward-seeking signals. The ChooseInflate*P(explode) regressor showed increasing activity (positive correlation; βs > 0) between brain activity and the probability of explosion in the left ventromedial prefrontal cortex (vmPFC) during inflation choices (cluster size k = 1,313, p < .001, cluster-corrected, peak voxel = MNI –12, 36, –18) (see Fig. 3C). Of note, this finding aimed at discriminating between loss aversion and reward seeking (Hypothesis 1) could not rule out infrequency effects when reward-seeking effects were found, as in the vmPFC. Such infrequency effects, if they existed, could be searched for separately as described below.
Loss aversion versus infrequency effects: ChooseWin parametric modulator
Consistent with loss aversion signals but not with infrequency effects, the ChooseWin*P(explode) regressor showed increasing activity (positive correlation, βs > 0) as the probability of explosion increased and participants chose to take their winnings and stop inflating. The effect was centered in biIFG and extended into the anterior insula [(left IFG, k = 125, peak voxel = MNI –40, 18, –4), t(15) = 7.61, p < .05 FDR; (right IFG, cluster size k = 209, peak voxel = MNI 40, 18, –6), t(15) = 7.47, p < .05 FDR] (see Fig. 3D). This finding by itself is consistent with loss aversion or reward-seeking signals but inconsistent with infrequency effects, because participants are more likely to stop inflating the balloon at higher levels of inflation. The complementary analysis of the ChooseInflate*P(explode) regressor above provides evidence to rule out reward seeking in these regions.
We also carried out a whole-brain search for regions that showed effects of infrequency, looking for where the ChooseWin*P(explode) regressor showed decreasing activity (negative correlation, βs < 0). No regions passed whole-brain correction at the level of individual voxels. Instead, only when an uncorrected threshold of p < .001 with corrected cluster-level p < .01 was used did the ChooseWin*P(explode) regressor show decreased activity (negative correlation, βs < 0) in the left middle temporal gyrus (cluster size k = 163, p < .001, cluster corrected, peak voxel = MNI –58, –42, –6), bilateral medial frontal gyrus (cluster size k = 285, p < .001, cluster corrected, peak voxel = MNI 12, –32, 62), left angular gyrus (cluster size k = 264, p < .001, cluster corrected, peak voxel = MNI –52, –68, 36), and left middle frontal gyrus (cluster size k = 419, p < .001, cluster corrected, peak voxel = MNI –26, 28, 46). Activity in these regions may be consistent with an effect of infrequency at the time of decision making. Note that we found no effects in the vmPFC region described above showing reward-seeking effects.
Given our hypothesis that ACC reflects loss aversion, we were surprised to note that the ChooseWin*P(explode) regressor did not show increasing activity (positive correlation, βs > 0) at the level of whole-brain correction, as did bilateral IFG/insula. To address this concern, we conducted a follow-up ROI analysis using the rACC region extracted from the ChooseInflate*P(explode) regressor (negative correlation, βs < 0). Using this region as a mask, we examined for additional evidence to support the role of the ACC in loss aversion, specifically for trials in which participants voluntarily chose to discontinue inflating the balloon [ChooseWin*P(explode) regressor]. Consistent with our hypothesis, we found a significantly positive relationship (βs > 0) with the ChooseWin*P(explode) regressor in the rACC region [t(15) = 2.05, p = .029], thus providing full evidence to support the role of the ACC in loss aversion [by showing a negative relationship with the ChooseInflate*P(explode) regressor, but a positive relationship with the ChooseWin*P(explode) regressor].
If ACC drives loss aversion signals, these signals should be stronger in participants who do not inflate the balloons as much on average. To test this, we computed for each participant the average inflation number at which participants voluntarily chose to stop inflating (i.e., only ChooseWin trials, not explosion trials), which we termed average inflations. Consistent with our hypotheses, we found that participants with lower average inflations showed a significantly stronger negative loading on the parametric modulator for the ChooseInflate*P(explode) regressor in the ACC, r(14) = .57, p < .05, whereas, for the vmPFC, participants with lower average inflations showed a significantly stronger positive loading on the parametric modulator for the ChooseInflate*P(explode) regressor, r(14) = –.56, p = .05. The absolute values of these parametric modulator beta weights were closest to zero for participants with higher average inflations. These results suggest that participants with higher average inflations (who continue to choose to inflate the balloon) have an overall reduced sensitivity to the probability of explosion in both the rACC and vmPFC.
Discussion
Using parametric modulation analysis in a modified BART task, we examined within-subjects correlations between BOLD activity and predetermined probabilities of explosion in distinct brain regions implicated in the neural correlates of risky decision making. We set out to clarify whether ACC and IFG/AI activities correspond to loss aversion at the time of decision making in a way that is not confounded with either reward-seeking (Hypothesis 1) or infrequency (Hypothesis 2) effects. The results were consistent with the hypothesis that ACC and IFG/AI signals are involved in loss aversion and are not attributable to the effects of reward seeking or the infrequency of risky choices. Specifically, in our initial whole-brain analysis, the ACC showed loss aversion signaling in the ChooseInflate condition, whereas the IFG showed loss aversion signaling in both the ChooseInflate and Choose Win regressors. However, a follow-up ROI analysis showed loss aversion signaling in the ACC also in response to the ChooseWin regressor.
To summarize, we used a variant BART paradigm to address the following concerns: First, we added temporal variability between the decision time and the time when the outcome was presented, allowing us to disentangle brain activity related to the time of decision from activity at the time of feedback. With this approach, we showed that activity at the time of decision making in the ACC and IFG was consistent with biasing decisions away from risk taking, in a way that was not confounded with the other effects described below. Logically, we have shown that ACC and IFG activity is sufficient to bias activity against risk, but we have not shown that it is necessary. A definitive inference of causation and necessity would be properly addressed with a lesion study. Second, the within-participants manipulation of risk across balloon inflation choices allowed us to assess whether brain activity in a given region increases or decreases for the choice to pursue reward despite the risk or to avoid loss. Collectively, our approach allowed us to disambiguate potential reward-seeking and loss aversion signals in the ACC and IFG/AI regions by analyzing choices to continue inflating separately from choices to stop inflating. We did not distinguish between risk and reward directly; the BART design is unable to separately evaluate the contributions of these factors, because they scale together as explosion probability increases. Finally, by looking at variations of activity within rather than between participants, we were able to control for variations in choice probability across participants that would otherwise lead to a confound between loss aversion effects and effects of the infrequency of risky choices.
ACC signals loss aversion and not the infrequency of risky choices
Some studies have reported ACC to be more active when participants avoid risky options (Fukui et al., 2005; Krawitz et al., 2010; Magno et al., 2006). In our recent studies of a modified Iowa Gambling Task, participants showed greater ACC and IFG/AI activity during the decision period when making a risky (bad) decision as compared to a safer (good) decision (Krawitz et al., 2010). These results appeared to show that participants with greater sensitivity to negative consequences were more likely to avoid the risky options, findings consistent with previous research (Brown & Braver, 2007; Magno et al., 2006).
While this account seemed plausible, it did not explain why participants continued to make bad decisions despite showing greater ACC activity, which has been shown to be associated with safer decisions across participants. One possible resolution to this conundrum was that greater ACC activity may only have a weak influence on loss avoidance and does not strongly determine behavioral choices. That is, ACC activity may be sensitive to loss, but it may not be centrally involved in a switch away from it. An alternative hypothesis, which we also considered in the present study, was based on previous literature that has suggested that greater ACC activity is a consequence of the infrequent occurrence of a particular option (e.g., selection of bad choices; Braver et al., 2001; Jessup et al., 2010). Under the infrequency hypothesis, the choice would drive ACC activity, rather than vice versa.
To discriminate between loss aversion signals and infrequency effects, we modified the BART in order to look within rather than across participants, thus creating a context in which we could ask whether there is greater or lesser activity in the ACC when participants choose a riskier versus a safer option. We predicted that if ACC drives loss aversion, this region would be less active when participants choose the risky option, even when such choices were infrequent.
The present results are consistent with the hypothesis that ACC drives loss aversion, even if it does not fully control decision making. Looking within participants, we found that the parametrically modulated regressors supported our predictions: ChooseInflate*P(explode) showed decreasing activity (negative correlation, βs < 0) in the ACC as participants continued to choose the risky option. In contrast, the ChooseWin*P(explode) regressor showed increasing activity (positive correlation, βs > 0) in the ACC (when applied as an ROI mask) and in biIFG extending into the AI as participants chose the safer option. An infrequency effect in the ChooseInflate*P(explode) regressor would have shown that increasing activity (positive correlation, βs > 0) in ACC occurs because participants rarely choose the risky option at higher inflation numbers, due to the greater probability of balloon explosion. Moreover, an infrequency effect in the ChooseWin*P(explode) regressor is not supported by the increasing activity (positive correlation, βs > 0) in biIFG, because stopping is more likely at higher inflation numbers.
IFG signals loss aversion and not reward seeking
To discriminate between loss aversion signals and reward seeking, we investigated whether the neural activity corresponding to when participants chose the riskier option (i.e., inflation) correlated either positively or negatively with greater explosion probability and available reward. We predicted that if IFG/AI drives reward seeking, this region would be more active when participants chose the risky option, whereas, in support of the loss aversion effects, regions would be less active when participants chose a riskier versus a safer option due to reduced loss aversion activity.
Intriguingly, the present results for the parametrically modulated regressor ChooseInflate*P(explode) were consistent with reward-seeking signals not in the IFG/AI regions, but in the left vmPFC, showing a positive relationship (positive correlation, βs > 0) between brain activity and the probability of explosion during choices to inflate. This is consistent with reward-seeking activity in this region, in agreement with previous reports that have found this region to be associated with experienced and stated preferences (Deppe, Schwindt, & Kugel, 2005; Koenigs & Tranel, 2008; McClure et al., 2004; Plassmann, Kenning, & Deppe, 2008). This positive correlation also could in principle reflect an infrequency effect, but that possibility was ruled out by the lack of any negative loading in the vmPFC for the ChooseWin*P(explode) regressor. In contrast, as we predicted for the ChooseWin*P(explode) regressor, we found loss aversion effects in bilateral IFG and to some extent in the neighboring AI, as well as in the ACC. These regions showed a positive relationship (positive correlation, βs > 0) between brain activity and the probability of explosion in the ChooseWin condition. Thus, our main findings reveal opposing signals mainly in the ACC, IFG/AI, and left vmPFC, specifically implicating the ACC and IFG/AI regions as loss aversion signals and vmPFC as a reward-seeking signal.
Alternative interpretations
Despite our hypothesis-driven approach and results, our findings are subject to alternative interpretations. The ACC has been proposed to signal response conflict (Botvinick, Braver, & Barch, 2001). The uncertainty in decision making, and especially the tension between reward seeking and loss avoidance, might be construed as a state of conflict (Paulus et al., 2001). If this were the case, then higher explosion probabilities should be associated with greater conflict, regardless of whether participants chose to inflate or stop inflating. This was not the case: When participants chose to inflate at higher explosion probabilities, ACC activity decreased. Such a pattern is consistent with reduced loss avoidance, but we cannot entirely rule out the possibility that a conflict signal was driving the loss avoidance and was less active on those trials when participants chose to inflate.
Newer findings suggest that medial prefrontal cortex (mPFC), and ACC in particular, is involved in the learned predictions of the probability and timing of the various possible outcomes of an action (Alexander & Brown, 2011). This theory is consistent with a role for the ACC in predicting likely losses, and these predictions would in turn provide a straightforward basis for driving loss avoidance. Other findings regarding the IFG/AI region show that it is highly modulated by personality traits (Feinstein, Stein, & Paulus, 2006). Also, the IFG has been implicated in risk-related signals (Bossaerts, 2010; Christopoulos et al., 2009), as well as in playing an inhibitory role across various tasks requiring the suppression of response tendencies (Aron, Fletcher, & Bullmore, 2003; Aron, Robbins, & Poldrack, 2004). These results are consistent with a role for the IFG/AI region in inhibiting behaviors that are likely to lead to losses (but see Magno et al., 2006).
Limitations
In the present study, we did not focus on discriminating among the range of proposed definitions of risk (see, e.g., Yates, 1992) or intend to identify differences in brain activity representing various measurements of risk assessment. Risk has multiple facets, including the recent link made between awareness and risk assessment (Bossaerts, 2010), which is why we do not suggest that the IFG/AI and ACC are the only crucial brain regions involved in risky decision making. Future research should consider disentangling risk aversion versus loss aversion (Schonberg, Fox, & Poldrack, 2011), including the measurement of cognitive constructs, such as variance, loss probability, and the potential magnitude of a loss.
Conclusion
In this study, we used ChooseInflate*P(explode) and ChooseWin*P(explode) to test competing hypotheses of loss avoidance against the confounds of either reward seeking or the infrequency of risky choices. In general, the observed effects for these two parametrically modulated regressors may be consistent with one or more possible cognitive processes, but taken together, they constrain the solution space so that a single account remains the most defensible one.
Specifically, we found loss aversion signals in bilateral IFG and in ACC, in contrast to reward-seeking signals found in the left vmPFC. The pattern of effects supported our predictions that the IFG and ACC are associated with loss aversion (see Fig. 1A) and not with reward seeking (see Fig. 1B) or the infrequency of risky choices (see Fig. 1C). These results support previous findings implicating these regions in modulating risk appraisals (Krawitz et al., 2010). Additionally, our findings demonstrate the utility of using parametric modulation analysis in order to examine the neural correlates of risky decision making based on within-subjects correlations between BOLD activity and risk-taking choices and outcomes.
References
Alexander, W. H., & Brown, J. W. (2011). Medial prefrontal cortex as an action–outcome predictor. Nature Neuroscience, 14, 1338–1344.
Aron, A. R., Fletcher, P. C., Bullmore, E. T., Sahakian, B. J., & Robbins, T. W. (2003). Stop-signal inhibition disrupted by damage to right inferior frontal gyrus in humans. Nature Neuroscience, 6, 115–116.
Aron, A. R., Robbins, T. W., & Poldrack, R. A. (2004). Inhibition and the right inferior frontal cortex. Trends in Cognitive Sciences, 8, 170–177.
Bossaerts, P. (2010). Risk and risk prediction error signals in anterior insula. Brain Structure & Function, 214, 645–653.
Botvinick, M. M., Braver, T. S., Barch, D. M., Carter, C. S., & Cohen, J. D. (2001). Conflict monitoring and cognitive control. Psychological Review, 108, 624–652. doi:10.1037/0033-295X.108.3.624
Braver, T. S., Barch, D. M., Gray, J. R., Molfese, D. L., & Snyder, A. (2001). Anterior cingulate cortex and response conflict: Effects of frequency, inhibition and errors. Cerebral Cortex, 11, 825–836.
Brett, M., Anton, J., Valabrègue, R., & Poline, J. (2002). Marseille Boite à Region d’Interet (MarsBaR) [Computer software]. Retrieved from http://marsbar.sourceforge.net/
Brown, J. W., & Braver, T. S. (2005). Learned predictions of error likelihood in the anterior cingulate cortex. Science, 307, 1118–1121. doi:10.1126/science.1105783
Brown, J., & Braver, T. (2007). Risk prediction and aversion by anterior cingulate cortex. Cognitive, Affective, & Behavioral Neuroscience, 7, 266–277.
Christopoulos, G. I., Tobler, P. N., Bossaerts, P., Dolan, R. J., & Schultz, W. (2009). Neural correlates of value, risk, and risk aversion contributing to decision making under risk. Journal of Neuroscience, 29, 12574–12583.
Cohen, M. X., Heller, A. S., & Ranganath, C. (2005). Functional connectivity with anterior cingulate and orbitofrontal cortices during decision-making. Cognitive Brain Research, 23, 61–70.
Craig, A. D. (2002). How do you feel? Interoception: The sense of the physiological condition of the body. Nature Reviews Neuroscience, 3, 655–666. doi:10.1038/nrn894
Craig, A. D. (2009). How do you feel—now? The anterior insula and human awareness. Nature Reviews Neuroscience, 10, 59–70. doi:10.1038/nrn2555
Dale, A. M. (1999). Optimal experimental design for event-related fMRI. Human Brain Mapping, 8, 109–114.
Deppe, M., Schwindt, W., Kugel, H., Plassmann, H., & Kenning, P. (2005). Nonlinear responses within the medial prefrontal cortex reveal when specific implicit information influences economic decision making. Journal of Neuroimaging, 15, 171–182.
Ernst, M., & Paulus, M. P. (2005). Neurobiology of decision making: A selective review from a neurocognitive and clinical perspective. Biological Psychiatry, 58, 597–604.
Feinstein, J. S., Stein, M. B., & Paulus, M. P. (2006). Anterior insula reactivity during certain decisions is associated with neuroticism. Social Cognitive and Affective Neuroscience, 1, 136–142.
Fukui, H., Murai, T., Fukuyama, H., Hayashi, T., & Hanakawa, T. (2005). Functional activity related to risk anticipation during performance of the Iowa gambling task. NeuroImage, 24, 253–259.
Gehring, W. J., & Knight, R. T. (2000). Prefrontal–cingulate interactions in action monitoring. Nature Neuroscience, 3, 516.
Gehring, W. J., & Willoughby, A. R. (2002). The medial frontal cortex and the rapid processing of monetary gains and losses. Science, 295, 2279–2282.
Gray, M. A., & Critchley, H. D. (2007). Interoceptive basis to craving. Neuron, 54, 183–186.
Hayden, B. Y., Heilbronner, S. R., Pearson, J. M., & Platt, M. L. (2011). Surprise signals in anterior cingulate cortex: Neuronal encoding of unsigned reward prediction errors driving adjustment in behavior. Journal of Neuroscience, 31, 4178–4187.
Holmes, K. M., Bearden, C. E., Barguil, M., Fonseca, M., Serap Monkul, E., Nery, F. G., . . . Glahn, D. C. (2009). Conceptualizing impulsivity and risk taking in bipolar disorder: Importance of history of alcohol abuse. Bipolar Disorders, 11, 33–40.
Holroyd, C. B., & Coles, M. G. H. (2002). The neural basis of human error processing: Reinforcement learning, dopamine, and the error-related negativity. Psychological Review, 109, 679–709. doi:10.1037/0033-295X.109.4.679
Jabbi, M., & Keysers, C. (2008). Inferior frontal gyrus activity triggers anterior insula response to emotional facial expressions. Emotions, 8, 775–780.
Jessup, R. K., Busemeyer, J. R., & Brown, J. W. (2010). Error effects in anterior cingulate cortex reverse when error likelihood is high. Journal of Neuroscience, 30, 3467–3472.
Kiehl, K. A., Liddle, P. F., & Hopfinger, J. B. (2000). Error processing and the rostral anterior cingulate: An event-related fMRI study. Psychophysiology, 37, 216–223.
Knutson, B., Fong, G. W., Bennett, S. M., Adams, C. M., & Hommer, D. (2003). A region of mesial prefrontal cortex tracks monetarily rewarding outcomes: Characterization with rapid event-related fMRI. NeuroImage, 18, 263–272.
Koenigs, M., & Tranel, D. (2008). Prefrontal cortex damage abolishes brand-cued changes in cola preference. Social Cognitive and Affective Neuroscience, 3, 1–6.
Krain, A. L., Wilson, A. M., Arbuckle, R., Castellanos, F. X., & Milham, M. P. (2006). Distinct neural mechanisms of risk and ambiguity: A meta-analysis of decision-making. NeuroImage, 32, 477–484.
Krawitz, A., Fukunaga, R., & Brown, J. W. (2010). Anterior insula activity predicts the influence of positively framed messages on decision making. Cognitive, Affective, & Behavioral Neuroscience, 10, 392–405. doi:10.3758/CABN.10.3.392
Kuhnen, C. M., & Knutson, B. (2005). The neural basis of financial risk taking. Neuron, 47, 763–770.
Lancaster, J. L., Woldoroff, M. G., Parsons, L. M., Liotti, M., Freitas, C. S., Rainey, L., . . . Fox, P. T. (2000). Automated Talairach atlas labels for functional brain mapping. Human Brain Mapping, 10, 120–131.
Lawrence, N. S., Jollant, F., O’Daly, O., Zelaya, F., & Phillips, M. L. (2009). Distinct roles of prefrontal cortical subregions in the Iowa Gambling Task. Cerebral Cortex, 19, 1134–1143.
Ljuez, C. W., Read, J. P., Kahler, C. W., Richards, J. B., Ramsey, S. E., Stuart, G. L., . . . Brown, R. A. (2002). Evaluation of a behavioral measure of risk taking: The Balloon Analogue Risk Task (BART). Journal of Experimental Psychology: Applied, 8, 75–84.
Magno, E., Foxe, J. J., Molholm, S., Robertson, I. H., & Garavan, H. (2006). The anterior cingulate and error avoidance. Journal of Neuroscience, 26, 4769–4773.
Maldjian, J. A., Laurienti, P. J., Kraft, R. A., & Burdette, J. H. (2003). An automated method for neuroanatomic and cytoarchitectonic atlas-based interrogation of fMRI data sets. NeuroImage, 19, 1233–1239.
Martins, S. S., Tavares, H., da Silva Lobo, D. S., Galetti, A. M., & Gentil, V. (2004). Pathological gambling, gender, and risk-taking behaviors. Addictive Behaviors, 29, 1231–1235.
McClure, S. M., Li, J., Tomlin, D., Cypert, K. S., Montague, L. M., & Montague, P. R. (2004). Neural correlates of behavioral preference for culturally familiar drinks. Neuron, 44, 379–387.
Naqvi, N. H., & Bechara, A. (2009). The hidden island of addiction: The insula. Trends in Neurosciences, 32, 56–67. doi:10.1016/j.tins.2008.09.009
Naqvi, N. H., Rudrauf, D., Damasio, H., & Bechara, A. (2007). Damage to the insula disrupts addiction to cigarette smoking. Science, 315, 531–534. doi:10.1126/science.1135926
Nee, D. E., Kastner, S., & Brown, J. W. (2011). Functional heterogeneity of conflict, error, task-switching, and unexpectedness effects within medial prefrontal cortex. NeuroImage, 54, 528–540.
Oliveira, F. T. P., McDonald, J. J., & Goodman, D. (2007). Performance monitoring in the anterior cingulate is not all error related: Expectancy deviation and the representation of action–outcome associations. Journal of Cognitive Neuroscience, 19, 1994–2004.
Oppenheim, A. V., Schafer, R. W., & Buck, J. R. (1999). Discrete-time signal processing. Upper Saddle River, NJ: Prentice Hall.
Paulus, M. P., Hozack, N., Zauscher, B., McDowell, J. E., Frank, L., Brown, G. G., . . . Braff, D. L. (2001). Prefrontal, parietal, and temporal cortex networks underlie decision-making in the presence of uncertainty. NeuroImage, 13, 91–100.
Paulus, M. P., Rogalsky, C., Simmons, A., Feinstein, J. S., & Stein, M. B. (2003). Increased activation in the right insula during risk-taking decision making is related to harm avoidance and neuroticism. NeuroImage, 19, 1439–1448.
Péchaud, M., Jenkinson, M., & Smith, S. (2006). Brain Extraction Tool (BET) [Computer software]. Oxford, U.K.: Oxford University Centre for Functional MRI of the Brain.
Plassmann, H., Kenning, P., Deppe, M., Kugel, H., & Schwindt, W. (2008). How choice ambiguity modulates activity in brain areas representing brand preference: Evidence from consumer neuroscience. Journal of Consumer Behaviour, 7, 360–367.
Platt, M. L., & Huettel, S. A. (2008). Risky business: The neuroeconomics of decision making under uncertainty. Nature Neuroscience, 11, 398–403. doi:10.1038/nn2062
Preuschoff, K., Quartz, S. R., & Bossaerts, P. (2008). Human insula activation reflects risk prediction errors as well as risk. Journal of Neuroscience, 28, 2745–2752.
Rao, H., Korczykowski, M., Pluta, J., Hoang, A., & Detre, J. (2008). Neural correlates of voluntary and involuntary risk taking in the human brain: An fMRI study of the Balloon Analog Risk Task (BART). NeuroImage, 42, 902–910.
Rogers, R. D., Ramnani, N., Mackay, C., Wilson, J. L., Jezzard, P., Carter, C. S., . . . Smith, S. M. (2004). Distinct portions of anterior cingulate cortex and medial prefrontal cortex are activated by reward processing in separable phases of decision-making cognition. Biological Psychiatry, 55, 594–602.
Rothman, A. J., & Salovey, P. (1997). Shaping perceptions to motivate healthy behavior: The role of message framing. Psychological Bulletin, 121, 3–19.
Saaty, T. L. (1987). Risk—Its priority and probability: The analytic hierarchy process. Risk Analysis, 7, 159–172.
Schoenbaum, G., & Setlow, B. (2003). Lesions of nucleus accumbens disrupt learning about aversive outcomes. Journal of Neuroscience, 23, 9833–9841.
Schonberg, T., Fox, C. R., & Poldrack, R. A. (2011). Mind the gap: Bridging economic and naturalistic risk-taking with cognitive neuroscience. Trends in Cognitive Sciences, 15, 11–19.
Singer, T., Seymour, B., O’Doherty, J., Kaube, H., Dolan, R. J., & Frith, C. D. (2004). Empathy for pain involves the affective but not sensory components of pain. Science, 303, 1157–1161. doi:10.1126/science.1093535
Swann, A. C., Lijffijt, M., Lane, S. D., Steinberg, J. L., & Moeller, F. G. (2009). Increased trait-like impulsivity and course of illness in bipolar disorder. Bipolar Disorders, 11, 280–288.
Tversky, A., & Kahneman, D. (1981). The framing of decisions and the psychology of choice. Science, 211, 453–458. doi:10.1126/science.7455683
van Veen, V., Holroyd, C. B., Cohen, J. D., Stenger, V. A., & Carter, C. S. (2004). Errors without conflict: Implications for performance monitoring theories of anterior cingulate cortex. Brain and Cognition, 56, 267–276.
Verdejo-García, A., & Bechara, A. (2009). A somatic marker theory of addiction. Neuropharmacology, 56, 48–62.
Weber, E., Blais, A.-R., & Betz, N. E. (2002). A domain-specific risk-attitude scale: Measuring risk perceptions and risk behaviors. Journal of Behavioral Decision Making, 15, 263–290. doi:10.1002/bdm.414
Wellcome Trust Centre for Neuroimaging. (2005). Statistical parametric mapping (SPM) [Computer software]. London, U.K.: Wellcome Trust Centre for Neuroimaging.
Yates, J. F. (1992). Risk-taking behavior. Chichester, U.K.: Wiley.
Author note
This research was supported by National Institute on Alcohol Abuse and Alcoholism (NIAAA) Grant K99 AA017877 to T.B., with additional support from NIAAA Grants R01 AA13650 to Peter R. Finn and DA026457 to J.W.B. We thank Peter R. Finn for use of his facility, and Thea Atwood and Rebecca Ward for help with the data collection.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fukunaga, R., Brown, J.W. & Bogg, T. Decision making in the Balloon Analogue Risk Task (BART): Anterior cingulate cortex signals loss aversion but not the infrequency of risky choices. Cogn Affect Behav Neurosci 12, 479–490 (2012). https://doi.org/10.3758/s13415-012-0102-1
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13415-012-0102-1