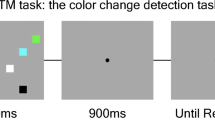
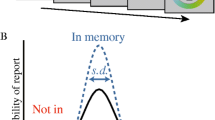
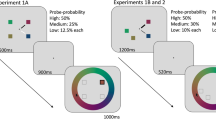
Abstract
Visual short-term memory (VSTM), the ability to store information no longer visible, is essential for human behavior. VSTM limits vary across the population and are correlated with overall cognitive ability. It has been proposed that low-memory individuals are unable to select only relevant items for storage and that these limitations are greatest when memory demands are high. However, it is unknown whether these effects simply reflect task difficulty and whether they impact the quality of memory representations. Here we varied the number of items presented, or set size, to investigate the effect of memory demands on the performance of visual short-term memory across low- and high-memory groups. Group differences emerged as set size exceeded memory limits, even when task difficulty was controlled. In a change-detection task, the low-memory group performed more poorly when set size exceeded their memory limits. We then predicted that low-memory individuals encoding items beyond measured memory limits would result in the degraded fidelity of memory representations. A continuous report task confirmed that low, but not high, memory individuals demonstrated decreased memory fidelity as set size exceeded measured memory limits. The current study demonstrates that items held in VSTM are stored distinctly across groups and task demands. These results link the ability to maintain high quality representations with overall cognitive ability.




Similar content being viewed by others
References
Adam, K. C., Mance, I., Fukuda, K., & Vogel, E. K. (2015). The contribution of attentional lapses to individual differences in visual working memory capacity. Journal of Cognitive Neuroscience, 27, 1601–1616.
Alvarez, G. A., & Cavanagh, P. (2004). The capacity of visual short-term memory is set both by visual information load and by number of objects. Psychological Science, 15, 106–111.
Awh, E., Barton, B., & Vogel, E. K. (2007). Visual working memory represents a fixed number of items regardless of complexity. Psychological Science, 18, 622–628.
Awh, E., & Vogel, E. K. (2008). The bouncer in the brain. Nature Neuroscience, 11, 5–6.
Barton, B., Ester, E. F., & Awh, E. (2009). Discrete resource allocation in visual working memory. Journal of Experimental Psychology: Human Perception and Performance, 35, 1359–1367.
Bays, P. M., Catalao, R. F. G., & Husain, M. (2009). The precision of visual working memory is set by allocation of a shared resource. Journal of Vision, 9, 1–11.
Bays, P. M., & Husain, M. (2008). Dynamic shifts of limited working memory resources in human vision. Science, 321, 851–854.
Brainard, D. H. (1997). The Psychophysics Toolbox. Spatial Vision, 10, 433–436.
Conway, A. R., Cowan, N., Bunting, M. F., Therriault, D. J., & Minkoff, S. R. (2002). A latent variable analysis of working memory capacity, short-term memory capacity, processing speed, and general fluid intelligence. Intelligence, 30, 163–183.
Cowan, N. (2001). The magical number 4 in short-term memory: A reconsideration of mental storage capacity. Behavioral and Brain Sciences, 24, 87–185.
Cowan, N. (2005). Working memory capacity limits in a theoretical context. In C. Izawa & N. Ohta (Eds.), Human learning and memory: Advances in theory and application (pp. 155–175). Erlbaum.
Cowan, N., Fristoe, N. M., Elliott, E. M., Brunner, R. P., & Saults, J. S. (2006). Scope of attention, control of attention, and intelligence in children and adults. Memory & Cognition, 34, 1754–1768.
Dux, P. E., & Marois, R. (2008). Distractor inhibition predicts individual differences in the attentional blink. PLOS ONE, 3, e3330.
Engle, R. W., Kane, M. J., & Tuholski, S. W. (1999). Individual differences in working memory capacity and what they tell us about controlled attention, general fluid intelligence, and functions of the prefrontal cortex. In A. Miyake & P. Shah (Eds.), Models of working memory: Mechanisms of active maintenance and executive control (pp. 102–134). Cambridge University Press.
Ester, E. F., Sprague, T. C., & Serences, J. T. (2015). Parietal and frontal cortex encode stimulus-specific mnemonic representations during visual working memory. Neuron, 87, 893–905.
Fukuda, K., & Vogel, E. K. (2009). Human variation in overriding attentional capture. Journal of Neuroscience, 29, 8726–8733.
Fukuda, K., & Vogel, E. K. (2011). Individual differences in recovery time from attentional capture. Psychological Science, 22, 361–368.
Fukuda, K., Vogel, E., Mayr, U., & Awh, E. (2010). Quantity, not quality: The relationship between fluid intelligence and working memory capacity. Psychonomic Bulletin & Review, 17, 673–679.
Fukuda, K., Woodman, G. F., & Vogel, E. K. (2015). Individual differences in visual working memory capacity: Contributions of attentional control to storage. In P. Jolicoeur, C. Lefebvre, & J. Martinez-Trujillo (Eds.), Mechanisms of sensory working memory: Attention and performance XXV (pp. 105–119). ScienceDirect.
Gaspar, J. M., Christie, G. J., Prime, D. J., Jolicœur, P., & McDonald, J. J. (2016). Inability to suppress salient distractors predicts low visual working memory capacity. Proceedings of the National Academy of Sciences, 113, 3693–3698.
Hardman, K. O., Vergauwe, E., & Ricker, T. J. (2017). Categorical working memory representations are used in delayed estimation of continuous colors. Journal of Experimental Psychology: Human Perception and Performance, 43(1), 30–54.
Harrison, S. A., & Tong, F. (2009). Decoding reveals the contents of visual working memory in early visual areas. Nature, 458, 632–635.
Jaeggi, S. M., Buschkuehl, M., Jonides, J., & Perrig, W. J. (2008). Improving fluid intelligence with training on working memory. Proceedings of the National Academy of Sciences, 105, 6829–6833.
Kamitani, Y., & Tong, F. (2005). Decoding the visual and subjective contents of the human brain. Nature Neuroscience, 8, 679–685.
Kumle, L., Võ, M. L. H., & Draschkow, D. (2021). Estimating power in (generalized) linear mixed models: An open introduction and tutorial in R. Behavior Research Methods, 53(6), 2528–2543.
Luck, S. J., & Vogel, E. K. (1997). The capacity of visual working memory for features and conjunctions. Nature, 309, 279–281.
MacCallum, R. C., Zhang, S., Preacher, K. J., & Rucker, D. D. (2002). On the practice of dichotomization of quantitative variables. Psychological Methods, 7(1), 19–40.
McNab, F., & Klingberg, T. (2008). Prefrontal cortex and basal ganglia control access to working memory. Nature Neuroscience, 11, 103–107.
Murray, J. D., Bernacchia, A., Roy, N. A., Constantinidis, C., Romo, R., & Wang, X. J. (2017). Stable population coding for working memory coexists with heterogeneous neural dynamics in prefrontal cortex. Proceedings of the National Academy of Sciences, 114(2), 394–399.
Pashler, H. (1988). Familiarity and visual change detection. Perception & Psychophysics, 44, 369–378.
Peirce, J. W. (2009). Generating stimuli for neuroscience using PsychoPy. Frontiers in Neuroinformatics, 2, 343
Pelli, D. G. (1997). The VideoToolbox software for visual psychophysics: Transforming numbers into movies. Spatial Vision, 10, 437–442.
Pinheiro, J., Bates, D., R Core Team. (2023). Nlme: linear and nonlinear mixed effects models. R package version 3.1-162, https://CRAN.R-project.org/package=nlme
Rademaker, R. L., Chunharas, C., & Serences, J. T. (2019). Coexisting representations of sensory and mnemonic information in human visual cortex. Nature Neuroscience, 22, 1336–1344.
Robison, M. K., Miller, A. L., & Unsworth, N. (2018). Individual differences in working memory capacity and filtering. Journal of Experimental Psychology: Human Perception and Performance, 44, 1038–1053.
Schurgin, M. W., Wixted, J. T., & Brady, T. F. (2020). Psychophysical scaling reveals a unified theory of visual memory strength. Nature Human Behaviour, 4(11), 1156–1172.
Serences, J. T., Ester, E. F., Vogel, E. K., & Awh, E. (2009). Stimulus-specific delay activity in human primary visual cortex. Psychological Science, 20, 207–214.
Sheremata, S., & Shomstein, S. (2014). Hemifield asymmetries differentiate VSTM for single-and multiple-feature objects. Attention, Perception, & Psychophysics, 76, 1609–1619.
Shipstead, Z., Redick, T. S., Hicks, K. L., & Engle, R. W. (2012). The scope and control of attention as separate aspects of working memory. Memory, 20, 608–628.
Tanner, W. P., Jr., & Swets, J. A. (1954). A decision-making theory of visual detection. Psychological Review, 61, 401–409.
Todd, J. J., & Marois, R. (2004). Capacity limit of visual short-term memory in human posterior parietal cortex. Nature, 428, 751–754.
Unsworth, N., Fukuda, K., Awh, E., & Vogel, E. K. (2014). Working memory and fluid intelligence: Capacity, attention control, and secondary memory retrieval. Cognitive Psychology, 71, 1–26.
Unsworth, N., Fukuda, K., Awh, E., & Vogel, E. K. (2015). Working memory delay activity predicts individual differences in cognitive abilities. Journal of Cognitive Neuroscience, 27, 853–865.
Van den Berg, R., Shin, H., Chou, W. C., George, R., & Ma, W. J. (2012). Variability in encoding precision accounts for visual short-term memory limitations. Proceedings of the National Academy of Sciences, 109, 8780–8785.
Vogel, E. K., & Machizawa, M. G. (2004). Neural activity predicts individual differences in visual working memory capacity. Nature, 428, 748–751.
Vogel, E. K., McCollough, A. W., & Machizawa, M. G. (2005). Neural measures reveal individual differences in controlling access to working memory. Nature, 438, 500–503.
Vogel, E. K., Woodman, G. F., & Luck, S. J. (2001). Storage of features, conjunctions, and objects in visual working memory. Journal of Experimental Psychology: Human Perception and Performance, 27, 92–114.
von Allmen, D. Y., Wurmitzer, K., Martin, E., & Klaver, P. (2013). Neural activity in the hippocampus predicts individual visual short-term memory capacity. Hippocampus, 23, 606–615.
Wilken, P., & Ma, W. J. (2004). A detection theory account of change detection. Journal of Vision, 4, 1120–1135.
Zhang, W., & Luck, S. J. (2008). Discrete fixed-resolution representations in visual working memory. Nature, 453, 233–235.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
None
Additional information
Open practices statement
Neither experiment was preregistered. The data of this study are available from the corresponding author, Y.S.S, upon reasonable request.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Public significance statement
Visual short-term memory selects and holds a few items from the external world by constructing stable internal images for further analysis. However, the amount of information available is severely limited, with limits varying within the population. In the present study, we investigate (1) whether limits between individuals exist when controlling for overall task difficulty and (2) whether memory fidelity is affected by differences in memory limits. We demonstrate that when asked to remember many items, individuals are less likely to detect a change in the remembered items and remember more items at a cost of memory fidelity.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shin, Y.S., Sheremata, S.L. When remembering less is more: Unfiltered items are associated with reduced memory fidelity in visual short-term memory. Atten Percept Psychophys 86, 1248–1258 (2024). https://doi.org/10.3758/s13414-024-02891-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13414-024-02891-w