Abstract
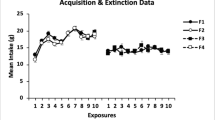
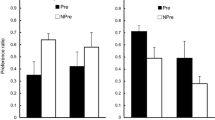
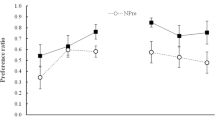
In two experiments, we examined the effect of reversal learning on the status of initially learned associations. In Experiment 1, thirsty rats were first taught to associate one flavor with sucrose and another flavor with Polycose. These relations were then reversed in a subsequent phase. One of the nutrients was then devalued by being paired with lithium chloride. The results of a two-bottle flavor-choice test revealed that the most recently learned associations governed performance. In Experiment 2, we aimed to discern whether the initially learned associations in Experiment 1 were weakened or masked by reversal learning. In order to address this question, either a 1-day (Group Immediate) or a 21-day (Group Delayed) retention interval was interpolated between the reversal and devaluation phases. Subsequent flavor-choice tests revealed that Group Immediate avoided the flavor most recently associated with the devalued nutrient but that Group Delayed avoided the flavor that was initially associated with the devalued nutrient. These findings suggest that the second-learned associations do not erase, but transiently mask, the first-learned associations, which subsequently recover over a retention interval. These results suggest a parallel in the mechanisms of extinction and reversal learning.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aguado, L., Symonds, M., & Hall, G. (1994). Interval between preexposure and test determines the magnitude of latent inhibition: Implications for an interference account. Learning & Behavior, 22, 188–194.
Albertella, L., & Boakes, R. A. (2006). Persistence of conditioned flavor preferences is not due to inadvertent food reinforcement. Journal of Experimental Psychology: Animal Behavior Processes, 32, 386–395.
Bouton, M. E. (1991). Context and retrieval in extinction and in other examples of interference in simple associative learning. In L. Dachowski & C. F. Flaherty (Eds.), Current topics in animal learning: Brain, emotion, and cognition (pp. 25–53). Hillsdale, NJ: Erlbaum.
Bouton, M. E. (2004). Context and behavioral processes in extinction. Learning & Memory, 11, 485–494.
Bouton, M. E., & Peck, C. A. (1992). Spontaneous recovery in cross-motivational transfer (counterconditioning). Learning & Behavior, 20, 313–321.
Bouton, M. E., Westbrook, R. F., Corcoran, K. A., & Maren, S. (2006). Contextual and temporal modulation of extinction: Behavioral and biological mechanisms. Biological Psychiatry, 60, 352–360.
Cappell, H., & Le {FnBlanc}, A. E. (1977). Parametric investigations of the effects of prior exposure to amphetamine and morphine on conditioned gustatory aversion. Psychopharmacology, 51, 265–271.
Delamater, A. R. (1996). Effects of several extinction treatments upon the integrity of Pavlovian stimulus-outcome associations. Learning & Behavior, 24, 437–449.
Delamater, A. R. (2004). Experimental extinction in Pavlovian conditioning: Behavioural and neuroscience perspectives. Quarterly Journal of Experimental Psychology, 57B, 97–132.
Delamater, A. R. (2007a). Extinction of conditioned flavor preferences. Journal of Experimental Psychology: Animal Behavior Processes, 33, 160–171.
Delamater, A. R. (2007b). The role of the orbitofrontal cortex in sensory-specific encoding of associations in Pavlovian and instrumental conditioning. In G. Schoenbaum, J. A. Gottfried, E. A. Murray, & S. J. Ramus (Eds.), Linking affect to action: Critical contributions of the orbitofrontal cortex (Annals of the New York Academy of Sciences, Vol. 1121, pp. 152–173). New York: New York Academy of Sciences.
Delamater, A. R., Campese, V., LoLordo, V. M., & Sclafani, A. (2006). Unconditioned stimulus devaluation effects in nutrient-conditioned flavor preferences. Journal of Experimental Psychology: Animal Behavior Processes, 32, 295–306.
Drucker, D. B., Ackroff, K., & Sclafani, A. (1994). Nutrient-conditioned flavor preference and acceptance in rats: Effects of deprivation state and nonreinforcement. Physiology & Behavior, 56, 701–707.
Dwyer, D. M. (2005). Reinforcer devaluation in palatability-based learned flavor preferences. Journal of Experimental Psychology: Animal Behavior Processes, 31, 487–492.
Elizalde, G., & Sclafani, A. (1990). Flavor preferences conditioned by intragastric Polycose infusions: A detailed analysis using an electronic esophagus preparation. Physiology & Behavior, 47, 63–77.
Hall, G. (1996). Learning about associatively activated stimulus representations: Implications for acquired equivalence and perceptual learning. Learning & Behavior, 24, 233–255.
Harris, J. A., Gorissen, M. C., Bailey, G. K., & Westbrook, R. F. (2000). Motivational state regulates the content of learned flavor preferences. Journal of Experimental Psychology: Animal Behavior Processes, 26, 15–30.
Harris, J. A., Shand, F. L., Carroll, L. Q., & Westbrook, R. F. (2004). Persistence of preference for a flavor presented in simultaneous compound with sucrose. Journal of Experimental Psychology: Animal Behavior Processes, 30, 177–189.
Holland, P. C. (1981). Acquisition of representation-mediated conditioned food aversions. Learning & Motivation, 12, 1–18.
Lipatova, O., Wheeler, D. S., Vadillo, M. A., & Miller, R. R. (2006). Recency-to-primacy shift in cue competition. Journal of Experimental Psychology: Animal Behavior Processes, 32, 396–406.
Lubow, R. E., & De la Casa, L. G. (2002). Superlatent inhibition and spontaneous recovery: Differential effects of pre- and postconditioning CS-alone presentations after long delays in different contexts. Learning & Behavior, 30, 376–386.
Nelson, J. B. (2002). Context specificity of excitation and inhibition in ambiguous stimuli. Learning & Motivation, 33, 284–310.
Quirk, G. J., & Mueller, D. (2008). Neural mechanisms of extinction learning and retrieval. Neuropsychopharmacology, 33, 56–72.
Randich, A., & LoLordo, V. M. (1979). Associative and nonassociative theories of the UCS preexposure phenomenon: Implications for Pavlovian conditioning. Psychological Bulletin, 86, 523–548.
Rescorla, R. A. (1992). Associations between an instrumental discriminative stimulus and multiple outcomes. Journal of Experimental Psychology: Animal Behavior Processes, 18, 95–104.
Rescorla, R. A. (1995). Full preservation of a response-outcome association through training with a second outcome. Quarterly Journal of Experimental Psychology, 48B, 252–261.
Rescorla, R. A. (1996). Preservation of Pavlovian associations through extinction. Quarterly Journal of Experimental Psychology, 49B, 245–258.
Rescorla, R. A. (1997). Spontaneous recovery after Pavlovian conditioning with multiple outcomes. Learning & Behavior, 25, 99–107.
Rescorla, R. A. (2004). Spontaneous recovery. Learning & Memory, 11, 501–509.
Rhodes, S. E. V., & Killcross, [A.] S. (2004). Lesions of rat infralimbic cortex enhance recovery and reinstatement of an appetitive Pavlovian response. Learning & Memory, 11, 611–616.
Rhodes, S. E. V., & Killcross, A. S. (2007). Lesions of rat infralimbic cortex enhance renewal of extinguished appetitive Pavlovian responding. European Journal of Neuroscience, 25, 2498–2503.
Rosas, J. M., & Bouton, M. E. (1996). Spontaneous recovery after extinction of a conditioned taste aversion. Learning & Behavior, 24, 341–348.
Rosas, J. M., & Bouton, M. E. (1998). Context change and retention interval can have additive, rather than interactive, effects after taste aversion extinction. Psychonomic Bulletin & Review, 5, 79–83.
Santini, E., Muller, R. U., & Quirk, G. J. (2001). Consolidation of extinction learning involves transfer from NMDA-independent to NMDA-dependent memory. Journal of Neuroscience, 21, 9009–9017.
Schoenbaum, G., Chiba, A. A., & Gallagher, M. (1999). Neural encoding in orbitofrontal cortex and basolateral amygdala during olfactory discrimination learning. Journal of Neuroscience, 19, 1876–1884.
Schoenbaum, G., Saddoris, M. P., & Stalnaker, T. A. (2007). Reconciling the roles of orbitofrontal cortex in reversal learning and the encoding of outcome expectancies. In G. Schoenbaum, J. A. Gottfried, E. A. Murray, & S. J. Ramus (Eds.), Linking affect to action: Critical contributions of the orbitofrontal cortex (Annals of the New York Academy of Sciences, Vol. 1121, pp. 320–335). New York: New York Academy of Sciences.
Swartzentruber, D., & Bouton, M. E. (1992). Context sensitivity of conditioned suppression following preexposure to the conditioned stimulus. Learning & Behavior, 20, 97–103.
Urushihara, K., Wheeler, D. S., & Miller, R. R. (2004). Outcome pre- and postexposure effects: Retention interval interacts with primacy and recency. Journal of Experimental Psychology: Animal Behavior Processes, 30, 283–298.
Author information
Authors and Affiliations
Corresponding author
Additional information
This research was supported by NIMH Grant 065947 awarded to A.R.D. The authors gratefully acknowledge Leib Litman for his constructive input on these data.
Rights and permissions
About this article
Cite this article
Scarlet, J., Campese, V. & Delamater, A.R. Sensory-specific associations in flavor-preference reversal learning. Learning & Behavior 37, 179–187 (2009). https://doi.org/10.3758/LB.37.2.179
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.3758/LB.37.2.179