Abstract
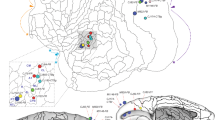
It is generally agreed that information flow through the cortex is constrained by a hierarchical architecture. Recent experimental evidence suggests that projections descending the hierarchy and targeting the primary visual cortex (area V1) may play an essential role in perceptual processes. We have, therefore, reexamined feedback projections to area V1, using retrograde tracer injections in this area. In addition to well-known areas, quantification of labeling in higher cortical areas reveals a number of hitherto unknown long-distance feedback connections originating from auditory (A1), multisensory (STP) cortices, but also from a perirhinal area (36). These feedback projections from advanced cortical stations, a global feature shared by areas that belong to the ventral visual stream, could play an important role in early multisensory integration and spatial awareness and could provide the physical substrate for the involvement of area V1 in visual consciousness.
Article PDF
Similar content being viewed by others
References
Allman, J., Miezin, R., & McGuinness, E. (1985). Stimulus specific responses from beyond the classical receptive field: Neurophysiological mechanisms for local-global comparisons in visual neurons. Annual Review of Neuroscience, 8, 407–430.
Amaral, D. G., Behniea, H., & Kelly, J. L. (2003). Topographic organization of projections from the amygdala to the visual cortex in the macaque monkey. Neuroscience, 118, 1099–1120.
Angelucci, A., Clasca, F., & Sur, M. (1998). Brainstem inputs to the ferret medial geniculate nucleus and the effect of early deafferentation on novel retinal projections to the auditory thalamus. Journal of Comparative Neurology, 400, 417–439.
Angelucci, A., Levitt, J. B., Walton, E. J., Hupe, J. M., Bullier, J., & Lund, J. S. (2002). Circuits for local and global signal integration in primary visual cortex. Journal of Neuroscience, 22, 8633–8646.
Barone, P., Batardière, A., Knoblauch, K., & Kennedy, H. (2000). Laminar distribution of neurons in extrastriate areas projecting to visual areas V1 and V4 correlates with the hierarchical rank and indicates the operation of a distance rule. Journal of Neuroscience, 20, 3263–3281.
Barone, P., Clarey, J. C., Irons, W. A., & Imig, T. J. (1996). Cortical synthesis of azimuth-sensitive single-unit responses with nonmonotonic level tuning: A thalamocortical comparison in the cat. Journal of Neurophysiology, 75, 1206–1220.
Bartolomeo, P. (2002). The relationship between visual perception and visual mental imagery: A reappraisal of the neuropsychological evidence. Cortex, 38, 357–378.
Batardière, A., Barone, P., Dehay, C., & Kennedy, H. (1998). Areaspecific laminar distribution of cortical feedback neurons projecting to cat area 17: Quantitative analysis in the adult and during ontogeny. Journal of Comparative Neurology, 396, 493–510.
Benevento, L. A., Fallon, J., Davis, B. J., & Rezak, M. (1977). Auditory-visual interaction in single cells in the cortex of the superior temporal sulcus and the orbital frontal cortex of the macaque monkey. Experimental Neurology, 57, 849–872.
Bisiach, E. V., & Vallar, G. (1988). Hemineglect in humans. In F. Boller & J. Grafman (Eds.), Handbook of Neuropsychology (Vol. 1, pp. 195–222). Amsterdam: Elsevier.
Bruce, C., Desimone, R., & Gross, C. G. (1981). Visual properties of neurons in a polysensory area in superior temporal sulcus of the macaque. Journal of Neurophysiology, 46, 369–384.
Buchel, C., Price, C., Frackowiak, R. S., & Friston, K. (1998). Different activation patterns in the visual cortex of late and congenitally blind subjects. Brain, 121 (Pt. 3), 409–419.
Bullier, J., Girard, P., & Salin, P. A. (1994). The role of area 17 in the transfer of information to extrastriate visual cortex. In A. Peters & K. S. Rockland (Eds.), Primary visual cortex in primates (Vol. 10, pp. 301–330). New York: Plenum.
Bullier, J., Kennedy, H., & Salinger, W. (1984). Branching and laminar origin of projections between visual cortical areas in the cat. Journal of Comparative Neurology, 228, 329–341.
Calvert, G. A., Brammer, M. J., & Iversen, S. D. (1998). Crossmodal identification. Trends in Cognitive Sciences, 7, 287–253.
Calvert, G. A., Bullmore, E. T., Brammer, M. J., Campbell, R., Williams, S. C. R., McGuire, P. K., Woodruff, P. W. R., Iversen, S. D., & David, A. S. (1997). Activation of auditory cortex during silent lipreading. Science, 276, 593–596.
Calvert, G. A., Campbell, R., & Brammer, M. J. (2000). Evidence from functional magnetic resonance imaging of crossmodal binding in the human heteromodal cortex. Current Biology, 10, 649–657.
Calvert, G. A., Hansen, P. C., Iversen, S. D., & Brammer, M. J. (2001). Detection of audio-visual integration sites in humans by application of electrophysiological criteria to the BOLD effect. Neuro-Image, 14, 427–438.
Cauller, L. (1995). Layer I of primary sensory neocortex: Where topdown converges upon bottom-up. Behavioural Brain Research, 71, 163–170.
Cohen, L. G., Weeks, R. A., Sadato, N., Celnik, P., Ishii, K., & Hallett, M. (1999). Period of susceptibility for cross-modal plasticity in the blind. Annals of Neurology, 45, 451–460.
Cusick, C. G. (1997). The superior temporal polysensory region in monkeys. In E. G. Jones & A. Peters (Eds.), Cerebral cortex (Vol. 12, pp. 435–468). New York: Plenum.
Dehay, C., Kennedy, H., & Bullier, J. (1988). Characterization of transient cortical projections from auditory, somatosensory, and motor cortices to visual areas 17, 18, and 19 in the kitten. Journal of Comparative Neurology, 272, 68–89.
Dehner, L. R., Keniston, L. P., Clemo, H. R., & Meredith, M. A. (2004). Cross-modal circuitry between auditory and somatosensory areas of the cat anterior ectosylvian sulcal cortex: A “new” inhibitory form of multisensory convergence. Cerebral Cortex, 14, 387–403.
Deibert, E., Kraut, M., Kremen, S., & Hart, J., Jr. (1999). Neural pathways in tactile object recognition. Neurology, 52, 1413–1417.
Doty, R. W. (1983). Nongeniculate afferents to striate cortex in macaques. Journal of Comparative Neurology, 218, 159–173.
Falchier, A., Clavagnier, S., Barone, P., & Kennedy, H. (2002). Anatomical evidence of multimodal integration in primate striate cortex. Journal of Neuroscience, 22, 5749–5759.
Farah, J. M. (1995). The neural bases of mental imagery. In M. S. Gazzaniga (Ed.), The cognitive neurosciences (pp. 963–974). Cambridge, MA: MIT Press.
Felleman, D. J., & Van Essen, D. C. (1991). Distributed hierarchical processing in the primate cerebral cortex. Cerebral Cortex, 1, 1–47.
Foxe, J. J., & Simpson, G. V. (2002). Flow of activation from V1 to frontal cortex in humans: A framework for defining “early” visual processing. Experimental Brain Research, 142, 139–150.
Giard, M. H., & Peronnet, F. (1999). Auditory-visual integration during multimodal object recognition in humans: A behavioral and electrophysiological study. Journal of Cognitive Neuroscience, 11, 473–490.
Giraud, A. L., Price, C. J., Graham, J. M., Truy, E., & Frackowiak, R. S. (2001). Cross-modal plasticity underpins language recovery after cochlear implantation. Neuron, 30, 657–663.
Goldman-Rakic, P. S. (1988). Topography of cognition: Parallel distributed networks in primate association cortex. Annual Review of Neuroscience, 11, 137–156.
Goldring, J. E., Dorris, M. C., Corneil, B. D., Ballantyne, P. A., & Munoz, D. P. (1996). Combined eye-head gaze shifts to visual and auditory targets in humans. Experimental Brain Research, 111, 68–78.
Heffner, R. S., & Heffner, H. E. (1992). Visual factors in sound localization in mammals. Journal of Comparative Neurology, 317, 219–232.
Hikosaka, K., Iwai, E., Saito, H., & Tanaka, K. (1988). Polysensory properties of neurons in the anterior bank of the caudal superior temporal sulcus of the macaque monkey. Journal of Neurophysiology, 60, 1615–1637.
Hochstein, S., & Ahissar, M. (2002). View from the top: Hierarchies and reverse hierarchies in the visual system. Neuron, 36, 791–804.
Hupé, J. M., James, A. C., Payne, B. R., Lomber, S. G., Girard, P., & Bullier, J. (1998). Cortical feedback improves discrimination between figure and background by V1, V2 and V3 neurons. Nature, 394, 784–787.
Innocenti, G. M., Berbel, P., & Clarke, S. (1988). Development of projections from auditory to visual areas in the cat. Journal of Comparative Neurology, 272, 242–259.
Ishai, A., & Sagi, D. (1995). Common mechanisms of visual imagery and perception. Science, 268, 1772–1774.
Jones, E. G., & Powell, T. P. (1970). An anatomical study of converging sensory pathways within the cerebral cortex of the monkey. Brain, 93, 793–820.
Juan, C. H., & Walsh, V. (2003). Feedback to V1: A reverse hierarchy in vision. Experimental Brain Research, 150, 259–263.
Kaas, J. H., & Hackett, T. A. (2000). Subdivisions of auditory cortex and processing streams in primates. Proceedings of the National Academy of Sciences, 97, 11793–11799.
Karnath, H. O., Ferber, S., & Himmelbach, M. (2001). Spatial awareness is a function of the temporal not the posterior parietal lobe. Nature, 411, 950–953.
Karnath, H. O., & Perenin, M. T. (1998). Tactile exploration of peripersonal space in patients with neglect. NeuroReport, 9, 2273–2277.
Kastner, S., & Ungerleider, L. G. (2000). Mechanisms of visual attention in the human cortex. Annual Review of Neuroscience, 23, 315–341.
Kennedy, H., & Bullier, J. (1985). A double-labeling investigation of the afferent connectivity to cortical areas V1 and V2 of the macaque monkey. Journal of Neuroscience, 5, 2815–2830.
Klein, I., Paradis, A. L., Poline, J. B., Kosslyn, S. M., & Le Bihan, D. (2000). Transient activity in the human calcarine cortex during visualmental imagery: An event-related fMRI study. Journal of Cognitive Neuroscience, 12(Suppl. 2), 15–23.
Korte, M., & Rauschecker, J. P. (1993). Auditory spatial tuning of cortical neurons is sharpened in cats with early blindness. Journal of Neurophysiology, 70, 1717–1721.
Kosaki, H., Hashikawa, T., He, J., & Jones, E. G. (1997). Tonotopic organization of auditory cortical fields delineated by parvalbumin immunoreactivity in macaque monkeys. Journal of Comparative Neurology, 386, 304–316.
Kosslyn, S. M., Ganis, G., & Thompson, W. L. (2001). Neural foundations of imagery. Nature Reviews Neuroscience, 2, 635–642.
Kreiman, G., Koch, C., & Fried, I. (2000). Imagery neurons in the human brain. Nature, 408, 357–361.
Lamme, V. A., & Roelfsema, P. R. (2000). The distinct modes of vision offered by feedforward and recurrent processing. Trends in Neurosciences, 23, 571–579.
Leopold, D. A., & Logothetis, N. K. (1996). Activity changes in early visual cortex reflect monkeys’ percepts during binocular rivalry. Nature, 379, 549–553.
Linden, J. F., Grunewald, A., & Andersen, R. A. (1999). Responses to auditory stimuli in macaque lateral intraparietal area:. II. Behavioral modulation. Journal of Neurophysiology, 82, 343–358.
Luh, K. E., Butter, C. M., & Buchtel, H. A. (1986). Impairments in orienting to visual stimuli in monkeys following unilateral lesions of the superior sulcal polysensory cortex. Neuropsychologia, 24, 461–470.
Lyon, D. C., & Kaas, J. H. (2002). Evidence from V1 connections for both dorsal and ventral subdivisions of V3 in three species of New World monkeys. Journal of Comparative Neurology, 449, 281–297.
Macaluso, E., Frith, C. D., & Driver, J. (2000). Modulation of human visual cortex by crossmodal spatial attention. Science, 289, 1206–1208.
Martinich, S., Pontes, M. N., & Rocha-Miranda, C. E. (2000). Patterns of corticocortical, corticotectal, and commissural connections in the opossum visual cortex. Journal of Comparative Neurology, 416, 224–244.
McDonald, J. J., Teder-Salejarvi, W. A., & Hillyard, S. A. (2000). Involuntary orienting to sound improves visual perception. Nature, 407, 906–908.
Mehta, A. D., Ulbert, I., & Schroeder, C. E. (2000a). Intermodal selective attention in monkeys: I: Distribution and timing of effects across visual areas. Cerebral Cortex, 10, 343–358.
Mehta, A. D., Ulbert, I., & Schroeder, C. E. (2000b). Intermodal selective attention in monkeys: II: Physiological mechanisms of modulation. Cerebral Cortex, 10, 359–370.
Miller, M. W., & Vogt, B. A. (1984). Direct connections of rat visual cortex with sensory, motor, and association cortices. Journal of Comparative Neurology, 226, 184–202.
Miyashita, Y. (1995). How the brain creates imagery: Projection to primary visual cortex. Science, 268, 1719–1720.
Montero, V. M. (1993). Retinotopy of cortical connections between the striate cortex and extrastriate visual areas in the rat. Experimental Brain Research, 94, 1–15.
Morel, A., Garraghty, P. E., & Kaas, J. H. (1993). Tonotopic organization, architectonic fields, and connections of auditory cortex in macaque monkeys. Journal of Comparative Neurology, 335, 437–459.
Motter, B. C. (1993). Focal attention produces spatially selective processing in visual cortical areas V1, V2, and V4 in the presence of competing stimuli. Journal of Neurophysiology, 70, 909–919.
Nothdurft, H. C., Gallant, J. L., & Van Essen, D. C. (1999). Response modulation by texture surround in primate area V1: Correlates of “popout” under anesthesia. Visual Neuroscience, 16, 15–34.
Nowak, L. G., Munk, M. H., James, A. C., Girard, P., & Bullier, J. (1999). Cross-correlation study of the temporal interactions between areas V1 and V2 of the macaque monkey. Journal of Neurophysiology, 81, 1057–1074.
Pascual-Leone, A., & Walsh, V. (2001). Fast backprojections from the motion to the primary visual area necessary for visual awareness. Science, 292, 510–512.
Pavani, F., Làdavas, E., & Driver, J. (2003). Auditory and multisensory aspects of visuospatial neglect. Trends in Cognitive Sciences, 7, 407–414.
Perkel, D. J., Bullier, J., & Kennedy, H. (1986). Topography of the afferent connectivity of area 17 in the macaque monkey: A doublelabeling study. Journal of Comparative Neurology, 253, 374–402.
Petroni, F., Panzeri, S., Hilgetag, C. C., Kotter, R., & Young, M. P. (2001). Simultaneity of responses in a hierarchical visual network. NeuroReport, 12, 2753–2759.
Pons, T. (1996). Novel sensations in the congenitally blind. Nature, 380, 479–480.
Raiguel, S. E., Lagae, L., Gulyas, B., & Orban, G. A. (1989). Response latencies of visual cells in macaque areas V1, V2 and V5. Brain Research, 493, 155–159.
Rauschecker, J. P. (1995). Compensatory plasticity and sensory substitution in the cerebral cortex. Trends in Neurosciences, 18, 36–43.
Rauschecker, J. P., & Kniepert, U. (1994). Auditory localization be haviour in visually deprived cats. European Journal of Neuroscience, 6, 149–160.
Rauschecker, J. P., & Korte, M. (1993). Auditory compensation for early blindness in cat cerebral cortex. Journal of Neuroscience, 13, 4538–4548.
Recanzone, G. H. (2000). Spatial processing in the auditory cortex of the macaque monkey. Proceedings of the National Academy of Sciences, 97, 11829–11835.
Recanzone, G. H., Guard, D. C., & Phan, M. L. (2000). Frequency and intensity response properties of single neurons in the auditory cortex of the behaving macaque monkey. Journal of Neurophysiology, 83, 2315–2331.
Rockland, K. S. (1994). The organization of feedback connections from area V2 (18) to V1 (17). In A. Peters & K. S. Rockland (Eds.), Primary visual cortex in primates (Vol. 10, pp. 261–299). New York: Plenum.
Rockland, K. S., & Ojima, H. (2003). Multisensory convergence in calcarine visual areas in macaque monkey. International Journal of Psychophysiology, 50, 19–26.
Rockland, K. S., & Van Hoesen, G. W. (1994). Direct temporaloccipital feedback connections to striate cortex (V1) in the macaque monkey. Cerebral Cortex, 4, 300–313.
Roder, B., Teder-Salejarvi, W., Sterr, A., Rosler, F., Hillyard, S. A., & Neville, H. J. (1999). Improved auditory spatial tuning in blind humans. Nature, 400, 162–166.
Sadato, N., Pascual-Leone, A., Grafman, J., Ibanez, V., Deiber, M. P., Dold, G., & Hallett, M. (1996). Activation of the primary visual cortex by Braille reading in blind subjects. Nature, 380, 526–528.
Salin, P. A., Girard, P., Kennedy, H., & Bullier, J. (1992). Visuotopic organization of corticocortical connections in the visual system of the cat. Journal of Comparative Neurology, 320, 415–434.
Sanderson, K. J., Dreher, B., & Gayer, N. (1991). Prosencephalic connections of striate and extrastriate areas of rat visual cortex. Experimental Brain Research, 85, 324–334.
Schmolesky, M. T., Wang, Y., Hanes, D. P., Thompson, K. G., Leutgeb, S., Schall, J. D., & Leventhal, A. G. (1998). Signal timing across the macaque visual system. Journal of Neurophysiology, 79, 3272–3278.
Schroeder, C. E., & Foxe, J. J. (2002). The timing and laminar profile of converging inputs to multisensory areas of the macaque neocortex. Cognitive Brain Research, 14, 187–198.
Schroeder, C. E., Smiley, J., Fu, K. G., McGinnis, T., O’Connell, M. N., & Hackett, T. A. (2003). Anatomical mechanisms and functional implications of multisensory convergence in early cortical processing. International Journal of Psychophysiology, 50, 5–17.
Somers, D. C., Dale, A. M., Seiffert, A. E., & Tootell, R. B. (1999). Functional MRI reveals spatially specific attentional modulation in human primary visual cortex. Proceedings of the National Academy of Sciences, 96, 1663–1668.
Stein, B. E. (1998). Neural mechanisms for synthesizing sensory information and producing adaptive behaviors. Experimental Brain Research, 123, 124–135.
Stein, B. E., & Meredith, M. A. (1993). The merging of the senses. Cambridge, MA: MIT Press.
Suzuki, W. A., & Amaral, D. G. (2003). Perirhinal and parahippocampal cortices of the macaque monkey: Cytoarchitectonic and chemoarchitectonic organization. Journal of Comparative Neurology, 463, 67–91.
Symonds, L. L., & Rosenquist, A. C. (1984). Laminar origins of visual corticocortical connections in the cat. Journal of Comparative Neurology, 229, 39–47.
Thompson, W. L., & Kosslyn, S. M. (2000). Neural systems activated during visual memory imagery: A review and meta-analyses. In A.W. Toga & J. C. Mazziotta (Eds.), Brain mapping: II. The systems (pp. 535–560). San Diego: Academic Press.
Tian, B., Reser, D., Durham, A., Kustov, A., & Rauschecker, J. P. (2001). Functional specialization in rhesus monkey auditory cortex. Science, 292, 290–293.
Tolias, A. S., Smirnakis, S. M., Augath, M. A., Trinath, T., & Logothetis, N. K. (2001). Motion processing in the macaque: Revisited with functional magnetic resonance imaging. Journal of Neuroscience, 21, 8594–8601.
Vanduffel, W., Payne, B. R., Lomber, S. G., & Orban, G. A. (1997). Functional impact of cerebral connections. Proceedings of the National Academy of Sciences, 94, 7617–7620.
Vanduffel, W., Tootell, R. B., & Orban, G. A. (2000). Attentiondependent suppression of metabolic activity in the early stages of the macaque visual system. Cerebral Cortex, 10, 109–126.
Vezoli, J., Falchier, A., Jouve, B., Knoblauch, K., Young, M. A., & Kennedy, H. (in press). Quantitative analysis of connectivity in the visual cortex: Extracting structure from function. The Neuroscientist.
Wallace, M. T., & Stein, B. E. (1994). Cross-modal synthesis in the midbrain depends on input from cortex. Journal in Neurophysiology, 71, 429–432.
Wanet-Defalque, M. C., Veraart, C., De Volder, A., Metz, R., Michel, C., Dooms, G., & Goffinet, A. (1988). High metabolic activity in the visual cortex of early blind human subjects. Brain Research, 446, 369–373.
Watson, R. T., Valenstein, E., Day, A., & Heilman, K. M. (1994). Posterior neocortical systems subserving awareness and neglect: Neglect associated with superior temporal sulcus but not area 7 lesions. Archives of Neurology, 51, 1014–1021.
Weeks, R., Horwitz, B., Aziz-Sultan, A., Tian, B., Wessinger, C. M., Cohen, L. G., et al. (2000). A positron emission tomographic study of auditory localization in the congenitally blind. Journal of Neuroscience, 20, 2664–2672.
Yaka, R., Yinon, U., & Wollberg, Z. (1999). Auditory activation of cortical visual areas in cats after early visual deprivation. European Journal of Neuroscience, 11, 1301–1312.
Zeki, S. (1978). Functional specialisation in the visual cortex of the rhesus monkey. Nature, 274, 423–428.
Zeki, S. (1993). A vision of the brain. Oxford: Blackwell.
Zipser, K., Lamme, V. A., & Schiller, P. H. (1996). Contextual modulation in primary visual cortex. Journal of Neuroscience, 16, 7376–7389.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by EU Grant“Nets and Representations” QLG3-1999-0106, Human Frontier Science Program Grant RG0133/ 2000-B, and European Community FP5 Quality of Life Grant QLG3-1999-01064, A.F. was supported by the Fondation pour la Recherche Médicale.
Rights and permissions
About this article
Cite this article
Clavagnier, S., Falchier, A. & Kennedy, H. Long-distance feedback projections to area V1: Implications for multisensory integration, spatial awareness, and visual consciousness. Cognitive, Affective, & Behavioral Neuroscience 4, 117–126 (2004). https://doi.org/10.3758/CABN.4.2.117
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.3758/CABN.4.2.117