Abstract
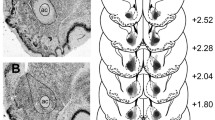
The nucleus accumbens (NAcc) is implicated in reward-related processes and in reinforcement learning. However, its precise role in associative processes is unclear and may not be related solely to appetitive learning. In the present study, the differential effects of selective excitotoxic lesions of the NAcc core and shell were studied on the acquisition of an aversive Pavlovian conditioning task that assessed conditioning to both discrete and contextual cues. Rats with selective lesions of the NAcc shell were not impaired on measures of aversive Pavlovian conditioning to either discrete or contextual cues. In contrast, animals with lesions of the NAcc core showed an impairment in conditioning to discrete cues and an enhancement in conditioning to contextual cues. The NAcc is thus implicated in aspects of aversive Pavlovian conditioning; the significance of this finding for theories of cortico-striatal function is discussed.
Article PDF
Similar content being viewed by others
References
Alexander, G. E., Delong, M. R., & Strick, P. L. (1986). Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annual Review of Neuroscience, 9, 357–381.
Annett, L. E., McGregor, A., & Robbins, T. W. (1989). The effects of ibotenic acid lesions of the nucleus accumbens on spatial-learning and extinction in the rat. Behavioural Brain Research, 31, 231–242.
Balleine, B., & Killcross, S. (1994). Effects of ibotenic acid lesions of the nucleus accumbens on instrumental action. Behavioural Brain Research, 65, 181–193.
Beck, C. H. M., & Fibiger, H. C. (1995). Conditioned fear-induced changes in behavior and in the expression of the immediate-early gene c-fos—With and without diazepam pretreatment. Journal of Neuroscience, 15 (1, Pt. 2), 709–720.
Blaha, C. D., Floresco, S. B., Phillips, A. G., & Yang, C. R. (1997). Gating of hippocampal and amygdala inputs to the nucleus accumbens by dopamine: A combined in vivo extracellular electrophysiological and electrochemical recording study. Society for Neuroscience Abstracts, 23, 505.6.
Brog, J. S., Salyapongse, A., Deutch, A. Y., & Zahm, D. S. (1993). The patterns of afferent innervation of the core and shell in the accumbens part of the rat ventral striatum—Immunohistochemical detection of retrogradely transported fluorogold. Journal of Comparative Neurology, 338, 255–278.
Buchanan, S. L., & Powell, D. A. (1982). Cingulate damage attenuates conditioned bradycardia. Neuroscience Letters, 29, 261–268.
Burns, L. H., Annett, L., Kelley, A. E., Everitt, B. J., & Robbins, T. W. (1996). Effects of lesions to amygdala, ventral subiculum, medial prefrontal cortex, and nucleus-accumbens on the reaction to novelty—Implication for limbic–striatal interactions. Behavioral Neuroscience, 110, 60–73.
Cador, M., Robbins, T. W., & Everitt, B. J. (1989). Involvement of the amygdala in stimulus reward associations—Interaction with the ventral striatum. Neuroscience, 30, 77–86.
Campeau, S., Falls, W. A., Cullinan, W. E., Helmreich, D. L., Davis, M., & Watson, S. J. (1997). Elicitation and reduction of fear: Behavioural and neuroendocrine indices and brain induction of the immediate-early gene c-fos. Neuroscience, 78, 1087–1104.
Deutch, A. Y., & Cameron, D. S. (1992). Pharmacological characterization of dopamine systems in the nucleus-accumbens core and shell. Neuroscience, 46, 49–56.
Devinsky, O., Morrell, M. J., & Vogt, B. A. (1995). Contributions of anterior cingulate cortex to behavior. Brain, 118 (Pt. 1), 279–306.
Di Chiara, G. (1998). A motivational learning hypothesis of the role of mesolimbic dopamine in compulsive drug use. Journal of Psychopharmacology, 12, 54–67.
Dickinson, A. (1980). Contemporary animal learning theory. Cambridge: Cambridge University Press.
Dweck, C. S., & Wagner, A. R. (1970). Situational cues and correlation between CS and US as determinants of the conditioned emotional response. Psychonomic Science, 18, 145–147.
Everitt, B. J., Morris, K. A., O’Brien, A., & Robbins, T. W. (1991). The basolateral amygdala ventral striatal system and conditioned place preference—Further evidence of limbic striatal interactions underlying reward-related processes. Neuroscience, 42, 1–18.
Feenstra, M. G. P., Botterblom, M. H. A., & Vanuum, J. F. M. (1995). Novelty-induced increase in dopamine release in the rat prefrontal cortex in-vivo—Inhibition by diazepam. Neuroscience Letters, 189, 81–84.
Good, M., & Honey, R. C. (1991). Conditioning and contextual retrieval in hippocampal rats. Behavioral Neuroscience, 105, 499–509.
Grace, A. A., & Moore, H. (1998). Regulation of information flow in the nucleus accumbens: A model for the pathophysiology of schizophrenia. In M. F. Lenzenweger & R. H. Dworkin (Eds.), The pathogenesis of schizophrenia: An experimental psychopathology perspective (pp. 123–157). Washington, DC: American Psychological Association.
Groenewegen, H. J., Vermeulen-Van der Zee, E., Kortschot, A. T., & Witter, M. P. (1987). Organization of the projections from the subiculum to the ventral striatum in the rat—A study using anterograde transport of phaseolus-vulgaris leucoagglutinin. Neuroscience, 23, 103–120.
Holland, P. C. (1997). Brain mechanisms for changes in processing of conditioned stimuli in Pavlovian conditioning: Implications for behavior theory. Animal Learning & Behavior, 25, 373–399.
Imperato, A., Angelucci, L., Casolini, P., Zocchi, A., & Puglisiallegra, S. (1992). Repeated stressful experiences differently affect limbic dopamine release during and following stress. Brain Research, 577, 194–199.
Jongen-Relo, A. L., Groenewegen, H. J., & Voorn, P. (1993). Evidence for a multicompartmental histochemical organization of the nucleus-accumbens in the rat. Journal of Comparative Neurology, 337, 267–276.
Kalivas, P. W., & Duffy, P. (1995). Selective activation of dopamine transmission in the shell of the nucleus-accumbens by stress. Brain Research, 675, 325–328.
Kalivas, P. W., & Duffy, P. (1997). Dopamine regulation of extracellular glutamate in the nucleus accumbens. Brain Research, 761, 173–177.
Killcross, A. S., Robbins, T. W., & Everitt, B. J. (1997a). Different types of fear-conditioned behaviour mediated by separate nuclei within amygdala. Nature, 388, 377–380.
Killcross, A. S., Robbins, T. W., & Everitt, B. J. (1997b). Is it time to invoke multiple fear learning systems in the amygdala? Trends in Cognitive Sciences, 1, 244–246.
Kim, J. J., & Fanselow, M. S. (1992). Modality-specific retrograde-amnesia of fear. Science, 256, 675–677.
King, D., & Finlay, J. M. (1997). Loss of dopamine terminals in the medial prefrontal cortex increased the ratio of DOPAC to DA in tissue of the nucleus accumbens shell: Role of stress. Brain Research, 767, 192–200.
LeDoux, J. E., Cicchetti, P., Xagoraris, A., & Romanski, L. M. (1990). The lateral amygdaloid nucleus–sensory interface of the amygdala in fear conditioning. Journal of Neuroscience, 10, 1062–1069.
Mackintosh, N. J. (1975). A theory of attention: Variation in the associability of stimuli with reinforcement. Psychological Review, 82, 276–298.
Maldonado-Irizarry, C. S., & Kelley, A. E. (1995). Excitatory amino-acid receptors within nucleus-accumbens subregions differentially mediate spatial-learning in the rat. Behavioural Pharmacology, 6, 527–539.
Maren, S., & Fanselow, M. S. (1996). The amygdala and fear conditioning: Has the nut been cracked? Neuron, 16, 237–240.
Mark, G. P., Blander, D. S., & Hoebel, B. G. (1991). A conditioned-stimulus decreases extracellular dopamine in the nucleus-accumbens after the development of a learned taste-aversion. Brain Research, 551, 308–310.
Marlin, N. A. (1981). Contextual associations in trace conditioning. Animal Learning & Behavior, 9, 519–523.
McCullough, L. D., Sokolowski, J. D., & Salamone, J. D. (1993). A neurochemical and behavioral investigation of the involvement of nucleus-accumbens dopamine in instrumental avoidance. Neuroscience, 52, 919–925.
Mirenowicz, J., & Schultz, W. (1996). Preferential activation of midbrain dopamine neurons by appetitive rather than aversive stimuli. Nature, 379, 449–451.
Moser, E., Moser, M. B., & Andersen, P. (1993). Spatial-learning impairment parallels the magnitude of dorsal hippocampal-lesions, but is hardly present following ventral lesions. Journal of Neuroscience, 13, 3916–3925.
Mulder, A. B., Hodenpijl, M. G., & Lopes da Silva, F. H. (1998). Electrophysiology of the hippocampal and amygdaloid projections to the nucleus accumbens of the rat: Convergence, segregation, and interaction of inputs. Journal of Neuroscience, 18, 5095–5102.
Nader, K., & LeDoux, J. E. (1997). Is it time to invoke multiple fear learning systems in the amygdala. Trends in Cognitive Sciences, 1, 241–244.
Odling-Smee, F. J. (1975a). Background stimuli and the inter-stimulus interval during Pavlovian conditioning. Quarterly Journal of Experimental Psychology, 27, 387–392.
Odling-Smee, F. J. (1975b). The role of background stimuli during Pavlovian conditioning. Quarterly Journal oj’Experimental Psychology, 27, 201–209.
Odling-Smee, F. J. (1978). The overshadowing of background stimuli by an informative CS in aversive Pavlovian conditioning with rats. Animal Learning & Behavior, 6, 43–51.
Parkinson, J. A., Olmstead, M. C., Robbins, T. W., & Everitt, B. J. (1999). Dissociation in effects of lesions of the nucleus accumbens core and shell in appetitive Pavlovian approach behavior and the potentiation of conditioned reinforcement and locomotor activity by d-amphetamine. Journal of Neuroscience, 19, 2401–2411.
Paxinos, G., & Watson, C. (1998). The rat brain in stereotaxic coordinates. San Diego: Academic Press.
Pearce, J. M., & Hall, G. (1980). A model for Pavlovian learning: Variations in the effectiveness of conditioned but not of unconditioned stimuli. Psychological Review, 87, 532–552.
Pennartz, C. M. A., Dasilva, F. H. L., & Groenewegen, H. J. (1994). The nucleus-accumbens as a complex of functionally distinct neuronal ensembles—an integration of behavioral, electrophysiological and anatomical data. Progress in Neurobiology, 42, 719.
Phillips, R. G., & LeDoux, J. E. (1992). Differential contribution of amygdala and hippocampus to cued and contextual fear conditioning. Behavioral Neuroscience, 106, 274–285.
Powell, D. A., Watson, K., & Maxwell, B. (1994). Involvement of subdivisions of the medial prefrontal cortex in learned cardiac adjustments in rabbits. Behavioral Neuroscience, 108, 294–307.
Reading, P. J., Dunnett, S. B., & Robbins, T. W. (1991). Dissociable roles of the ventral, medial and lateral striatum on the acquisition and performance of a complex visual stimulus–response habit. Behavioural Brain Research, 45, 147–161.
Rescorla, R. A. (1968). Probability of shock in the presence and absence of CS in fear conditioning. Journal of Comparative & Physiological Psychology, 66, 1–5.
Rescorla, R. A., & Wagner, A. R. (1972). A theory of Pavlovian conditioning: Variations in the effectiveness of reinforcement and non-reinforcement. In A. H. Black & W. F. Prokasy (Eds.), Classical conditioning II: Current research and theory (pp. 64–99). New York: Appleton-Century-Crofts.
Riedel, G., Harrington, N. R., Hall, G., & MacPhail, E. M. (1997). Nucleus accumbens lesions impair context, but not cue, conditioning in rats. NeuroReport, 8, 2477–2481.
Robbins, T. W., & Everitt, B. J. (1995). Arousal systems and attention. In M. Gazzaniga (Ed.), The cognitive neurosciences (pp. 703–725). Cambridge, MA: MIT Press.
Salamone, J. D. (1994). The involvement of nucleus-accumbens dopamine in appetitive and aversive motivation. Behavioural Brain Research, 61, 117–133.
Sananes, C. B., & Davis, M. (1992). N-methyl-d-aspartate lesions of the lateral and basolateral nuclei of the amygdala block fear-potentiated startle and shock sensitization of startle. Behavioral Neuroscience, 106, 72–80.
Schwarting, R., & Carey, R. J. (1985). Deficits in inhibitory avoidance after neurotoxic lesions of the ventral striatum are neurochemically and behaviorally selective. Behavioural Brain Research, 18, 279–283.
Selden, N. R. W., Everitt, B. J., Jarrard, L. E., & Robbins, T. W. (1991). Complementary roles for the amygdala and hippocampus in aversive conditioning to explicit and contextual cues. Neuroscience, 42, 335–350.
Selden, N. R. W., Robbins, T. W., & Everitt, B. J. (1990). Enhanced behavioral conditioning to context and impaired behavioral and neuroendocrine responses to conditioned-stimuli following ceruleocortical noradrenergic lesions—support for an attentional hypothesis of central noradrenergic function. Journal of Neuroscience, 10, 531–539.
Thierry, A. M., Tassin, J. P., Blanc, G., & Glowinski, J. (1976). Selective activation of mesocortical dopaminergic system by stress. Nature, 263, 242–244.
Westbrook, R. F., Good, A. J., & Kiernan, M. J. (1997). Microinjection of morphine into the nucleus accumbens impairs contextual learning in rats. Behavioral Neuroscience, 111, 996–1013.
Wilkinson, L. S., Humby, T., Killcross, A. S., Torres, E. M., Everitt, B. J., & Robbins, T. W. (1998). Dissociations in dopamine release in medial prefrontal cortex and ventral striatum during the acquisition and extinction of classical aversive conditioning in the rat. European Journal of Neuroscience, 10, 1019–1026.
Williams, G. V., Rolls, E. T., Leonard, C. M., & Stern, C. (1993). Neuronal responses in the ventral striatum of the behaving macaque. Behavioural Brain Research, 55, 243–252.
Wise, R. A., & Bozarth, M. A. (1987). A psychomotor stimulant theory of addiction. Psychological Review, 94, 469–492.
Wise, R. A., & Rompre, P. P. (1989). Brain dopamine and reward. Annual Review of Psychology, 40, 191–225.
Wright, C. I., Beijer, A. V. J., & Groenewegen, H. J. (1996). Basal amygdaloid complex afferents to the rat nucleus-accumbens are compartmentally organized. Journal of Neuroscience, 16, 1877–1893.
Young, A. M. J., Ahier, R. G., Upton, R. L., Joseph, M. H., & Gray, J. A. (1998). Increased extracellular dopamine in the nucleus accumbens of the rat during associative learning of neutral stimuli. Neuroscience, 83, 1175–1183.
Young, A. M. J., Joseph, M. H., & Gray, J. A. (1993). Latent inhibition of conditioned dopamine release in rat nucleus-accumbens. Neuroscience, 54, 5–9.
Zahm, D. S., & Brog, J. S. (1992). On the significance of subterritories in the accumbens part of the rat ventral striatum. Neuroscience, 50, 751–767.
Zahm, D. S., & Heimer, L. (1993). Specificity in the efferent projections of the nucleus-accumbens in the rat—comparison of the rostral pole projection patterns with those of the core and shell. Journal of Comparative Neurology, 327, 220–232.
Author information
Authors and Affiliations
Corresponding author
Additional information
J.A.P. was in receipt of a BBSRC research fellowship and the Oon Khye Beng Ch’hia Tsio Scholarship, awarded by Downing College, Cambridge.
This work was supported by a Wellcome Trust Programme Grant (T.W.R., B.J.E., A. Roberts, and B. J. Sahakian).
The authors thank Caroline Morrison and Helen Sweet for their assistance with the histological assessment for this study.
Rights and permissions
About this article
Cite this article
Parkinson, J.A., Robbins, T.W. & Everitt, B.J. Selective excitotoxic lesions of the nucleus accumbens core and shell differentially affect aversive Pavlovian conditioning to discrete and contextual cues. Psychobiology 27, 256–266 (1999). https://doi.org/10.3758/BF03332119
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3758/BF03332119