Abstract
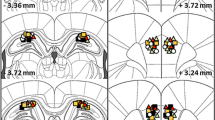
This study examined the effects of ibotenate lesions of the hippocampal CA1 subfield on spatial working memory (delayed-nonmatching-to-place [DNMTP] task) as a function of the existence of preoperative training in mice. The task was varied in its level of difficulty according to either the number of interpolated arm visits (occupied delays) or the length of free time delays imposed between place sample presentation and subsequent recognition. Results indicate that when the tasks were first well learned and the CA1 subfield was subsequently lesioned, only slight impairments were observed during both reacquisition of the DNMTP rule and the problem containing five interpolated visits. When CA1 pyramidal cells were lesioned prior to any training, CA1-lesioned subjects showed general performance deficits regardless of the delay studied (occupied or free). A shift from isolated rule task to the same task tested simultaneously with more difficult variants produced marked performance deficits even for the previously mastered DNMTP rule task. These results suggest that preoperative training reduces subsequent lesion-induced memory deficits and modifies the pattern of postoperative working-memory performance; lesions of the hippocampal CA1 field contribute to the impairments of not only mnemonic capability per se but also reference memory components of the DNMTP task underlying the procedural and cognitive demands required for correct performance of the task.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Auer, R. N., Jensen, M. L., & Whishaw, I. Q. (1989). Neurobehavioral deficits due to ischemic brain damage limited to half of the CA1 sector of the hippocampus. Journal of Neuroscience, 9, 1641–1647.
Barnes, C. A. (1988). Spatial learning and memory processes: The search for their neurobiological mechanisms in the rat. Trends in Neurosciences, 11, 163–169.
Cho, Y. H., Beracochea, D., & Jaffard, R. (1991). Temporally graded retrograde and anterograde amnesia following ibotenic entorhinal cortex lesion in mice. Society for Neuroscience Abstracts, 17, 1045.
Cirillo, R. A., Horel, J. A., & George, P. J. (1989). Lesions of the anterior temporal stem and the performance of delayed match-to-sample and visual discriminations in monkeys. Behavioural Brain Research, 34, 55–69.
Cumming, J. L., Tomiyasu, U., Read, S., & Benson, D. F. (1984). Amnesia with hippocampal lesions after cardiopulmonary arrest. Neurology, 34, 679–681.
Davis, H. P., Baranowski, J. R., Pulsinelli, W. A., & Volpe, B. T. (1987). Retention of reference memory following ischemic hippocampal damage. Physiology & Behavior, 39, 783–786.
Eichenbaum, H., Mathews, P., & Cohen, N. J. (1989). Further studies of hippocampal representation during odor discrimination learning. Behavioral Neuroscience, 103, 1207–1216.
Eichenbaum, H., Stewart, C., & Morris, R. G. M. (1990). Hippocampal representation in place learning. Journal of Neuroscience, 10, 3531–3542.
Gaffan, D. (1974). Recognition impaired and association intact in the memory of monkeys after transection of the fornix. Journal of Comparative & Physiological Psychology, 86, 1100–1109.
Gaffan, D. (1985). Hippocampus: Memory, habit and voluntary movement. Philosophical Transactions of the Royal Society of London, B308, 87–99.
Gaffan, D., Gaffan, E. A., & Harrison, S. (1984). Effects of fornix transection upon spontaneous and trained non-matching by monkeys. Quarterly Journal of Experimental Psychology: Comparative & Physiological Psychology, 36B, 285–303.
Gaffan, D., Sheilds, C., & Harrison, S. (1984). Delayed matching by fornix-transected monkeys: The sample, the push and the bait. Quarterly Journal of Experimental Psychology: Comparative & Physiological Psychology, 36B, 305–317.
Gage, P. D. (1985). Performance of hippocampectomized rats in a reference/working-memory task: Effects of preoperative versus postoperative training. Physiological Psychology, 13, 235–242.
Gionet, T. X., Thomas, J. D., Warner, D. S., Goodlett, C. R., Wasserman, E. A., & West, J. R. (1991). Forebrain ischemia induces selective behavioral impairments associated with hippocampal injury in rats. Stroke, 22, 1040–1047.
Goldschmidt, R. B., & Steward, O. (1980). Preferential neurotoxicity of colchicine for granule cells of the dentate gyrus of the adult rat. Proceedings of the National Academy of Sciences, 77, 3047–3051.
Hagan, J. J., & Beaughard, M. (1990). The effects of forebrain ischaemia on spatial learning. Behavioural Brain Research, 41, 151–160.
Jaffard, R., Beracochea, D., & Cho, Y. (1991). The hippocampalmamillary system: Anterograde and retrograde amnesia. Hippocampus, 1, 275–278.
Jarrard, L. E. (1978). Selective hippocampal lesions: Differential effects on performance by rats of a spatial task with preoperative versus postoperative training. Journal of Comparative & Physiological Psychology, 92, 119–127.
Jarrard, L. E. (1986). Selective hippocampal lesions and behavior: Implications for current research and theorizing. In R. Isaacson & K. H. Pribram (Eds.), The hippocampus (Vol. 4, pp. 93–126). New York: Plenum.
Jarrard, L. E. (1989). On the use of ibotenic acid to lesion selectively different components of the hippocampal formation. Journal of Neuroscience Methods, 29, 251–259.
Jarrard, L. E. (1991). On the neural bases of the spatial mapping system: Hippocampus vs. hippocampal formation. Hippocampus, 1, 236–239.
Jarrard, L. E., Okaiche, H., Steward, O., & Goldschmidt, R. B. (1984). On the role of hippocampal connections in the performance of place and cue tasks: Comparisons with damage to hippocampus. Behavioral Neuroscience, 95, 946–954.
Kesner, R. P. (1986). Neurobiological views of memory. In J. L. Martinez & R. P. Kesner (Eds.), Learning and memory: A biological view (pp. 399–438). Orlando: Academic Press.
Kiyota, Y., Miyamoto, M., & Nagaoka, A. (1991). Relationship between brain damage and memory impairment in rats exposed to transient forebrain ischemia. Brain Research, 538, 295–302.
McLamb, R. L., Mundy, W. R., & Tilson, H. A. (1988). Destruction of dentate gyrus cells with colchicine interferes with the acquisition and retention of a working memory task in rats. Neurotoxicology & Teratology, 9, 521–528.
McNaughton, B. L., Barnes, C. A., Meltzer, J., & Sutherland, R. J. (1989). Hippocampal granule cells are necessary for normal spatial learning but not for spatially-selective pyramidal cell discharge. Experimental Brain Research, 76, 485–496.
Mishkin, M. (1978). Memory in monkeys severely impaired by combined but not by separate removals of amygdala and hippocampus. Nature, 273, 297–298.
Morris, R. G. M. (1983). An attempt to dissociate “spatial-mapping” and “working-memory” theories of hippocampal function. In W. Seifert (Ed.), Neurobiology of the hippocampus (pp. 405–432). New York: Academic Press.
Murray, E. A., & Mishkin, M. (1984). Severe tactual as well as visual memory deficits following combined removal of the amygdala and hippocampus in monkeys. Journal of Neuroscience, 4, 2565–2580.
Oakley, D. A. (1983). The varieties of memory: A phylogenetic approach. In A. Mayes (Ed.), Memory in animals and humans (pp. 20–82). New York: Van Nostrand Reinhold.
O’Keefe, J., & Nadel, L. (1978). The hippocampus as a cognitive map. Oxford: Clarendon Press.
Olton, D. S., Becker, J. T., & Handelmann, G. E. (1979). Hippocampus, space and memory. Behavioral & Brain Science, 2, 487–533.
Pulsinelli, W., Brierley, J., & Plum, F. (1982). Temporal profile of neuronal damage in model of transient forebrain ischemia. Annals of Neurology, 11, 491–498.
Ridley, R. M., & Baker, H. F. (1991). A critical evaluation of monkey models of amnesia and dementia. Brain Research Reviews, 16, 15–37.
Saunders, R. C., & Weiskrantz, L. (1989). The effects of fornix transection and combined fornix transection, mammillary body lesions and hippocampal ablations or object-pair association memory in the rhesus monkey. Behavioural Brain Research, 35, 85–94.
Sidman, M., Stoddard, L. T., & Mohr, J. P. (1968). Some additional quantitative observations of immediate memory in a patient with bilateral hippocampal lesions. Neuropsychologia, 6, 245–254.
Squire, L. R., Zola-Morgan, S., & Chen, K. S. (1988). Human amnesia and animal models of amnesia: Performance of amnesic patients on tests designed for the monkey. Behavioral Neuroscience, 102, 210–221.
Sutherland, R. J., Whishaw, I. Q., & Kolb, B. (1983). A behavioural analysis of spatial localisation following electrolytic kainate or colchicine induced damage to the hippocampal formation in the rat. Behavioural Brain Research, 7, 133–153.
Tulving, E. (1983). Elements of episodic memory. Oxford: Clarendon.
Volpe, B. T., Davis, H. P., & Colombo, P. J. (1989). Preoperative training modifies radial maze performance in rats with ischemic hippocampal injury. Stroke, 20, 1700–1706.
Volpe, B. T., & Hirst, W. (1983). The characterization of an amnesic syndrome following hypoxic ischemic injury. Archives of Neurology, 40, 436–440.
Volpe, B. T., Waczek, B., & Davis, H. P. (1988). Modified T-maze training demonstrated dissociated memory loss in rats with hippocampal injury. Behavioural Brain Research, 27, 259–268.
Walsh, T. J., Schulz, D. W., Tilson, H. A., & Shmechel, D. E. (1986). Colchicine-induced granule cell loss in rat hippocampus: Selective behavioral and histological alterations. Brain Research, 398, 23–36.
Whishaw, I. Q. (1987). Hippocampal granule cell and CA3-4 lesions impair formation of a place learning-set in the rat and induce reflex epilepsy. Behavioural Brain Research, 24, 59–72.
Zola-Morgan, S., & Squire, L. R. (1986). Memory impairment in monkeys following lesions limited to the hippocampus. Behavioral Neuroscience, 100, 155–160.
Zola-Morgan, S., & Squire, L. R. (1990). The primate hippocampal formation: Evidence for a time-limited role in memory storage. Science, 250, 288–290.
Zola-Morgan, S., Squire, L. R., & Amaral, D. G. (1986). Human amnesia and the medial temporal region: Enduring memory impairment following a bilateral lesion limited to the CA1 field of hippocampus. Journal of Neuroscience, 6, 2950–2967.
Zola-Morgan, S., Squire, L. R., Amaral, D. G., & Suzuki, W. A. (1989). Lesions of perirhinal and parahippocampal cortex that spare the amygdala and hippocampal formation produce severe memory impairment. Journal of Neuroscience, 9, 4355–4370.
Zola-Morgan, S., Squire, L. R., & Mishkin, M. (1982). The neuroanatomy of amnesia: Amygdala-hippocampus versus temporal stem. Science, 218, 1337–1339.
Author information
Authors and Affiliations
Additional information
This study was supported by the Centre National de la Recherche Scientifique (CNRS) and the Fondation pour la Recherche Médicale.
The authors wish to thank L. E. Jarrard for his evaluation of ibotenate CA1 field lesions, and T. Durkin for help in preparing this paper.
Rights and permissions
About this article
Cite this article
Cho, Y.H., Beracochea, D. & Jaffard, R. Differential effects of ibotenate lesions of the CA1 subfield of the hippocampus on a delayed-nonmatching-to-place task as a function of preoperative training in mice. Psychobiology 20, 261–269 (1992). https://doi.org/10.3758/BF03332058
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3758/BF03332058