Abstract
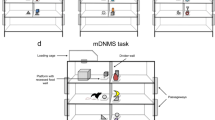
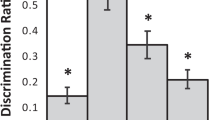
Assessment of recognition memory was performed in a group of 12 rhesus monkeys, 19–24 years old (an age estimated to be comparable with early senescence in humans—i.e., 60s to early 70s). Their performance was compared with that of 14 young adult animals (5–14 years old, comparable to humans from the late teens to early 40s) on two tasks of recognition memory: trial-unique delayed nonmatching to sample (DNMS) and the delayed recognition span task (DRST). The DNMS is a benchmark visual recognition memory task that requires the identification of a novel object when paired with a familiar object that was presented 10 sec earlier. After reaching learning criterion, the memory demands of the task are increased by lengthening the interval between stimulus presentation and recognition up to 600 sec and lengthening the list length of samples presented before recognition up to 10 objects. The DRST is a task used to assess memory capacity by determining memory span for number of stimuli in different stimulus conditions. In the present study, memory span was determined for spatial, color, and object stimuli using both a unique series of stimuli and an embedded repeated series. On DNMS, only 4 of 12 early senescent monkeys were impaired, compared with the range of young adult monkeys in the acquisition of the task, but as a group, they were significantly impaired on the delay and list performance measures. On the DRST, early senescent monkeys, as a group, were impaired on both the spatial condition and the object condition of the task, but not on the color condition. The results reveal a cognitive profile of early senescent monkeys that, unlike monkeys of more advanced age, is characterized by a relatively moderate impairment in recognition memory and memory span.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aggleton, J. P., & Mishkin, M. (1983). Visual recognition impairment following medial thalamic lesions in monkeys. Neuropsychologia, 21, 189–197.
Albert, M. S., & Moss, M. B. (1984). The assessment of memory disorders in patients with Alzheimer’s disease. In L. Squire & N. Butters (Eds.), Neuropsychology of memory (pp. 236–246). New York: Guilford.
Albert, M. S., & Moss, M. B. (1999). Profiles of cognition in normal aging. In A. Peters & J. Morrison (Eds.), Cerebal cortex (Vol. 13, pp. 1–20). New York: Plenum.
Alverez, P., Zola-Morgan, S., & Squire, L. R. (1995). Damage limited to the hippocampal region produces long-lasting memory impairment in monkeys. Journal of Neuroscience, 15, 3796–3807.
Amaral, D. G. (1993). Morphological analyses of the brains of behaviorally characterized aged nonhuman primates. Neurobiology of Aging, 14, 671–612.
Arnsten, A. F. T., & Goldman-Rakic, P. S. (1985). A-2 adrenergic mechanisms in prefrontal cortex associated with cognitive decline in aged nonhuman primates. Science, 230, 1273–1276.
Arnsten, A. F. T., & Goldman-Rakic, P. S. (1990). Analysis of a-2 adrenergic agonist effects on the delayed nonmatch-to-sample performance of aged rhesus monkeys. Neurobiology of Aging, 11, 583–590.
Bachevalier, J. (1993). Behavioral changes in aged rhesus monkeys. Neurobiology of Aging, 14, 619–621.
Bachevalier, J., Landis, L. C., Walker, M., Brickson, M., Mishkin, M., Price, D. L., & Cork, L. C. (1991). Aged monkeys exhibit behavioral deficits indicative of widespread cerebral dysfunction. Neurobiology of Aging, 12, 99–111.
Bachevalier, J., & Mishkin, M. (1986). Visual recognition impairment follows ventromedial but not dorsolateral prefrontal lesions in monkeys. Behavioral Brain Research, 20, 249–261.
Bachevalier, J., & Mishkin, M. (1989). Mnemonic and neuropathological effects of occluding the posterior cerebral artery in macaca mulatta. Neuropsychologia, 27, 83–105.
Bartus, R. T. (1979). Effects of aging on visual memory, sensory processing and discrimination learning in a nonhuman primate. In J. M. Ordy & K. Brizzee (Eds.), Sensory systems and communication in the elderly (pp. 137–145). New York: Raven.
Bartus, R. T., & Dean, R. L, III (1979). Recent memory in aged non-human primates: Hypersensitivity to visual interference during retention. Experimental Aging Research, 5, 385–400.
Bartus, R. T., Dean, R. L., III, & Fleming, D. L. (1979). Aging in the rhesus monkey: Effects on visual discrimination learning and reversal learning. Journal of Gerontology, 34, 209–219.
Bartus, J. M., Fleming, D., & Johnson, H. R. (1978). Aging in the rhesus monkey: Debilitating effects on short-term memory. Journal of Gerontology, 34, 209–219.
Beason-Held, L., Rosene, D. L., Killiany, R. J., & Moss, M. B. (1999). Memory deficits associated with ibotenic acid lesions of the hippocampal formation in rhesus monkeys. Hippocampus, 9, 562–574.
Blackford, R. C., & La Rue, A. (1989). Criteria for diagnosing age associated memory impairment: Proposed improvements from the field. Developmental Neuropsychology, 5, 295–306.
Comparet, P., Darriet, D., & Jaffard, R. (1992). Demonstration of dissociation between frontal and temporal lesions in man on two versions of delayed nonmatching recognition tests used in monkeys. CR Academy of Science, 314, 515–518.
Crook, T., Bartus, R. T., Ferris, S. H., Whitehouse, P., Cohen, G. D., & Gershon, S. (1986). Age associated memory impairment: Proposed diagnostic criteria and measures of clinical change—Report of a National Institute of Mental Health work group. Developmental Neuropsychology, 2, 261–276.
Davis, R. T., Bennet, C. L., & Weisenburger, R. P. (1982). Repeated measurements of forgetting by rhesus monkeys (Macaca mulatta). Perceptual Motor Skills, 55, 703–709.
Gaffan, D. (1974). Recognition impaired and association intact in the memory of monkeys after transection of the fornix. Journal of Comparative & Physiological Psychology, 86, 1100–1109.
Gellerman, L. W. (1933). Chance orders of alternating stimuli in visual discrimination experiments. Journal of General Psychology, 42, 207–208.
Gilbert, J. G., & Levee, R. F. (1971). Patterns of declining memory. Journal of Gerontology, 26, 70–75.
Goldman-Rakic, P. S. (1987). Circuitry of primate prefrontal cortex and regulation of behavior by representational memory. Handbook of Physiology, 5, 373–417.
Goldman-Rakic, P. S., & Brown, R. M. (1981). Regional changes in monoamines in cerebral cortex and subcortical structures of aging rhesus monkeys. Neuroscience, 6, 177–178.
Harlow, H., & Bromer, J. A. (1938). A test-apparatus for monkeys. Psychological Review, 19, 434–438.
Hayes, W. L. (1988). Statistics (4th ed.). New York: Holt, Rinehart & Winston.
Inouye, S. K., Albert, M. S., Mohs, R., Sun, K., & Berkman, L. F. (1993). Cognitive performance in a high-functioning community-dwelling elderly population. Journal of Gerontology, 48, 146–151.
Kemper, T. L. (1993). The relationship of cerebral cortical changes to nuclei in the brainstem. Neurobiology of Aging, 14, 659–660.
Kemper, T. L., Moss, M. B., Rosene, D. L., & Killiany, R. J. (1997). Age-related neuronal loss in the nucleus centralis superior of the rhesus monkey. Acta Neuropathologia, 94, 124–130.
Kirk, R. E. (1982). Experimental design: Procedures for the behavioral scientist (2nd ed.). Pacific Grove, CA: Brooks/Cole.
Lai, Z. C., Moss, M. B., Killiany, R. J., Rosene, D. L., & Herndon, J. G. (1995). Executive system dysfunction in the aged monkey: Spatial and object reversal learning. Neurobiology of Aging, 16, 947–954.
Lange, K. W., Robbins, T. W., Marsden, C. D., James, M., Owen, A. M., & Paul, G. M. (1992). L-dopa withdrawal in Parkinson’s disease selectively impairs cognitive performance in tests sensitive to frontal lobe function. Psychopharmacology, 107, 394–404.
La Rue, A. (1982). Memory loss and aging: Distinguishing dementia from benign senescent forgetfulness and depressive pseudodementia. Psychiatric Clinics of North America, 5, 89–103.
Mahut, H., Moss, M. B., & Zola-Morgan, S. (1982). Hippocampal resections impair associative learning and recognition memory in the monkey. Journal of Neuroscience, 2, 1214–1229.
Marcusson, J. O., Morgan, D. G., Winblad, B., & Finch, C. E. (1984). Serotonin-2 binding sites in human frontal cortex and hippocampus. Selective loss of S-2A sites with age. Brain Research, 311, 51–56.
Marriott, J. G., & Abelson, J. S. (1980). Age differences in short-term memory of test-sophisticated rhesus monkeys. Age, 3, 7–9.
Medin, D. L., & Davis, R. T. (1974). Memory. In A. M. Shrier & F. Stollmitz (Eds.), Behavior of non-human primates (pp. 1–47). New York: Academic Press.
Meunier, M., Hadfield, W., Bachevalier, J., & Murray, E. A. (1996). Effects of rhinal cortex lesions combined with hippocampectomy on visual recognition memory in rhesus monkeys. Journal of Neurophysiology, 75, 1190–1205.
Mishkin, M. (1978). Memory in monkeys severely impaired by combined but not by separate removal of amygdala and hippocampus. Nature, 273, 297–298.
Moore, T. L., Killiany, R. J., Rosene, D. L., & Moss, M. B. (1998). Executive system dysfunction in the aged rhesus monkey using an analog of the Wisconsin Card Sorting Test. Society for Neuroscience Abstracts, 28, 1764.
Moss, M. B. (1983). Assessment of memory in amnesic and dementia patients: Adaptation of behavioral tests used with non-human primates. INS Bulletin, 12, 25.
Moss, M. B., Albert, M. S., Butters, N., & Payne, M. (1986). Differential patterns of memory loss among patients with Alzheimer’s disease, Huntington’s disease, and alcoholic Korsakoff’s syndrome. Archives of Neurology, 43, 239–246.
Moss, M. B., Killiany, R. J., Lai, Z. C., Rosene, D. L., & Herndon, J. G. (1997). Recognition span in monkeys of advanced age. Neurobiology of Aging, 18, 13–19.
Moss, M. B., Killiany, R. J., Rosene, D. L., & Herndon, J. G. (2000). Recognition memory in aged rhesus monkeys: Oldest of the old. Manuscript submitted for publication.
Moss, M. B., Rosene, D. L., & Peters, A. (1988). Effects of aging on visual recognition memory in the rhesus monkey. Neurobiology of Aging, 9, 495–502.
Murray, E. A., & Mishkin, M. (1984). Severe tactual as well as visual memory deficits follow combined removal of the amygdala and hippocampus in monkeys. Journal of Neuroscience, 4, 2565–2580.
Murray, E. A., & Mishkin, M. (1986). Visual recognition in monkeys following rhinal cortical ablations combined with either amygdalectomy or hippocampectomy. Journal of Neuroscience, 6, 1991–2003.
O’Boyle, V. J., Murray, E. A., & Mishkin, M. (1993). Effects of excitotoxic amygdalo-hippocampal lesions on visual recognition in rhesus monkeys. Society for Neuroscience Abstracts, 19, 438.
Peters, A., Leahu, D., Moss, M. B., & McNally, K. J. (1994). The effects of aging on area 46 of the frontal cortex of the rhesus monkey. Cerebral Cortex, 6, 621–635.
Peters, A., Morrison, J. H., Rosene, D. L., & Hyman, B. T. (1998). Are neurons lost from the primate cortex during normal aging? Cerebral Cortex, 8, 295–300.
Peters, A., Rosene, D. L., Moss, M. B., Kemper, T. L., Abraham, C. R., Tigges, J., & Albert, M. S. (1997). Neurobiological bases of age related cognitive decline in the rhesus monkey. Journal of Neuropathology & Experimental Neurology, 55, 861–874.
Petrinovich, L. F., & Hardyck, C. D. (1969). Error rates for multiple comparison methods: Some evidence concerning the frequency of erroneous conclusions. Psychological Bulletin, 71, 43–54.
Presty, S. K., Bachevalier, J., Walker, L. C., Struble, R. G., Price, D. L., Mishkin, M., & Cork, L. C. (1987). Age differences in recognition memory of the rhesus monkey (Macaca mulatta). Neurobiology of Aging, 8, 435–440.
Rapp, P. R. (1990). Visual discrimination and reversal learning in the aged monkey (Macaca mulatta). Behavioral Neuroscience, 104, 876–884.
Rapp, P. R., & Amaral, D. G. (1989). Evidence for a task-dependent memory dysfunction in the aged monkey. Journal of Neuroscience, 9, 3568–3576.
Rapp, P. R., & Amaral, D. G. (1991). Recognition memory deficits in a subpopulation of aged monkeys resemble the effects of medial temporal lobe damage. Neurobiology of Aging, 12, 481–486.
Rehbein, L. (1985). Long-term effects of early hippocampectomy in the monkey. Unpublished doctoral dissertation, Northeastern University.
Rosene, D. L. (1993). Comparing age-related changes in the basal fore-brain and hippocampus of the rhesus monkey. Neurobiology of Aging, 14, 669–670.
Rosene, D. L., Lai, Z. C., Killiany, R. J., Moss, M. B., Jolesz, F., & Albert, M. S. (2000). Age-related loss of white matter with preservation of gray matter in the forebrain of the rhesus monkey—An MRI study. Manuscript submitted for publication.
Salmon, D. P., Granholm, E., McCullough, D., Butters, N., & Grant, I. (1989). Recognition memory span in mild and moderately demented patients with Alzheimer’s disease. Journal of Clinical & Experimental Neuropsychology, 4, 429–443.
Saunders, R. C., Murray, E. A., & Mishkin, M. (1984). Further evidence that amygdala and hippocampus contribute equally to recognition memory. Neuropsychologia, 22, 758–796.
Sloane, J., Rosene, D. L., Moss, M. B., Kemper, T., & Abraham, C. R. (1997). Lack of correlation between plaque burden and cognition in the aged monkey. Acta Neuropathologica, 94, 471–478.
Smith, G., Ivnik, R. J., Petersen, R. C., Malec, J. F., Kokmen, E., & Tangalos, E. (1991). Age-associated memory impairment diagnoses: Problems of reliability and concerns for terminology. Psychology & Aging, 6, 551–558.
Squire, L., Zola-Morgan, S., & Chen, K. S. (1988). Human amnesia and animal models of amnesia: Performance of amnesic patients on tests designed for the monkey. Behavioral Neuroscience, 102, 210–221.
Struble, R. G., Price, D. L., Jr., Cork, L. C., & Price, D. L. (1985). Senile plaques in cortex of aged normal monkeys. Brain Research, 361, 267–275.
Tigges, J., Gordon, T. P., McClure, H. M., Hall, E. C., & Peters, A. (1988). Survival rate and life span of rhesus monkeys at the Yerkes Regional Primate Research Center. American Journal of Primatology, 15, 263–273.
Tigges, J., Herndon, J. G., & Rosene, D. L. (1996). Preservation into old age of synaptic number and size in the supragranular layer of the dentate gyrus in rhesus monkeys. Acta Anatomica, 157, 63–72.
Walker, L. C., Kitt, C. A., Struble, R. G., Wagster, M. V., Price, D. L., & Cork, L. C. (1988). The neural basis of memory decline in aged monkeys. Neurobiology of Aging, 9, 657–666.
West, M. J. (1993). Regionally specific loss of neurons in the aging human hippocampus. Neurobiology of Aging, 14, 287–293.
Zola-Morgan, S., & Squire, L. R. (1985). Medial temporal lesions in monkeys impair memory on a variety of tasks sensitive to human amnesia. Behavioral Neuroscience, 99, 22–23.
Zola-Morgan, S., & Squire, L. R. (1986). Memory impairment in monkeys following lesions limited to the hippocampus. Behavioral Neuroscience, 100, 155–160.
Zola-Morgan, S., Squire, L. R., & Amaral, D. G. (1989). Lesions of the hippocampal formation but not lesions of the fornix or the mammillary nuclei produce long-lasting memory impairment in monkeys. Journal of Neuroscience, 9, 898–913.
Zola-Morgan, S., Squire, L. R., Clower, R. P., & Rempel, N. L. (1993). Damage to the perirhinal cortex exacerbates memory impairment following lesions to the hippocampal formation. Journal of Neuroscience, 13, 251–265.
Author information
Authors and Affiliations
Corresponding author
Additional information
This research was supported in part by Grant PO1-AG00001 from the National Institute on Aging, Bethesda, MD, and by a research training grant in the structural basis of neurobiology T32-NS07152 from the National Institute of Neurological Disorders and Stroke, Bethesda, MD. In addition, this study was supported by NIH Grant RR-00165 from the National Center for Research Resources to the Yerkes Regional Primate Research Center. Boston University School of Medicine and the Yerkes Regional Primate Research Center are both fully accredited by the American Association for Accreditation of Laboratory Animal Care. The authors thank Tara Moore, Beverly Duryea-Steiger, and John Herndon for their valuable assistance with this project.
Rights and permissions
About this article
Cite this article
Killiany, R.J., Moss, M.B., Rosene, D.L. et al. Recognition memory function in early senescent rhesus monkeys. Psychobiology 28, 45–56 (2000). https://doi.org/10.3758/BF03330628
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3758/BF03330628