Abstract
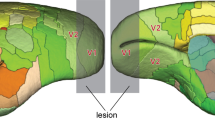
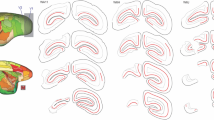
The influence of lesions located generally within four regions of the rat#’s posterior neocortex upon the acquisition and retention of two-choice visual discriminations was assessed using a Thompson-Bryant apparatus. The ability to distinguish visual patterns was tested by utilizing stimuli composed of 45° versus 135° alternating black and white stripes (both total and local luminance cues were controlled). “Brightness” discrimination was assessed by using black and white card discriminanda. Posterior parietal injuries resulted in a dysfunction on each of the four tasks, but rats with this injury eventually attained criterion for each of the habits. Smaller, more anteromedial parietal lesions failed to disturb pattern discrimination and were not studied in the other paradigms. The only deficit observed in rats with medial peristriate injuries was a slight retardation in acquisition of the pattern discrimination. Since the effects of lateral peristriate and striate injuries upon pattern vision have been well described previously, the effects of these lesions on the retention and acquisition of the black-white discrimination were studied. Lateral peristriate lesions, but not striate lesions, were associated with a retention loss of the brightness problem. This loss was detected even with a recovery interval of 60 days. Both striate and lateral peristriate lesions retarded black-white discrimination acquisition. Discussion focuses on the probable natures of the visual dysfunctions associated with these injuries.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Boyd, M. G., & Thomas, R. K. (1977). The effects of posterior association cortex lesions in rats: Mazes, pattern discrimination, and reversal learning. Physiological Psychology, 5, 455–461.
Cooper, R. M., Blochert, K. P., Gillespie, L. A., & Miller, L. G. (1972). Translucent occluders and lesions of posterior neocortex in the rat. Physiology & Behavior, 8, 693–697.
Dean, P. (1981). Visual pathways and visual acuity in hooded rats. Behavioural Brain Research, 3, 239–271.
Espinoza, S. G., & Thomas, H. C. (1983). Retinotopic organization of striate and extrastriate visual cortex in the hooded rat. Brain Research, 272, 137–144.
Fellows, B. J. (1967). Chance stimulus sequences for discrimination tasks. Psychological Bulletin, 67, 87–92.
Foreman, N., & Stevens, R. (1982). Visual lesions and radial maze performance in rats. Behavioral & Neural Biology, 36, 126–136.
Gallardo, L., Mottles, M., Vera, L., Carrasco, M. A., Torrealba, F., Montero, V. M., & Pinto-Hamuy, T. (1979). Failure by rats to learn a visual conditional discrimination after lateral peristriate cortical lesions. Physiological Psychology, 7, 173–177.
Gavin, M. R., & Isaac, W. (1986). Recovery of function of a conditioned avoidance response in rats with serial and single-stage bilateral occipital ablation. Physiological Psychology, 14, 31–35.
Goldstein, L. H., & Oakley, D. A. (1987). Visual discrimination in the absence of visual cortex. Behavioural Brain Research, 24, 181–193.
Goodale, M. A., & Dale, R. H. I. (1981). Radial-maze performance in the rat following lesions of posterior neocortex. Behavioural Brain Research, 3, 273–288.
Horel, J. A., Bettinger, L. A., Royce, G. J., & Meyer, D. R. (1966). Role of neocortex in the learning and relearning of two visual habits by the rat. Journal of Comparative & Physiological Psychology, 61, 66–78.
Hughes, H. C. (1977). Anatomical and neurobehavioral investigations concerning the thalamocortical organization of the rat#’s visual system. Journal of Comparative Neurology, 175, 311–336.
Kesner, R. P., DiMattia, B. A., & Crutcher, K. A. (1987). Evidence for neocortical involvement in reference memory. Behavioral & Neural Biology, 47, 40–53.
Kolb, B., Sutherland, R. J., & Whishaw, I. Q. (1983). A comparison of the contributions of the frontal and parietal association cortex to spatial localization in rats. Behavioral Neuroscience, 97, 13–27.
Kolb, B., & Walkey, J. (1987). Behavioural and anatomical studies of the posterior parietal cortex in the rat. Behavioural Brain Research, 23, 127–145.
Lashley, K. S. (1935). The mechanism of vision: XII. Nervous structures concerned in habits based on reactions to light. Comparative Psychology Monographs, 11, 43–79.
Lavond, D. G., & Dewbury, R. G. (1980). Visual form perception in a function of the visual cortex: II. The rotated horizontal-vertical and oblique-stripes pattern problems. Physiological Psychology, 8, 1–8.
Lavond, D., Hata, M. E., Gray, T. S., Geckler, C. L., Meyer, P. M., & Meyer, D. R. (1978). Visual form perception is a function of visual cortex. Physiological Psychology, 6, 471–477.
LeVere, T. E. (1980). Recovery of function after brain damage: A theory of the behavioral deficit. Physiological Psychology, 8, 297–308.
Lindsay, J. F., Jr., & McDaniel, W. F. (1987). Neural tissue transplants in rats with lateral peristriate lesions: II. Behavior. Medical Science Research, 15, 655–656.
McDaniel, W. F. (1985). Functions of the posterior neocortex of the rat. IRCS Journal of Medical Science, 13, 286–289.
McDaniel, W. F., & Brown, R. G. (1984). Radial maze performance following restricted posterior neocortical lesions. IRCS Medical Science, 12, 807–808.
McDaniel, W. F., Coleman, J., & Lindsay, J. E., Jr. (1982). A comparison of lateral peristriate and striate neocortical ablations in the rat. Behavioural Brain Research, 6, 249–272.
McDaniel, W. F., & Noble, L. M. (1984). Visual pattern discrimination following bilateral striate or lateral peristriate neocortical injuries. IRCS Medical Science, 12, 1084–1085.
McDaniel, W. F., & Thomas, R. K. (1978). Temporal and parietal association cortex lesions and spatial and black-white reversal learning in the rat. Physiological Psychology, 6, 300–305.
McDaniel, W. F.. Wildman, L. D., & Spears, R. H. (1979). Posterior association cortex and visual pattern discrimination in the rat. Physiological Psychology, 7, 241–244.
Meyer, D. R., & Meyer, P. M. (1977). Dynamics and bases of recoveries of functions after injuries to the cerebral cortex. Physiological Psychology, 5, 133–165.
Meyer, P. M., Meyer, D. R., & Cloud, M. D. (1986). Temporal neocortical injuries in rats impair attending but not complex visual processing. Behavioral Neuroscience, 100, 845–851.
Miller, M. W., & Vogt, B. A. (1984). Direct connections of rat visual cortex with sensory, motor, and association cortex. Journal of Comparative Neurology, 226, 184–202.
Milner, A. D., Goodale, M. A., & Morton, M. C. (1979). Visual sampling after lesions of superior colliculus in rats. Journal of Comparative & Physiological Psychology, 93, 1015–1023.
Mishkin, M. (1972). Cortical visual areas and their interaction. In A. G. Karczmar & J. C. Eccles (Eds.), The brain and human behavior (pp. 187–208). Berlin: Springer-Verlag.
Mishkin, M., Lewis, M. E., & Ungerleider, L. G. (1982). Equivalence of parieto-preoccipital subareas for visuospatial ability in monkeys. Behavioural Brain Research, 6, 41–55.
Mishkin, M., & Ungerleider, L. G. (1982). Contribution of striate inputs to the visuospatial functions of parieto-preoccipital cortex in monkeys. Behavioural Brain Research, 6, 57–77.
Montero, V. M., Rojas, A., & Torrealba, F. (1973). Retinotopic organization of striate and peristriate visual cortex in the albino rat. Brain Research, 53, 197–201.
Pasik, T., & Pasik, P. (1971). The visual world of monkeys deprived of striate cortex: Effective stimulus parameters and the importance of the accessory optic system. Vision Research, Suppl. No. 3, 419–435.
Pinto-Hamuy, T., Olavarria, J., Guic-Robles, E., Morgues, M., Nassal, O., & Petit, D. (1987). Rats with lesions in anteromedial extrastriate cortex fail to learn a visuosomatic conditional response. Behavioural Brain Research, 25, 221–231.
Thomas, R. K., & Peacock, L. J. (1965). A method of measuring brain lesions. Psychonomic Science, 3, 184.
Thomas, R. K., & Weir, V. R. (1975). The effects of lesions in the frontal or posterior association cortex of rats on Maze III. Physiological Psychology, 3, 210–214.
Thompson, R. (1979). Hippocampal and cortical function in a maze devoid of left and right turns. Physiology & Behavior, 23, 601–603.
Thompson, R., & Bryant, J. H. (1955). Memory as affected by activity of the relevant receptor. Psychological Reports, 1, 393–400.
Thompson, R., Huestis, P. W., Crinella, F. M., & Yu, J. (1986). The neuroanatomy of mental retardation in the white rat. Neuroscience & Biobehavioral Reviews, 10, 317–338.
Thompson, R., & Yu, J. (1985). The comparative effects of frontal, parietal, occipitotemporal, and limbic forebrain lesions in weanling rats on learning. Physiology & Behavior, 15, 559–567.
Toga, A. W., & Collins, R. C. (1981). Metabolic response of optic centers to visual stimuli in the albino rat: Anatomical and physiological considerations. Journal of Comparative Neurology, 199, 443–464. Torrealba, F., Olavarria, J., & Carrasco, M. A. (1984). Cortical connections of the anteromedial extrastriate visual cortex in rat. Experimental Brain Researach, 56, 543–549.
Ware, C. B., Diamond, I. T., & Casagrande, V. A. (1974). Effects of ablating the striate cortex on a successive pattern discrimination: Further study of the visual system in the tree shrew (Tupaia glis). Brain, Behavior & Evolution, 9, 264–279.
Weiskrantz, L. (1980). Varieties of residual experience. Quarterly Journal of Experimental Psychology, 32, 365–386.
Weiskrantz, L., & Cowey, A. (1963). Striate cortex lesions and visual acuity of the rhesus monkey. Journal of Comparative & Physiological Psychology, 56, 225–231.
Whishaw, I. Q., & Kolb, B. (1984). Decortication abolishes place but not cue learning in rats. Behavioural Brain Research, 11, 123–134.
Author information
Authors and Affiliations
Additional information
Three faculty research grants to the first author, funded by the Georgia College Foundation, made this work possible.
Rights and permissions
About this article
Cite this article
McDaniel, W.F., Wall, T.T. Visuospatial functions in the rat following injuries to striate, peristriate, and parietal neocortical sites. Psychobiology 16, 251–260 (1988). https://doi.org/10.3758/BF03327315
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3758/BF03327315