Abstract
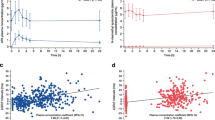
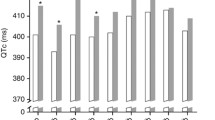
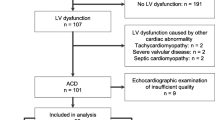
Objective: We designed a single-center retrospective study to assess the QT interval duration and to describe cardio vascular events among patients treated with mitotane for a adrenocortical carcinoma (ACC). Design: We selected 14 patients (6 males and 8 females) that met the following criteria: ACC treated with mitotane, for whom an electrocardiogram (ECG) at baseline (before mitotane initiation) was available and for whom at least one ECG was available during the course of mitotane therapy together with a concomitant mitotane plasma level determination. Results: Mean mitotane plasma level at baseline and after treatment showed a significant increase (mean level increased from 0 to 14.9±2 mg/l). At baseline and before mitotane was initiated all QTc intervals were <450 msec for men and <460 msec for women. During the treatment phase with mitotane, no QTc>470 msec was found in any patients respectively for men and women. In addition, no patient showed any significant QTc prolongation (>5% or >10 msec) at any time during mitotane treatment. During a mean follow-up of 15.9±3.5 months (range 2–45 months). No cardiovascular deaths or hospitalization for cardiovascular events was documented. No torsades de pointes were documented on ECG. No syncope, dizziness, heart failure were observed during follow up. Six out of 14 patients died during the follow-up, in five cases due to the progression of the disease, one patient died suddenly at home during follow-up. Conclusion: This short and retrospective series shows no evidence that mitotane induce any QT prolongation, even when plasma levels are well above the therapeutic window.
Similar content being viewed by others
References
Allolio B, Fassnacht M. Clinical review: Adrenocortical carcinoma: clinical update. J Clin Endocrinol Metab 2006, 91: 2027–37.
Abiven G, Coste J, Groussin L, et al. Clinical and biological features in the prognosis of adrenocortical cancer: poor outcome of cortisol-secreting tumors in a series of 202 consecutive patients. J Clin Endocrinol Metab 2006, 91: 2650–5.
Luton JP, Cerdas S, Billaud L, et al. Clinical features of adrenocortical carcinoma, prognostic factors, and the effect of mitotane therapy. N Engl J Med 1990, 322: 1195–2201.
Schulick RD, Brennan MF. Long-term survival after complete resection and repeat resection in patients with adrenocortical carcinoma. Ann Surg Oncol 1999, 6: 719–26.
Kendrick ML, Lloyd R, Erickson L, et al. Adrenocortical carcinoma: surgical progress or status quo? Arch Surg 2001, 136: 543–9.
Terzolo M, Angeli A, Fassnacht M, et al. Adjuvant mitotane treatment for adrenocortical carcinoma. N Engl J Med 2007, 356: 2372–80.
van Slooten H, Moolenaar AJ, van Seters AP, Smeenk D. The treatment of adrenocortical carcinoma with o,p′-DDD: prognostic implications of serum level monitoring Eur J Cancer Clin Oncol 1984, 20: 47–53.
Terzolo M, Pia A, Berruti A, et al. Low-dose monitored mitotane treatment achieves the therapeutic range with manageable side effects in patients with adrenocortical cancer. J Clin Endocrinol Metab 2000, 85: 2234–8.
Daffara F, De Francia S, Reimondo G, et al. Prospective evaluation of mitotane toxicity in adrenocortical cancer patients treated adjuvantly. Endocr Relat Cancer 2008, 15: 1043–53.
Veytsman I, Nieman L, Fojo T. Management of endocrine manifestations and the use of mitotane as a chemotherapeutic agent for adrenocortical carcinoma. J Clin Oncol 2009, 27: 4619–29.
Baudin E, Pellegriti G, Bonnay M, et al. Impact of monitoring plasma 1,1-dichlorodiphenildichloroethane (o,p′DDD) levels on the treatment of patients with adrenocortical carcinoma. Cancer 2001, 92: 1385–92.
Hermsen IG, Fassnacht M, Terzolo M, et al. Plasma concentrations of o,p′DDD, o,p′DDA, and o,p′DDE as predictors of tumor response to mitotane in adrenocortical carcinoma: results of a retrospective ENS@T multicenter study. J Clin Endocrinol Metab 2011, 96: 1844–51.
Pliquett RU, Eichfeld U, Stumvoll M, Koch CA. Long QT syndrome under mitotane therapy. J Endocrinol Invest 2007, 30: 167–8.
Drew BJ, Ackerman MJ, Funk M, et al. American Heart Association Acute Cardiac Care Committee of the Council on Clinical Cardiology, the Council on Cardiovascular Nursing, and the American College of Cardiology Foundation. Prevention of torsade de pointes in hospital settings: a scientific statement from the american heart association and the american college of cardiology foundation. Circulation 2010, 121: 1047–60.
Andersen A, Kasperlik-Zaluska AA, Warren DJ. Determination of mitotane (o,p-DDD) and its metabolites o,p-DDA and o,p-DDE in plasma by high-performance liquid chromatography. Ther Drug Monit 1999, 21: 355–9.
Nader N, Raverot G, Emptoz-Bonneton A, et al. Mitotane has an estrogenic effect on sex hormone-binding globulin and corticosteroid-binding globulin in humans. J Clin Endocrinol Metab 2006, 91: 2165–70.
Maher VM, Trainer PJ, Scoppola A, Anderson JV, Thompson GR, Besser GM. Possible mechanism and treatment of o,p′DDD-induced hypercholesterolaemia. Q J Med 1992, 84: 671–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ederhy, S., Cohen, A., Dufaitre, G. et al. No evidence for relevant QT interval prolongation in mitotane-treated patients with adrenocortical carcinoma. J Endocrinol Invest 35, 911–914 (2012). https://doi.org/10.3275/8616
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3275/8616