Abstract
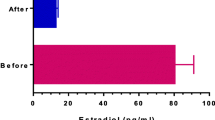
Background: Gonadotropins administration have been demonstrated effective for the treatment of idiopathic male infertility, even if no clear data regarding their specific mechanism of action on semen quality are at the moment available. Aim: To evaluate the effect of highly purified FSH (hpFSH) administration on standard semen parameters, sperm oxidative stress, and sperm chromatin structure and DNA fragmentation. Material and methods: In the current prospective baseline-controlled study, 36 subjects with male idiophatic infertility were enrolled. Baseline clinical and biochemical data were evaluated. Before and after 3 months of treatment with hpFSH, sperm samples were collected and standard semen analysis, reactive oxygen species (ROS) assessment and sperm DNA fragmentation were evaluated. Results: A significant (p<0.05) improvement from baseline in standard seminal parameters was observed. A significant (p<0.05) reduction in sperm ROS levels and DNA fragmentation was observed. Statistically significant (p<0.05) correlations between variation in ROS levels and both seminal parameters and DNA fragmentation variations were detected. Conclusions: Three months of hpFSH administration seems to reduce ROS and DNA damage in subjects with male idiopathic subfertility improving semen parameters. Further evidences from well-designed randomized double-blind placebo-controlled studies are needed in order to confirm our preliminary results.
Similar content being viewed by others
References
Thonneau P, Marchand S, Tallec A, et al. Incidence and main causes of infertility in a resident population (1,850,000) of three French regions (1988–1989). Hum Reprod 1991, 6: 811–6.
Hull MG, Eddowes HA, Fahy U, et al. Expectations of assisted conception for infertility. BMJ 1992, 304: 1465–9.
Nygren KG, Andersen AN. Assisted reproductive technology in Europe, 1999. Results generated from European registers by ESHRE. Hum Reprod 2002, 17: 3260–74.
Padron OF, Brackett NL, Sharma RK, Lynne CM, Thomas AJ, Agarwal A. Seminal reactive oxygen species and sperm motility and morphology in men with spinal cord injury. Fertil Steril 1997, 67: 1115–20.
Krausz C, West K, Buckingham D, Aitken RJ. Development of a technique for monitoring the contamination of human semen samples with leukocytes. Fertil Steril 1992, 57: 1317–25.
Ollero M, Gil-Guzman E, Lopez MC, et al. Characterization of subsets of human spermatozoa at different stages of maturation: implications in the diagnosis and treatment of male infertility. Hum Reprod 2001, 16: 1912–21.
Loft S, Kold-Jensen T, Hjollund NH, et al. Oxidative DNA damage in human sperm influences time to pregnancy. Hum Reprod 2003, 18: 1265–72.
Tesarik J, Mendoza-Tesarik R, Mendoza C. Sperm nuclear damage DNA change: update on the mechanism, diagnosis and treatment. Reprod Biomed Online 2006, 12: 715–21.
Aitken RJ, Fisher H. Reactive oxygen species generation and human spermatozoa: the balance of benefits and risk. Bioassays 1994, 16: 259–67.
De Lamirande E, Gagnon C. Impact of reactive species on spermatozoa: a balancing act between beneficial and detrimental effects. Hum Reprod 1995, 10: 15–21.
Ozdamar AS, Soylu AG, Culha M, Gökalp A. Testicular oxidative stress. Effects of experimental varicocele in adolescent rats. Urol Int 2004, 73: 343–7.
Yumura Y, Iwasaki A, Saito K, Ogawa T, Hirokawa M. Effect of reactive oxygen species in semen on the pregnancy of infertile couples. Int J Urol 2009, 16: 202–7.
Attia AM, Al-Inany HG, Farquhar C, Proctor M. Gonadotrophins for idiopathic male factor subfertility. Cochrane Database Syst Rev. 2007, 4: CD005071.
World Health Organization. WHO Laboratory Manual for the Examination of Human Semen and Sperm-Cervical Mucus Interaction. 3rd ed. Cambridge University Press, Cambridge, 1992.
World Health Organization. WHO Laboratory manual for the examination of human semen and sperm-cervical mucus interaction. Cambridge University Press, New York, 1999.
Orio F Jr, Palomba S, Di Biase S, et al. Homocysteine levels and C677T polymorphism of methylenetetrahydrofolate reductase in women with polycystic ovary sindrome. J Clin Endocrinol Metab 2003, 88: 673–9.
Kobayashi H, Gil-Guzman E, Mahran AM, et al. Quality control of reactive oxygen species measurement by luminol-dependent chemiluminescence assay. J Androl 2001, 22: 568–74.
Fernández JL, Muriel L, Goyanes V, et al. Simple determination of human sperm DNA fragmentation with an improved sperm chromatin dispersion test. Fertil Steril 2005, 84: 833–42.
Nieschlag E, Simoni M, Gromoll J, Weinbauer GF. Role of FSH in the regulation of spermatogenesis: clinical aspects. Clin Endocrinol (Oxf) 1999, 51: 139–46.
Foresta C, Bettella A, Merico M, et al. FSH in the treatment of oligozoospermia. Mol Cell Endocrinol 2000, 30: 89–97.
Foresta C, Bettella A, Merico M, Garolla A, Ferlin A, Rossato M. Use of recombinant human follicle-stimulating hormone in the treatment of male factor infertility. Fertil Steril 2002, 77: 238–44.
Foresta C, Selice R, Ferlin A, Garolla A. Recombinant FSH in the treatment of oligozoospermia. Expert Opin Biol Ther 2009, 9: 659–66.
Foresta C, Bettella A, Garolla A, Ambrosini G, Ferlin A. Treatment of male idiopathic infertility with recombinant human follicle-stimulating hormone: a prospective, controlled, randomized clinical study. Fertil Steril 2005, 84: 654–66.
Gualtieri AF, Mazzone GL, Rey RA, Schteingart HF. FSH and bFGF stimulate the production of glutathione in cultured rat Sertoli cells. Int J Androl 2009, 32: 218–25.
Ochsendorf FR, Buhl R, Bästlein A, Beschmann H. Glutathione in spermatozoa and seminal plasma of infertile men. Hum Reprod 1998, 13: 353–9.
Agarwal A, Makker K, Sharma R. Clinical relevance of oxidative stress in male factor infertility: an update. Am J Reprod Immunol 2008, 59: 2–11.
Coniglio JG. Testicular lipids. Prog Lipid Res 1994, 33: 387–401.
Sikka SC. Oxidative stress and role of antioxidants in normal and abnormal sperm function. Front Biosci 1996, 1: e78–86.
Venkatesh S, Deecaraman M, Kumar R, Shamsi MB, Dada R. Role of reactive oxygen species in the pathogenesis of mitochondrial DNA (mtDNA) mutations in male infertility. Indian J Med Res 2009, 129: 127–37.
Lopes S, Jurisicova A, Sun JG, Casper RF. Reactive oxygen species: potential cause for DNA fragmentation in human spermatozoa. Hum Reprod 1998, 13: 896–900.
Twigg J, Fulton N, Gòmez E. Analysis of the impact of intracellular reactive oxygen species generation on the structural and functional integrity of human spermatozoa: lipid peroxidation, DNA fragmentation and effective antioxidants. Hum Reprod 1998, 13: 1429–36.
Lin MH, Kuo-Kuang Lee R, Li SH, Lu CH, Sun FJ, Hwu YM. Sperm chromatin structure assay parameters are not related to fertilization rates, embryo quality, and pregnancy rates in in vitro fertilization and intracytoplasmic sperm injection, but might be related to spontaneous abortion rates. Fertil Steril 2008, 90: 352–9.
Appasamy M, Muttukrishna S, Pizzey AR, et al. Relationship between male reproductive hormones, sperm DNA damage and markers of oxidative stress in infertility. Reprod Biomed Online 2007, 14: 159–65.
Frank SA, Hurst LD. Mitochondria and male disease. Nature 1996, 383: 224.
Cummins JM, Jequier AM, Kan R. Molecular biology of human male infertility: links with aging, mitochondrial genetics, and oxidative stress? Mol Reprod Dev 1994, 37: 345–62.
St John JC, Cooke ID, Barratt CL. Mitochondrial mutations and male infertility. Nat Med 1997, 3: 124–5.
Moskovtsev SI, Willis J, White J, Mullen JB. Sperm DNA damage: correlation to severity of semen abnormalities. Urology 2009, 74: 789–93.
Smit M, Romijn JC, Wildhagen MF, Weber RF, Dohle GR. Sperm chromatin structure is associated with the quality of spermatogenesis in infertile patients. Fertil Steril 2010, 94: 1748–52.
Mehdi M, Khantouche L, Ajina M, Saad A. Detection of DNA fragmentation in human spermatozoa: correlation with semen parameters. Andrologia 2009, 41: 383–6.
Fernández JL, Muriel L, Goyanes V, et al. Halosperm is an easy, available, and cost-effective alternative for determining sperm DNA fragmentation. Fertil Steril 2005, 84: 860.
Chohan KR, Griffin JT, Lafromboise M, De Jonge CJ, Carrell DT. Comparison of chromatin assays for DNA fragmentation evaluation in human sperm. J Androl 2006, 27: 53–9.
Enciso M, Muriel L, Fernández JL, et al. Infertile men with varicocele show a high relative proportion of sperm cells with intense nuclear damage level, evidenced by the sperm chromatin dispersion test. J Androl 2006, 27: 106–11.
Velez de la Calle JF, Muller A, Walschaerts M, et al. Sperm deoxyribonucleic acid fragmentation as assessed by the sperm chromatin dispersion test in assisted reproductive technology programs: results of a large prospective multicenter study. Fertil Steril 2008, 90: 1792–9.
Saleh RA, Agarwal A, Nada EA, et al. Negative effects of increased sperm DNA damage in relation to seminal oxidative stress in men with idiopathic and male factor infertility. Fertil Steril 2003, 79: 1597–605.
Henkel R, Kierspel E, Hajimohammad M, et al. DNA fragmentation of spermatozoa and assisted reproduction technology. Reprod Biomed Online 2003, 7: 477–84.
Ménézo YJ, Hazout A, Panteix G, et al. Antioxidants to reduce sperm DNA fragmentation: an unexpected adverse effect. Reprod Biomed Online 2007, 14: 418–21.
Tunc O, Thompson J, Tremellen K. Improvement in sperm DNA quality using an oral antioxidant therapy. Reprod Biomed Online 2009, 18: 761–8.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Palomba, S., Falbo, A., Espinola, S. et al. Effects of highly purified follicle-stimulating hormone on sperm DNA damage in men with male idiopathic subfertility: A pilot study. J Endocrinol Invest 34, 747–752 (2011). https://doi.org/10.3275/7745
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3275/7745