Abstract
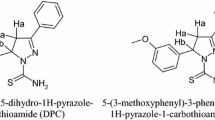
Six new pyrazole derivatives were successfully prepared and investigated as corrosion inhibitors for mild steel in 1 M H2SO4 solution by weight loss measurements for 24 h at 30°C. It was observed that at higher inhibitor concentrations, the rate of corrosion decreases, the inhibition efficiency increases as well as the surface coverage degree. The values of the standard free energy of adsorption showed that all prepared pyrazole derivatives afford physisorption effects. The correlation between experimental results and theoretical data could be concluded by using semiempirical molecular orbital calculations for the six studied inhibitors. Theoretical calculations were used to understand the interaction nature between the metal surface and the organic molecules as a corrosion inhibitors.







Similar content being viewed by others
REFERENCES
Bing, T.D.L., Fenglian, F., Yichun, X., Guojun, Y. and Jianyin, Z., Ind. Eng. Chem. Res., 2012, vol. 51, pp. 2615–2621.
Yan, L., Peng, Z., Qiang, L. and Baorong, H., Appl. Surf. Sci., 2005, vol. 252, pp. 1245–1253.
Shihab, M.S. and Al-Doori, H.H., J. Mol. Struct., 2014, vol. 1076, pp. 658–663.
Shihab, M.S. and Mahmood, A.F., Russ. J. Appl. Chem., 2016, vol. 89, pp. 505–516.
Shanmugakala, R., Tharmaraj, P., Sheela, C.D. and Anitha, C., Int. J. Inorg. Chem., 2012, vol. 2012, pp. 1–7.
Álvarez-Rodríguez, L., Cabeza, J.A., García-Álvarez, P., and Polo, D., Coord. Chem. Rev., 2015, vol. 300, pp. 1–28.
Escobar-Nuricumbo, J.J., Campos-Alvarado, C., Ríos-Moreno, G., Morales-Morales, D., Walsh, P.J., and Parra-Hake, M., Inorg. Chem., 2007, vol. 46, pp. 6182–6189.
Al-Saadawy, N.H. and Faraj, H.R., Am. Chem. Sci. J., 2016, vol. 10, pp. 1–5.
Abdel-Rehim, S.S., Khaled, K.F., and Al-Mobarak, N.A., Arab. J. Chem., 2011, vol. 4, pp. 333–337.
Abdallah, M., Megahed, H.E., and Motae, M.S., Monats Chem., 2010, vol. 141, pp. 1287–1295.
Abdel Hameed, R.S. and Abdallah, M., Prot. Met. Phys. Chem. Surf., 2018, vol. 54, pp. 113–121.
Vinutha, M.R. and Venkatesha, T.V., Port. Electrochim. Acta, 2016, vol. 34, pp. 157–184.
Vogel, A., Tatchell, A., Furnis, B., Hannaford, A. and Smith, P., Vogel’s Textbook of Practical Organic Chemistry, New York: Prentice Hall, 1996, 5th ed.
Conard, R.C. and Dolliver, A.M., Org. Synth.,Coll., 1943, vol. 2, p. 167.
Tirlapur, V., Gandhi, N., Basawaraj, R. and Prasad, R., Int. J. ChemTech Res., 2010, vol. 2, pp. 1434–1440.
Ajmal, M., Mideen, A.S. and Quraishi, M.A., Corros. Sci., 1994, vol. 36, pp. 79–84.
Scendo, M. and Hepel, M., Corros. Sci., 2007, vol. 49, pp. 3381–3407.
Scendo, M., Corros. Sci., 2007, vol. 49, pp. 3953–3968.
Herrag, L., Bouklah, M., Patel, N.S., Mistry, B.M., Hammouti, B., Elkadiri, S. and Bouachrine, M., Res. Chem. Intermed., 2012, vol. 38, pp. 1669–1690.
Verma, C., Quraishi, M.A. and Singh, A., J. Taibah Univ. Sci., 2016, vol. 10, pp. 718–733.
Xu, B., Ji, Y., Zhang, X., Jin, X., Yang, W., and Chen, Y., RSC Adv., 2015, vol. 5, pp. 56049–56059.
Stewart, J.P.J., J. Comput. Chem., 1989, vol. 10, pp. 209–220.
Obi-Egbedi, N.O. and Obot, I.B., Arab. J. Chem., 2013, vol. 6, pp. 211–223.
Kamis, E., Bellucci, F., Latanision, R.M., and El-Ashry, E.S.H., Corrosion, 1991, vol. 47, pp. 677–686.
Bentiss, F., Lebrini, M., and Lagrenée, M., Corros. Sci., 2005, vol. 47, pp. 2915–2931.
Obot, I.B., Gasem, Z.M. and Umoren, S.A., Int. J. Electrochem. Sci., 2014, vol. 9, pp. 510–522.
Zhang, K., Xu, B., Yang, W., Yin, X., Liu, Y. and Chen, Y., Corros. Sci., 2015, vol. 90, pp. 284–295.
Li, W., He, Q., Pei, C. and Hou, B., Electrochim. Acta, 2007, vol. 52, pp. 6386–6396.
Martinez, S., Mater. Chem. Phys., 2003, vol. 77, pp. 97–102.
Cao, Z., Tang, Y., Cang, H., Xu, J., Lu, G., and Jing, W., Corros. Sci., 2014, vol. 83, pp. 292–298.
Kumar, S., Sharma, D., Yadav, P., and Yadav, M., Ind. Eng. Chem. Res., 2013, vol. 52, pp. 14019–14029.
Sastri, V.S. and Perumareddi, J.R., Corros Sci., 1997, vol. 53, pp. 617–622.
Dewar, M.J.S. and Thiel, W., J. Am. Chem. Soc., 1977, vol. 99, pp. 4899–4907.
Dutta, A., Saha, S., Banerjee, P., and Sukul, D., Corros. Sci., 2015, vol. 98, pp. 541–550.
Lesar, A. and Milosev, I., Chem. Phys. Lett., 2009, vol. 483, pp. 198–203.
Obot, I.B. and Gasem, Z.M., Corros. Sci., 2014, vol. 83, pp. 359–366.
Singh, A.K., Ind. Eng. Chem. Res., 2012, vol. 51, pp. 3215–3223.
ACKNOWLEDGMENTS
The authors would like to thank the staff of the Department of Chemistry, College of Science, Al-Nahrain University, Iraq for their help and cooperation throughout this research.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Zahraa Talib Khudhair, Mehdi Salih Shihab Study of Synergistic Effect of Some Pyrazole Derivatives as Corrosion Inhibitors for Mild Steel in 1 M H2SO4 . Surf. Engin. Appl.Electrochem. 56, 601–609 (2020). https://doi.org/10.3103/S1068375520050142
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1068375520050142