Abstract
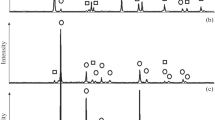
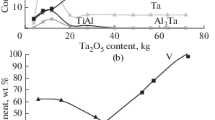
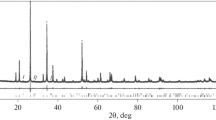
The features of phase formation during the joint aluminothermic reduction of titanium, niobium, tantalum, and vanadium from their oxides are studied using thermodynamic modeling and differential thermal (DTA) and X-ray diffraction phase analysis. Using computer thermodynamic modeling made it possible to predict the optimal temperature conditions in the metallothermic process, the composition and ratio of the reagents in the charge, the behavior of the elements, and the sequence of the phase formation. To identify the kinetic and thermochemical components of the process, thermodynamic calculations are supplemented by differential thermal studies using combined scanning calorimetry. An analysis of the theoretical and experimental data shows that the interaction of aluminum with titanium dioxide proceeds through the stage of formation of titanium monoxide and results in the formation of TixAly intermetallic compounds of various compositions (TiAl3, TiAl, and Ti2Al) depending on the Al to TiO2 ratio in the batch. When titanium dioxide is partially replaced by niobium, tantalum, and vanadium oxides, the metallothermic process under interactions in the Al–TiO2–Nb2O5, Al–TiO2–Ta2O5, and Al–TiO2–V2O5 systems is of the similar character; enters the active phase after the formation of liquid aluminum; is accompanied by exothermic effects; and is characterized by the priority formation of titanium aluminides and binary and ternary intermetallic aluminum compounds with rare refractory metals of group V such as AlNb3, Al3Nb, Al3Ta, Al3(Ti1 – х,Taх), Al3(Ti,Ta), and Al3(Ti0.8V0.2). The joint conversion of titanium dioxide and rare refractory metal pentoxides during the reduction process is affected through sequential and parallel stages of the formation of simple and complex element oxides with low oxidation states.












Similar content being viewed by others
REFERENCES
Chufarov, G.I. and Tatievskaya, E.P., Adsorption-catalytic theory of metal oxides reduction, in Problemy metallurgii (Problems on Metallurgy), Samarin, A.N., Ed., Moscow: USSR Acad. Sci., 1953, pp. 15–32.
Chufarov, G.I. and Tatievskaya, E.P., Mechanism and kinetics of metal oxides reduction, in Fiziko-khimicheskie osnovy domennogo protsessa i sovremennaya praktika proizvodstva chuguna (Physicochemical Foundations of the Blast Furnace Process and Modern Practice of Cast Iron Production), Mikhailov, V.V., Ed., Sverdlovsk: Gosudarstvennoe Nauchno-Tekhnicheskoe Izd. po Chernoi i Tsvetnoi Metallurgii, 1956, pp. 21–64.
Krutilin, A.N., Kukharchuk, M.N., and Sycheva, O.A., Solid phase recovery of iron oxides by carbon, Lit’e Metall., 2012, no. 2, pp. 11–16.
Egry, I., Brooks, R., Holland-Moritz, D., Novakovich, R., Matsushita, T., Ricci, E., Seetharaman, S., Wunderlich, R., and Jarvis, D., Thermophysical properties of γ-titanium aluminide: The European IMPRESS Project, Int. J. Thermophys., 2007, no. 28, pp. 1026–1036.
Mahdouk, K., Gachon, J.-C., and Boirden, L., Enthalpies of formation of the Al-Nb intermetallic compounds, J. Alloys Compd., 1998, no. 268, pp. 118–121.
Novakovic, R., Giuranno, D., Ricci, E., Tuissi, A., Wunderlich, R., Fecht, H.-J., and Egry, I., Surface, dynamic and structural properties of liquid Al–Ti alloys, Appl. Surf. Sci., 2012, no. 258, pp. 3269–3275.
Asta, M., de Fontaine, D., and Van Schilfgaarde, M., First-principles study of phase stability of Ti–Al intermetallic compounds, J. Mater. Res., 1993, vol. 8, p. 2554.
Boehlert, C.J., Part III. The tensile behavior of Ti–Al–Nb O + Bcc orthorhombic alloys, Metall. Mater. Trans. A, 2001, vol. 32, pp. 1977–1988.
Popil, F. and Douin, J., The dislocation microstructure in orthorhombic O Ti2AlNb deformed between room temperature and 800°C, Philos. Mag. A, 1996, vol. 73, no. 5, pp. 1401–1418.
Chungen Zhou, Huibin Xu, and Kyoo Young Kim, The influence of additions of Nb and Cr on the aluminizing behavior of TiAl alloy, Metall. Mater. Trans. A, 2000, vol. 31, no. 10, pp. 2391–2394.
Chen, W., Li, J.W., Xu, L., and Lu, B., Development of Ti2AlNb alloys: opportunities and challenges, Adv. Mater. Processes, 2014, vol. 175, no. 5, pp. 23–27.
Jihua Peng, Shiqiong Li, Yong Mao, and Xunfang Sun, Phase transformation and microstructures in Ti–Al–Nb–Ta system, Mater. Lett., 2002, no. 53, pp. 57–62.
Li-Hua Ye, Hao Wang, Gang Zhou, Qing-Miao Hu, and Rui Yang, Phase stability of TiAl–X (X = V, Nb, Ta, Cr, Mo, W, and Mn) alloys, J. Alloys Compd., 2020, vol. 819, article no. 153291. https://doi.org/10.1016/j.jallcom.2019.153291
Andreev, D.E., Sanin, V.N., and Yukhvid, V.I., SVS metallurgy of titanium aluminides, Int. J. Self-Propag. High-Temp. Synth., 2005, vol. 14, no. 3, pp. 219–234.
Napalkov, V.I. and Makhov, S.V., Legirovanie i modifitsirovanie alyuminiya i magniya (Alloying and Modifying of Aluminum and Magnesium), Moscow: Moscow State Institute of Steel and Alloys Technological Univ., 2002.
Murach, N.N. and Lisienko, V.T., Alyuminotermiya titana (Aluminothermy of Titanium), Moscow: TsNIITsvetmet, 1958.
Gasik, M.I., Lyakishev, N.P., and Emlin, B.I., Teoriya i tekhnologiya proizvodstva ferrosplavov (Theory and Technology of Ferroalloy Production), Moscow: Metallurgiya, 1988.
Krasikov, S.A., Nadol’skii, A.L., Ponomarenko, A.A., Sitnikova, O.A., and Zhidovinova, S.V., Metallothermic preparation of titanium-aluminum alloys under temperature-controlled conditions, Tsvetn. Met., 2012, no. 6, pp. 68–71.
Vedmid’, L.B., Krasikov, S.A., Zhilina, E.M., Nikitina, E.V., Evdokimova, I.V., and Merkushev, A.G., Evolution of phase formation during the aluminothermic reduction of titanium and zirconium from oxides, Russ. Metall. (Engl. Transl.), 2018, vol. 2018, no. 8, pp. 733–736.
Krasikov, S.A., Zhilina, E.M., Pichkaleva, O.A., Ponomarenko, A.A., Vedmid’, L.B., Zhidovinova, S.V., and Chentsov, V.P., Effect of the intermetallic compound composition of the character of interphase interactions during aluminothermic co-reduction of titanium, nickel, and molybdenum from their oxides, Russ. Metall. (Engl. Transl.), 2016, vol. 2016, no. 8. pp. 771–775.
Zhilina, E.M., Krasikov, S.A., Agafonov, S.N., Vedmid’, L.B., and Zhidovinova, S.V., Thermodynamic and kinetic peculiarities of joint aluminothermic reduction of titanium and zirconium from oxides, Butlerov. Soobshch., 2016, vol. 45, no. 1, pp. 130–135.
Nunes, C.A., Pinatti, D.G., and Robin, A., Nb–Ta alloys by aluminothermic reduction of Nb2O5/Ta2O5 mixtures and electron beam melting, Int. J. Refract. Met. Hard Mater., 1999, no. 17, pp. 305–314.
Roine, A., Outokumpu HSC Chemistry for Windows. Chemical Reaction and Equilibrium Software with Extensive Thermochemical Database, Pori: Outokumpu Research OY, 2006.
Feng, W., Wang, Q., Kong, Q., Zhu, X., Wu, J., and Sun, Ch., Influence of high-temperature water vapor on titanium film surface, Oxid. Met., 2016, vol. 86, pp. 179–192.
Funding
This work was perforned as part of a state assignment to the Institute of Metallurgy, Ural Branch, Russian Academy of Sciences, using the equipment of the Ural-M Shared Resource Center.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no conflict of interest.
Additional information
Translated by O. Lotova
About this article
Cite this article
Balakirev, V.F., Osinkina, T.V., Krasikov, S.A. et al. Joint Metallothermic Reduction of Titanium and Rare Refractory Metals of Group V. Russ. J. Non-ferrous Metals 62, 190–196 (2021). https://doi.org/10.3103/S1067821221020024
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1067821221020024