Abstract
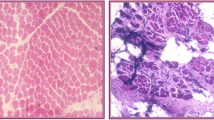
Congenital muscular dystrophy (MDC) is a group of rare hereditary myopathies with an early onset of progressive muscle weakness and dystrophic changes as evidenced by muscle biopsy. Some forms are associated with severe malformations of the brain. This study presented 2 pediatric patients with genetically diagnosed congenital muscular dystrophy 1A. The patients exhibited a typical combination of muscular hypotonia, joint contractures and elevated creatine kinase levels. Characteristic white matter lesions were not present in an early MRI scan of one patient, but could be detected at the age of 18 months. The second patient showed both severe white and grey matter abnormalities (pachy microgyria) in the MRI scan. In both cases, MRI findings did not correlate with the mental development of the patients.
Similar content being viewed by others
References
Reed U.C., Congenital muscular dystrophy. Part I: a review of phenotypical and diagnostic aspects, Arq. Neuro-Psiquiatr., 2009, 67, 144–168
Reed U.C., Congenital muscular dystrophy. Part II: a review of pathogenesis and therapeutic perspectives, Arq. Neuro-Psiquiatr., 2009, 67, 343–362
Darin N., Tulinius M., Neuromuscular disorders in childhood: a descriptive epidemiological study from western Sweden, Neuromusc. Disord., 2000, 10, 1–9
Mostacciuolo M.L., Angelini C., Marinelloa F., Miorin M., Perini P., Trevisan C.P., Genetic epidemiology of congenital muscular dystrophy in a sample from north-east Italy, Hum. Genet., 1996, 97, 277–279
Shorer Z., Philpot J., Muntoni F., Sewry C., Dubowitz V., Demyelinating peripheral neuropathy in merosin-deficient congenital muscular dystrophy, J. Child Neurol., 1995, 10, 472–475
Farina L., Morandi L., Milanesi I., Ciceri E., Mora M., Moroni I., et al., Congenital muscular dystrophy with merosin deficiency: MRI findings in five patients, Neuroradiology, 1998, 40, 807–811
Philpot J., Cowan F., Pennock J., Sewry C., Dubowitz V., Bydder G., et al. Merosin-deficient congenital muscular dystrophy: the spectrum of brain involvement on magnetic resonance imaging, Neuromusc. Disord., 1999, 9, 81–85
Schiffmann R., van der Knaap, M. S. An MRI-based approach to the diagnosis of white matter disorders, Neurology, 2009, 72, 750–759
Pini A., Merlini L., Tomé F.M., Chevallay M., Gobbi G., Merosin-negative congenital muscular dystrophy, occipital epilepsy with periodic spasms and focal cortical dysplasia: report of three Italian cases in two families, Brain Dev., 1996, 18, 316–322
Fujii Y., Sugiura C., Fukuda C., Maegaki Y., Ohno K., Sequential neuroradiological and neurophysiological studies in a Japanese girl with merosin-deficient congenital muscular dystrophy, Brain Dev., 2011, 33, 140–144
van der Knaap M.S., Smit L.M., Barth P.G., Catsman-Berrevoets C.E., Brouwer O.F., Begeer J.H., et al., Magnetic resonance imaging in classification of congenital muscular dystrophies with brain abnormalities Ann. Neurol., 1997, 42, 50–59
Caro P.A., Scavina M., Hoffman E., Pegoraro E., Marks H.G., MR imaging findings in children with merosin-deficient congenital muscular dystrophy, Am. J. Neuroradiol., 1999, 20, 3324–326
Villanova M., Malandrini A., Sabatelli P., Sewry C.A., Toti P., Torelli S., et al., Localization of laminin alpha 2 chain in normal human central nervous system: an immunofluorescence and ultrastructural study, Acta Neuropathol., 1997, 94, 576–571
Leite C.C., Reed U.C., Otaduy M.C., Lacerda M.T., Costa M.O., Ferreira L.G., et al., Congenital muscular dystrophy with merosin deficiency: 1H MR spectroscopy and diffusion-weighted MR imaging, Radiology, 2005, 235, 190–196
Sijens P.E., Fock J.M., Meiners L.C., Potze J.H., Irwan R., Oudkerk M., MR spectroscopy and diffusion tensor imaging of the brain in congenital muscular dystrophy with merosin deficiency: metabolite level decreases, fractional anisotropy decreases, and apparent diffusion coefficient increases in the white matter, Brain Dev., 2007, 29, 317–321
Sunada Y., Edgar T.S., Lotz B.P., Rust R.S., Campbell K.P., Merosinnegative congenital muscular dystrophy associated with extensive brain abnormalities, Neurology 1995, 45, 2084–2089
Messina S., Bruno C., Moroni I., Pegoraro E., D’Amico A., Biancheri R., et al., Congenital muscular dystrophies with cognitive impairment. A population study, Neurology 2010, 7. 75, 898–903
Vigliano P., Dassi P., Di Blasi C., Mora M., Jarre L., LAMA2 stop-codon mutation: merosin-deficient congenital muscular dystrophy with occipital polymicrogyria, epilepsy and psychomotor regression, Eur. J. Paediatr. Neurol., 2009, 13, 72–6
Matsumura K., Yamada H., Saito F., Sunada Y., Shimizu T., Peripheral nerve involvement in merosin-deficient congenital muscular dystrophy and dy mouse, Neuromusc. Disord., 1997, 7, 7–12
Louhichi N., Triki C., Morinière M., Rebai A., Richard P., et al., Severe MDC1A congenital muscular dystrophy due to a splicing mutation in the LAMA2 gene resulting in exon skipping and significant decrease of mRNA level, Genet. Test., 2007, 11: 199–207
Siala O., Louhichi N., Triki C., Morinière M., Fakhfakh F., Baklouti F., LAMA2 mRNA processing alterations generate a complete deficiency of laminin-alpha 2 protein and a severe congenital muscular dystrophy, Neuromusc. Disord., 2008, 18, 137–145
Oliveira J., Santos R., Soares-Silva I., Jorge P., Vieira E., Oliveira M.E., et al, LAMA2 gene alalysis in a cohort of 26 congenital muscular dystrophy patients, Clin. Genet., 2008, 74, 502–512
Geranmayeh F., Clement E., Feng L.H., Sewry C., Pagan J., Mein R., et al., Genotype-phenotype correlation in a large population of muscular dystrophy patients with LAMA2 mutations, Neuromusc. Disord., 2010, 20, 241–250
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributed equally
About this article
Cite this article
Köhler, C., Weigt-Usinger, K., Heyer, C.M. et al. CNS findings in congenital muscular dystrophy 1A (with laminin alpha-2-deficiency). Translat.Neurosci. 2, 138–141 (2011). https://doi.org/10.2478/s13380-011-0020-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s13380-011-0020-7