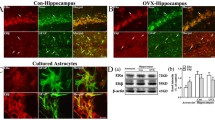
Abstract
17β-estradiol is known to exert neurotrophic and neuroprotective effects through classical estrogen receptors [ERs], ERα and ERβ, on a variety of cell types either by genomic or non-genomic actions. The actions of estradiol on glial cells are important to maintain metabolic functions of the nervous system. Astrocytes are considered to be active participants in brain activity because of their ability to release growth factors, including neurotrophins. Present in vitro studies show that 17β-estradiol modulates NGF and BDNF expression in time-dependent manner and ERK acts as secondary messenger for estradiol’s action. 17β-estradiol is involved in survival of cortical astrocytes. In conclusion, this study indicates vital role of ERβ mediated ERK signalling for regulation of NGF and BDNF expression along with cell viability of cortical astrocytes which further confirms the role of ERs, particularly ERβ in glial cells’ functions and viability.






Similar content being viewed by others
Abbreviations
- E2:
-
17β-estradiol
- ERs:
-
Estradiol Receptors
- ERα:
-
Estradiol Receptor alpha
- ERβ:
-
Estradiol Receptor beta
- NGF:
-
Nerve growth factor
- BDNF:
-
Brain derived neurotrophic factor
- NT3:
-
Neurotrophins 3
- NT4/5:
-
Neurotrophins 4/5
- ERK:
-
Extracellular regulated kinase
- MEK:
-
Mitogen activated protein kinase
- GFAP:
-
Glial fibrillary acidic protein
References
Abcam-protocols. (2016a) Immunocytochemistry and immunofluorescence protocol. Retrieved from http://www.abcam.com/protocols/immunocytochemistry-immunofluorescence-protocol
Abcam-protocols. (2016b) Western blot protocol. Retrieved from http://www.abcam.com/protocols/general-western-blot-protocol
Adams C, Kumar R (2013) The effect of estrogen in a man with Parkinson's disease and a review of its therapeutic potential. Int J Neurosci 123(10):741–742. https://doi.org/10.3109/00207454.2013.797903
Aiga A, Asaumi K, Lee YJ, Kadota H, Mitani S, Ozaki T, Takigawa M (2006) Expression of neurotrophins and their receptors tropomyosin-related kinases (Trk) under tension-stress during distraction osteogenesis. Acta Med Okayama 60(5):267–277. https://doi.org/10.18926/AMO/30739
Allen SJ, Dawbarn D (2006) Clinical relevance of the neurotrophins and their receptors. Clin Sci (Lond) 110(2):175–191. https://doi.org/10.1042/CS20050161
Allen SJ, Watson JJ, Shoemark DK, Barua NU, Patel NK (2013) GDNF, NGF and BDNF as therapeutic options for neurodegeneration. Pharmacol Ther 138(2):155–175. https://doi.org/10.1016/j.pharmthera.2013.01.004
Arita E, Kondoh M, Isoda K, Nishimori H, Yoshida T, Mizuguchi H, Yagi K (2008) Evaluation of promoter strength in mouse and rat primary hepatocytes using adenovirus vectors. Eur J Pharm Biopharm 70(1):1–6. https://doi.org/10.1016/j.ejpb.2008.03.013
Bains M, Cousins JC, Roberts JL (2007) Neuroprotection by estrogen against MPP+−induced dopamine neuron death is mediated by ERα in primary cultures of mouse mesencephalon. Exp Neurol 204(2):767–776. https://doi.org/10.1016/j.expneurol.2007.01.020
Balaratnasingam S, Janca A (2012) Brain derived neurotrophic factor: a novel neurotrophin involved in psychiatric and neurological disorders. Pharmacol Ther 134(1):116–124. https://doi.org/10.1016/j.pharmthera.2012.01.006
Birge SJ (1997) The role of estrogen in the treatment of Alzheimer's disease. Neurology 48(5 Suppl 7):36S–41S. https://doi.org/10.1212/WNL.48.5_Suppl_7.36S
Blázquez C, Galve-roperh I, GuzmÁn M (2000) De novo-synthesized ceramide signals apoptosis in astrocytes via extracellular signal-regulated kinase. FASEB J 14(14):2315–2322. https://doi.org/10.1096/fj.00-0122com
Bucci C, Alifano P, Cogli L (2014) The role of Rab proteins in neuronal cells and in the trafficking of neurotrophin receptors. Membranes (Basel) 4(4):642–677. https://doi.org/10.3390/membranes4040642
Cattaneo A, Calissano P (2012) Nerve growth factor and Alzheimer's disease: new facts for an old hypothesis. Mol Neurobiol 46(3):588–604. https://doi.org/10.1007/s12035-012-8310-9
Chan, JR, Watkins, TA, Cosgaya, JM, Zhang, C, Chen, L, Reichardt, LF, … Barres, BA (2004). NGF controls axonal receptivity to myelination by Schwann cells or oligodendrocytes. Neuron 43(2), 183–191. https://doi.org/10.1016/j.neuron.2004.06.024
Chen Y, Balasubramaniyan V, Peng J, Hurlock EC, Tallquist M, Li J, Lu QR (2007) Isolation and culture of rat and mouse oligodendrocyte precursor cells. Nat Protoc 2(5):1044–1051. https://doi.org/10.1038/nprot.2007.149
Cheng B, Mattson MP (1994) NT-3 and BDNF protect CNS neurons against metabolic/excitotoxic insults. Brain Res 640(1):56–67. https://doi.org/10.1016/0006-8993(94)91857-0
Choi K, Ni L, Jonakait GM (2011) Fas ligation and tumor necrosis factor alpha activation of murine astrocytes promote heat shock factor-1 activation and heat shock protein expression leading to chemokine induction and cell survival. J Neurochem 116(3):438–448. https://doi.org/10.1111/j.1471-4159.2010.07124.x
Dhandapani KM, Brann DW (2007) Role of astrocytes in estrogen-mediated neuroprotection. Exp Gerontol 42(1):70–75. https://doi.org/10.1016/j.exger.2006.06.032
Dittrich F, Feng Y, Metzdorf R, Gahr M (1999) Estrogen-inducible, sex-specific expression of brain-derived neurotrophic factor mRNA in a forebrain song control nucleus of the juvenile zebra finch. Proc Natl Acad Sci U S A 96(14):8241–8246. https://doi.org/10.1073/pnas.96.14.8241
Dong J, Song N, Xie J, Jiang H (2009) Ghrelin antagonized 1-methyl-4-phenylpyridinium (MPP+)-induced apoptosis in MES23. 5 cells. J Mol Neurosci 37(2):182–189. https://doi.org/10.1007/s12031-008-9162-7
Douglas-Escobar M, Rossignol C, Steindler D, Zheng T, Weiss MD (2012) Neurotrophin-induced migration and neuronal differentiation of multipotent astrocytic stem cells in vitro. PLoS One 7(12):e51706. https://doi.org/10.1371/journal.pone.0051706
Fuchikami M, Morinobu S, Kurata A, Yamamoto S, Yamawaki S (2009) Single immobilization stress differentially alters the expression profile of transcripts of the brain-derived neurotrophic factor (BDNF) gene and histone acetylation at its promoters in the rat hippocampus. Int J Neuropsychopharmacol 12(1):73–82. https://doi.org/10.1017/S1461145708008997
Fuente-Martin, E, Garcia-Caceres, C, Morselli, E, Clegg, D, Chowen, J, Finan, B, … Tschöp, M (2013). Estrogen, astrocytes and the neuroendocrine control of metabolism. Rev Endocr Metab Disord 14(4), 331–338. https://doi.org/10.1007/s11154-013-9263-7
Gould E, Woolley CS, Frankfurt M, McEwen BS (1990) Gonadal steroids regulate dendritic spine density in hippocampal pyramidal cells in adulthood. J Neurosci 10(4):1286–1291. https://doi.org/10.1523/JNEUROSCI.10-04-01286.1990
Hammes SR, Levin ER (2007) Extranuclear steroid receptors: nature and actions. Endocr Rev 28(7):726–741. https://doi.org/10.1210/er.2007-0022
Hannan JL, Albersen M, Stopak BL, Liu X, Burnett AL, Hoke A, Bivalacqua TJ (2015) Temporal changes in neurotrophic factors and neurite outgrowth in the major pelvic ganglion following cavernous nerve injury. J Neurosci Res 93(6):954–963. https://doi.org/10.1002/jnr.23553
Hasan W, Smith HJ, Ting AY, Smith PG (2005) Estrogen alters trkA and p75 neurotrophin receptor expression within sympathetic neurons. J Neurobiol 65(2):192–204. https://doi.org/10.1002/neu.20183
Johann S, Beyer C (2013) Neuroprotection by gonadal steroid hormones in acute brain damage requires cooperation with astroglia and microglia. J Steroid Biochem Mol Biol 137:71–81. https://doi.org/10.1016/j.jsbmb.2012.11.006
Kajta, M, Trotter, A, Lasoń, W, Beyer, C (2006). Impact of 17β-estradiol on cytokine-mediated apoptotic effects in primary hippocampal and neocortical cell cultures. Brain Res 1116(1), 64–74. https://doi.org/10.1016/j.brainres.2006.07.105
Karki P, Smith K, Johnson J Jr, Lee E (2014) Astrocyte-derived growth factors and estrogen neuroprotection: role of transforming growth factor-α in estrogen-induced upregulation of glutamate transporters in astrocytes. Mol Cell Endocrinol 389(1–2):58–64. https://doi.org/10.1016/j.mce.2014.01.010
Kwakowsky, A, Milne, MR, Waldvogel, HJ, Faull, RL (2016). Effect of Estradiol on Neurotrophin Receptors in Basal Forebrain Cholinergic Neurons: Relevance for Alzheimer’s Disease. Int J Mol Sci 17(12), 2122. https://doi.org/10.3390/ijms17122122
Labelle C, Leclerc N (2000) Exogenous BDNF, NT-3 and NT-4 differentially regulate neurite outgrowth in cultured hippocampal neurons. Brain Res Dev Brain Res 123(1):1–11. https://doi.org/10.1016/S0165-3806(00)00069-9
Lee SJ, McEwen BS (2001) Neurotrophic and neuroprotective actions of estrogens and their therapeutic implications. Annu Rev Pharmacol Toxicol 41(1):569–591. https://doi.org/10.1146/annurev.pharmtox.41.1.569
Levin ER (2005) Integration of the extranuclear and nuclear actions of estrogen. Mol Endocrinol 19(8):1951–1959. https://doi.org/10.1210/me.2004-0390
Lorenzo A, Diaz H, Carrer H, Caceres A (1992) Amygdala neurons in vitro: neurite growth and effects of estradiol. J Neurosci Res 33(3):418–435. https://doi.org/10.1002/jnr.490330308
Ma, Y, Guo, H, Zhang, L, Tao, L, Yin, A, Liu, Z, … Hou, W (2016). Estrogen replacement therapy-induced neuroprotection against brain ischemia-reperfusion injury involves the activation of astrocytes via estrogen receptor β. Sci Rep 6, 21467. https://doi.org/10.1038/srep21467
MacLusky NJ, Clark AS, Naftolin F, Goldman-Rakic PS (1987) Estrogen formation in the mammalian brain: possible role of aromatase in sexual differentiation of the hippocampus and neocortex. Steroids 50(4):459–474. https://doi.org/10.1016/0039-128X(87)90032-8
Matsumoto A, Arai Y (1981) Neuronal plasticity in the deafferented hypothalamic arcuate nucleus of adult female rats and its enhancement by treatment with estrogen. J Comp Neurol 197(2):197–205. https://doi.org/10.1002/cne.901970203
McEwen BS, Alves SE, Bulloch K, Weiland NG (1997) Ovarian steroids and the brain implications for cognition and aging. Neurology 48(5 Suppl 7):8S–15S. https://doi.org/10.1212/WNL.48.5_Suppl_7.8S
Miranda RC, Sohrabji F, Toran-Allerand D (1994) Interactions of estrogen with the neurotrophins and their receptors during neural development. Horm Behav 28(4):367–375. https://doi.org/10.1006/hbeh.1994.1033
Mitra, SW, Hoskin, E, Yudkovitz, J, Pear, L, Wilkinson, HA, Hayashi, S, … Schaeffer, JM (2003). Immunolocalization of estrogen receptor β in the mouse brain: comparison with estrogen receptor α. Endocrinology 144(5), 2055–2067. https://doi.org/10.1210/en.2002-221069
Nelson LR, Bulun SE (2001) Estrogen production and action. J Am Acad Dermatol 45(3):S116–S124. https://doi.org/10.1067/mjd.2001.117432
Register TC, Shively CA, Lewis CE (1998) Expression of estrogen receptor α and β transcripts in female monkey hippocampus and hypothalamus. Brain Res 788(1):320–322. https://doi.org/10.1016/S0006-8993(98)00036-5
Riss, TL, Moravec, RA, Niles, AL, Duellman, S, Benink, HA, Worzella, TJ, Minor, L (2016). Cell viability assays. https://www.ncbi.nlm.nih.gov/books/NBK144065/
Rocca WA, Grossardt BR, Shuster LT (2011) Oophorectomy, menopause, estrogen treatment, and cognitive aging: clinical evidence for a window of opportunity. Brain Res 1379:188–198. https://doi.org/10.1016/j.brainres.2010.10.031
Scharfman HE, MacLusky NJ (2006) The influence of gonadal hormones on neuronal excitability, seizures, and epilepsy in the female. Epilepsia 47(9):1423–1440. https://doi.org/10.1111/j.1528-1167.2006.00672.x
Sharma K, Singh J, Pillai PP, Frost EE (2015) Involvement of MeCP2 in regulation of myelin-related gene expression in cultured rat oligodendrocytes. J Mol Neurosci 57(2):176–184. https://doi.org/10.1007/s12031-015-0597-3
Shughrue PJ, Lane MV, Merchenthaler I (1997) Comparative distribution of estrogen receptor-α and-β mRNA in the rat central nervous system. J Comp Neurol 388(4):507–525. https://doi.org/10.1002/(SICI)1096-9861(19971201)388:4%3C507::AID-CNE1%3E3.0.CO;2-6
Singh M, Sétáló G, Guan X, Warren M, Toran-Allerand CD (1999) Estrogen-induced activation of mitogen-activated protein kinase in cerebral cortical explants: convergence of estrogen and neurotrophin signaling pathways. J Neurosci 19(4):1179–1188. https://doi.org/10.1523/JNEUROSCI.19-04-01179.1999
Sofroniew MV, Vinters HV (2010) Astrocytes: biology and pathology. Acta Neuropathol 119(1):7–35. https://doi.org/10.1007/s00401-009-0619-8
Sohrabji F, Lewis DK (2006) Estrogen–BDNF interactions: implications for neurodegenerative diseases. Front Neuroendocrinol 27(4):404–414. https://doi.org/10.1016/j.yfrne.2006.09.003
Sohrabji F, Miranda RC, Toran-Allerand CD (1994) Estrogen differentially regulates estrogen and nerve growth factor receptor mRNAs in adult sensory neurons. J Neurosci 14(2):459–471. https://doi.org/10.1523/JNEUROSCI.14-02-00459.1994
Solum DT, Handa RJ (2002) Estrogen regulates the development of brain-derived neurotrophic factor mRNA and protein in the rat hippocampus. J Neurosci 22(7):2650–2659. https://doi.org/10.1523/JNEUROSCI.22-07-02650.2002
Thomas P, Pang Y, Filardo E, Dong J (2005) Identity of an estrogen membrane receptor coupled to a G protein in human breast cancer cells. Endocrinology 146(2):624–632. https://doi.org/10.1210/en.2004-1064
TORAN-ALLERAND CD (2005) Estrogen and the brain: beyond ER-α, ER-β, and 17β-estradiol. Ann N Y Acad Sci 1052(1):136–144. https://doi.org/10.1196/annals.1347.009
Toran-Allerand CD, Miranda RC, Bentham W, Sohrabji F, Brown TJ, Hochberg RB, MacLusky n J (1992) Estrogen receptors colocalize with low-affinity nerve growth factor receptors in cholinergic neurons of the basal forebrain. Proc Natl Acad Sci U S A 89(10):4668–4672. https://doi.org/10.1073/pnas.89.10.4668
Toran-Allerand CD, Singh M, Sétáló G (1999) Novel mechanisms of estrogen action in the brain: new players in an old story. Front Neuroendocrinol 20(2):97–121. https://doi.org/10.1006/frne.1999.0177
Tripathi A, Parikh ZS, Vora P, Frost EE, Pillai PP (2017) pERK1/2 peripheral recruitment and filopodia protrusion augment oligodendrocyte progenitor cell migration: combined effects of PDGF-A and fibronectin. Cell Mol Neurobiol 37(2):183–194. https://doi.org/10.1007/s10571-016-0359-y
Tsiperson V, Huang Y, Bagayogo I, Song Y, VonDran MW, DiCicco-Bloom E, Dreyfus CF (2015) Brain-derived neurotrophic factor deficiency restricts proliferation of oligodendrocyte progenitors following cuprizone-induced demyelination. ASN Neuro 7(1):1759091414566878. https://doi.org/10.1177/1759091414566878
Vasudevan N, Pfaff DW (2007) Membrane-initiated actions of estrogens in neuroendocrinology: emerging principles. Endocr Rev 28(1):1–19. https://doi.org/10.1210/er.2005-0021
Woolley CS, Gould E, Frankfurt M, McEwen BS (1990) Naturally occurring fluctuation in dendritic spine density on adult hippocampal pyramidal neurons. J Neurosci 10(12):4035–4039. https://doi.org/10.1523/JNEUROSCI.10-12-04035.1990
Xiao J, Wong AW, Willingham MM, van den Buuse M, Kilpatrick TJ, Murray SS (2010) Brain-derived neurotrophic factor promotes central nervous system myelination via a direct effect upon oligodendrocytes. Neurosignals 18(3):186–202. https://doi.org/10.1159/000323170
Acknowledgments
The authors are highly thankful to the Animal House Facility, Department of Biochemistry, The Maharaja Sayajirao University of Baroda, Vadodara. Authors are also very thankful to DBT-ILSPARE Programme, for the Confocal microscope facility at Dr. Vikram Sarabhai Science Block, Faculty of Science, The Maharaja Sayajirao University of Baroda, Vadodara.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Animal ethical approval
All animal experiments were in compliance with the ethical committee approved by the Institutional Animal Ethical Committee, The Maharaja Sayajirao University of Baroda; ZD/02/2013, 2014, 2015.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Langhnoja, J.M., Buch, L.K. & Pillai, P.P. 17β-estradiol modulates NGF and BDNF expression through ERβ mediated ERK signaling in cortical astrocytes. Biologia 73, 907–915 (2018). https://doi.org/10.2478/s11756-018-0099-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-018-0099-1