Abstract
Effects of fusaproliferin (FUS) on membrane potential (E M), electrolyte leakage, enzymes activity and respiration of roots, were studied in two maize cultivars (Zea mays L.), differing in their susceptibility to this toxin. In short-term experiments (≤ 6 h), E M has been rapidly and significantly depolarized by FUS. The rapidity of E M depolarization in tolerant cv. Lucia was more expressive in comparison with susceptible cv. Pavla, but the extent of E M depolarization was higher in cv. Pavla. In both maize cultivars, higher depolarization of E M was registered in cells of root zone I. In long-term experiments after the first E M depolarization, which occurred during the first 6 h of FUS treatment, gradual depolarization continued up to 24 h and was represented not only by the active component (E P) but also by the passive component (E D) of E M. The decrease in E M and E D was followed by a loss of K+ ions from FUS treated roots of both cultivars. The leak of K+ ions from the root cells of both root zones as well as both maize cultivars increased with the time of FUS treatment and was significantly higher in susceptible cv. Pavla than in tolerant cv. Lucia. FUS treatment of maize roots resulted in a significant decrease of root respiration which was higher in susceptible cv. Pavla than in tolerant cv. Lucia.
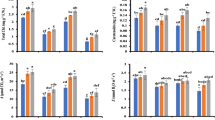
The analysis of enzyme activities revealed that FUS significantly stimulated POD activity in both maize cultivars. SOD activity was significantly increased only in susceptible cv. Pavla, while APX activity was not affected by the presence of FUS. GST activity was specifically induced by FUS only in tolerant cv. Lucia.
Due to the observed correlation between the extent of depolarization and the sensitivity of the studied maize cultivars to fusaproliferin, the E M parameters should be used for rapid screening of FUS-resistant cultivars for agricultural practice.
Similar content being viewed by others
Abbreviations
- APX:
-
ascorbate peroxidase
- FC:
-
fusicoccine
- FUS:
-
fusaproliferin
- GST:
-
glutathione-S-transferase
- PM:
-
plasma membrane
- POD:
-
guajacol peroxidase
- SOD:
-
superoxide dismutase
References
Atkin O.K., Edwards E.J. & Loveys B.R. 2000. Response of root respiration to changes in temperature and its relevance to global warming. New Phytol. 147: 141–154.
D’Alton A. & Etherton B. 1984. Effect of fusaric acid on tomato root hair membrane potentials and ATP levels. Plant Physiol. 74: 39–42.
DeGara L., de Pinto M.C. & Tommasi F. 2003. The antioxidant system vis-a-vis reactive oxygen species during plantpathogen interaction. Plan Physiol. Biochem. 41: 863–870.
Beyer W.F. & Fridovich I. 1987. Assaying for superoxide dismutase activity: some large consequence of minor changes in conditions. Anal. Biochem. 161: 559–566.
Bradford M.N. 1976. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Chance B. & Maehly A.C. 1955. Assay of catalases and peroxidases, pp. 764–775. In: Colowick P.S. & Kaplan N.O. (eds), Methods in Enzymology, Vol. 2, Academic Press, New York.
Chen G. & Asada K. 1989. Ascorbate peroxidase in tea leaves: occurrence of two isozymes and the differences in their enzymatic and molecular properties. Plant Cell Physiol. 30: 987–998.
Desjardins A.E. & Hohn T.M. 1997. Mycotoxins in plant pathogenesis. Molec. Plant-Microbe Interactions 10: 147–152.
Edwards R., Dixon D.P. & Walbot V. 2000. Plant glutathione-Stransferases: enzymes with multiple functions in sickness and in health. Trend Plant Sci. 5: 193–198.
Heiser I., Oßwald W. & Elstner E.F. 1998. The formation of reactive oxygen species by fungal and bacterial phytotoxins. Plant Physiol. Biochem. 36: 703–713.
Iannelli M.A., Pietrini F., Fiore L., Ppetrilli L. & Massacci A. 2002. Antioxidant response to cadmium in Phragmites australis plants. Plant Physiol. Biochem. 40: 977–982.
Köhler K. & Bentrup F.W. 1983. The effect of fusaric acid upon electrical membrane properties and ATP level in photoautotropic cell suspension cultures of Chenopodium rubrum L. Zeitschr. Pflanz. Physiol. 109: 355–361.
Gäumann E. 1957. Fusaric acid as a wilt toxin. Phytopathology 47: 342–357.
Lamprecht S.C., Marasas W.F.O., Alberts J.F., Cawood M., Gelderblom W.C.A. & Shepard G.S. 1994. Phytotoxicity of fumosins and AAL-Toxin to corn ant tomato. Phytopathology 84: 383–391.
Lemmens M., Joseph R., Schuhmacher R., Grausgruber H., Buerstmayr H., Ruckenbauer P., Neuhold G., Fidesser M. & Krska R. 1997. Head blight (Fusarium spp.) on wheat: investigation on the relationship between disease symptoms and mycotoxin content. Cereal Res. Comm. 25: 459–465.
Logrieco A., Moretti A., Fornelli F., Fogliano V., Ritieni A., Caiaffa M., Randazzo G., Bottalico A. & Macchia L. 1996. Fusaproliferin production by Fusarium subglutinans and its toxicity to Artemia salina, SF - 9 insect cells, and IARC/LCL 171 human B-lymphocytes. Appl. Environ. Microbiol. 62: 3378–3784.
Marrè E. 1979. Fusicoccin: a tool in plant physiology. Annu. Rev. Plant Physiol. 30: 273–288.
Marrè M.T., Vergani P. & Albergoni F.C. 1993. Relationship between fusaric acid uptake and its binding to cell structures by leaves of Egeria densa and its toxic effects on membrane permeability and respiration. Physiol. Mol. Plant Pathol. 42: 141–157.
Mauch F. & Dudler R. 1993. Differential induction of distinct glutathione-S-transferases of wheat by xenobiotics and by pathogen attack. Plant Physiol. 102: 1193–1201.
Miller J.D. & Young J.C. 1985. Deoxynivalenol in an experimental Fusarium graminearum infection of wheat. Can. J. Plant Path. 7: 132–134.
Nadubinská M., Ritieni A., Moretti A. & Šrobáro’a, A. 2003. Chlorophyll content in maize plants after treatment with fusariotoxins. Biologia 58: 115–119.
Neuhold G., Fidesser M. & Krska R. 1977. Head blight (Fusarium spp.) on wheat: investigation on the relationship between disease symptoms and mycotoxin content. Cereal Res. Comm. 25: 459–465.
Novacky A. 1980. Disease-related alteration in membrane function, pp. 369–380. In: Spanswick R.M., Lucas W.J. & Dainty J. (eds), Plant membrane transport. Current concept issues Elsevier/North Holland Biomedical Press.
Paciolla C., Dipierro N., Mulè G., Logrieco A. & Dipierro S. 2004. The mycotoxins beauvericin and T-2 induce cell deth and alteration to the ascorbate metabolism in tomato protoplasts. Physiol. Mol. Plant Pathol. 65: 49–56.
Pavlovkin J., Mistrík I., Zajchenko A.M. & Dugovič L. 1993. Some aspects of the phytotoxic action of trichothecene mycotoxin - roridin H on corn roots. Biologia 48: 435–439.
Pavlovkin J., Mistrík I. & Prokop M. 2004. Some aspects of the phytotoxic action of fusaric acid on primary Ricinus roots. Plant Soil Environ. 50: 397–401.
Pavlovkin J., Mistríková I., Luxová M. & Mistrík I. 2006. Effect of beauvericin on root transmembrane electric potential, electrolyte leakage and respiration of maize roots with dofferent susceptibility to Fusarium. Plant Soil Environ. 52: 492–498.
Pavlovkin J., Novacky A. & Ullrich-Eberius C.I. 1986. Membrane potential changes during bacteria-induced hypersensitive reaction. Physiol. Mol. Plant Pathol. 28: 125–135.
Placinta C. M., D’Mello J. P. F. & Macdonald A. M. C. 1999. A rewiew of worldwide contamination of cereal grains and animal feed with Fusarium mycotoxins. Feed Sci. Technol. 78: 21–37.
Ritieni A., Fogliano V., Randazzo G., Scarallo A., Logrieco A. & Moretti A. 1995. Isolation and characterisation of fusaproliferin, a new toxic metabolite from Fusarium proliferatum. Natural toxin 3: 17–20.
Ritieni A., Monti S.M., Randazzo G., Logrieco A., Meretti A. & Peluso G. 1997. Teratogenic effects of fusaproliferin on chicken. J. Agric. Food Chem. 45: 3039–3043.
Santini A., Šrobárová, A., Pavlovkin J., Čiamporová, M. & Ritieni A. 2008. Fusaproliferin effects on the photosystem in the cells of maize seedlings leaves. Eur. J. Plant Pathol. 120: 363–371.
Scott-Craig J.S., Kerby K.B., Stein B.D. & Somerville S.C. 1995. Expression of an extracellular peroxidase that is induced in barley (Hordeum vulgare) by powdery mildew pathogen (Erysiphe graminis f. sp. hordei). Physiol. Mol. Plant Pathol. 47: 407–418.
Šliková, S., Šrobárová, A., Šudyová, V., Polišenská, I., Gregová E. & Mihálik D. 2010. Response of oat cultivars to Fusarium infection with a view to their suitability for food use. Biologia 65: 609–614.
Snijders C.H.A. & Perkowski J. 1990. Effect of head blight caused by Fusarium culmorum on toxin content and weight of wheat kernels. Phytopathology 80: 566–570.
Šrobárová, A., Nadubinská M. & Čiamporoá, M. 2004. Relative efficacy of fusariotoxins on young maize plants. Cereal Res. Comm. 2: 241–248.
Šrobároá, A., Teixiera da Silva J. A., Kogan G., Ritieni A. & Santini A. 2009. Beauvericin dcreases cell viability of wheat. Chem. Biodiver. 6: 1208–1215.
Tamás, L., Dudíková, J., Ďurčeková, K., Huttová, J., Mistrík, I., & Zelinová V. 2008. The impact of heavy metals on the activity of some enzymes along the barley root. Env. Exp. Bot. 62: 86–91.
van Loon L.C. 1985. Pathogenesis-related proteins. Plant Mol. Biol. 4: 111–116.
Vianello M. & Macri F. 1978a. Inhibition of plant cell membrane transport phenomena induced by zearalenone. Planta 143: 51–57.
Vianello M. & Macri F. 1978b. Effect of zearalenone (F-2) on pea steam, maize root and rat liver mitochondria. Planta 153: 443–446.
Wang Y.Z. & Miller J.D. 1988. Effect of Fusarium graminearum metabolites on wheat tissue in relation to Fusarium head blight resistance. J. Phytopathol. 122: 118–125.
Ziegler W., Pavlovkin J., Raňansky J. & Zajchenko A.M. 1994. Ion channels induced by some mycotoxins in lipid bilayer membranes. Biologia 49: 855–862.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pavlovkin, J., Jašková, K., Mistríková, I. et al. Impact of fusaproliferin on primary roots of maize cultivars differing in their susceptibility to Fusarium . Biologia 66, 1044–1051 (2011). https://doi.org/10.2478/s11756-011-0114-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-011-0114-2