Abstract
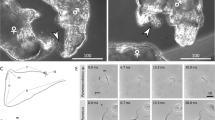
Sperm apical hooks in murine rodents play an important role in sperm competition. Apical hooks are more curved and longer in species with relatively larger testes, that is in species with a higher risk of sperm competition. The sperm can form aggregations, ‘trains’, that can move faster than individual sperm, thus reaching the egg earlier as was observed in Apodemus sylvaticus. The apical hook plays an important role for train formation. This study focuses on the changes in the curvature of sperm apical hooks during the final stages of spermiogenesis and stages before fertilization (sperm-life span). Apical hook curvatures of field mice (A. agrarius and A. sylvaticus) vary significantly between dormant and active sperm. In contrast, there are no significant differences among the stages in the eastern house mouse. Since there are high ranges of angle values in all stages, the mean angles of apical hook curvature are not appropriate for evaluating risk of sperm competiton. The ranges of angle values point to a level of flexibility of the apical hooks. The lengths of sperm hooks in individual species do not change during particular stages. The length and flexibility of the sperm apical hooks are important for the formation of sperm aggregations, thus these sperm characters indicate the risk of sperm competition and the sperm strategies in murine rodents.
Similar content being viewed by others
References
Breed W.G. 2004. The spermatozoon of Eurasian murine rodents: Its morphological diversity and evolution. J. Morphol. 261(1): 52–69. DOI: 10.1002/jmor.10228
Breed W.G. 2005. Evolution of the spermatozoon in muroid rodents. J. Morphol. 265(3): 271–290. DOI: 10.1002/jmor. 10357
Breed W.G. & Taylor J. 2000. Body mass, testes mass, and sperm size in murine rodents. J. Mammal. 81(3): 758–768. DOI: 10.1644/1545-1542(2000)081〈0758:BMTMAS〉2.3.CO;2
Bryja J., Patzenhauerová H., Albrecht T., Mošanský M. & Stopka P. 2008. Varying levels of female promiscuity in four Apodemus mice species. Behav. Ecol. Sociobiol. 63: 251–260. DOI: 10.1007/s00265-008-0656-7
Bryja J. & Stopka P. 2005. Facultative promiscuity in a presumably monogamous mouse Apodemus microps. Acta Theriologica 50(2): 189–196. DOI: 10.1007/BF03194482
Dean M.D., Ardlie K.G. & Nachman M.W. 2006. The frequency of multiple paternity suggests that sperm competition is common in house mice (Mus domesticus). Molec. Ecol. 15: 4141–4151. DOI: 10.1111/j.1365-294X.2006.03068.x
delBarco-Trillo J. & Ferkin M.H. 2004. Male mammals respond to a risk of sperm competition conveyed by odours of conspecific males. Nature 431: 446–449. DOI:10.1038/nature02845
Dvořáková K., Šandera M., Jursová M., Vašinová Pěknicová J. 2008. The influence of fluorides on mouse sperm capacitation. Anim. Reprod. Sci. 108: 157–170. DOI: 10.1016/j.anireprosci.2007.07.015
Firman R.C. & Simmons L.W. 2008. The frequency of multiple paternity predicts variation in testes size among island populations of house mice. J. Evol. Biol. 21(6): 1524–1533. DOI: 10.1111/j.1420-9101.2008.01612.x
Frynta D., Slábová M. & Vohralík V. 2009. Why do male house mice have such small testes? Zool. Sci. 26(1): 17–23. DOI: 10.2108/zsj.26.17
Harcourt A.H., Harvey P.H., Larson S.G. & Short R.V. 1981. Testes weight, body weight and breeding system in primates. Nature 293: 55–57. DOI:10.1038/293055a0
Kenagy G.J. & Trombulak S.G. 1986. Size and function of mammalian testes in relation to body size. J. Mammal. 67(1): 1–22.
Immler S. 2008. Sperm competition and sperm cooperation: the potential role of diploid and haploid expression. Reproduction 135: 275–283. DOI: 10.1530/REP-07-0482
Immler S., Moore H.D.M., Breed W.G. & Birkhead T.R. 2007. By hook or by crook? Morphometry, competition and cooperation in rodent sperm. PloS ONE. 2(1): e170. DOI:10.1371/journal.pone.0000170
Moore H., Dvořáková K., Jenkins N. & Breed W. 2002. Exceptional sperm cooperation in the wood mouse. Nature 418: 174–177. DOI:10.1038/nature00832
Ramm S.A. & Stockley P. 2007. Ejaculate allocation under varying sperm competition risk in the house mouse, Mus musculus domesticus. Behav. Ecol. 18: 491–495. DOI: 10.1093/beheco/arm003
Ramm S.A. & Stockley P. 2009. Adaptive plasticity of mammalian sperm production in response to social experience. Proc. Roy. Soc. B Biol. Sci. 276: 745–751. DOI: 10.1098/rspb.2008.1296
Roldan E.R.S., Gomendio M. & Vitullo A.D. 1992. The evolution of eutherian spermatozoa and underlying selective forces: female selection and sperm competition. Biol. Rev. 67(4): 551–593. DOI: 10.1111/j.1469-185X.1992.tb01193.x
Stopka P. & Macdonald D.W. 1998. Signal interchange during mating in the wood mouse (Apodemus sylvaticus): the concept of active and passive signalling. Behavior 135(2): 231–249.
Stopka P. & Graciasová R. 2001. Conditional allogrooming in the herb-field mouse. Behav. Ecol. 12(5): 584–589. DOI:10.1093/beheco/12.5.584
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Šandera, M., Andrlíková, P., Frolíková, M. et al. Changes in the curvature of sperm apical hooks in murine rodents. Biologia 66, 916–921 (2011). https://doi.org/10.2478/s11756-011-0095-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-011-0095-1