Abstract
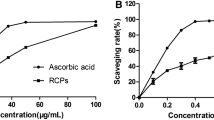
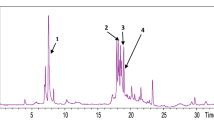
Previous work revealed that the defatted methanol (MeOH) extract of fruits of Conocarpus erectus L. (Combretaceae family) exhibited antioxidant, antibacterial and anti-cancer activities. In further studies of this valuable plant, the defatted MeOH extract of C. erectus fruits was subjected to chromatographic fractionation in a silica gel glass column followed by reversed-phase high-performance liquid chromatography-ultraviolet-electrospray ionisation spectrometry analysis (RP-HPLC-UV-ESI-MS). The major and sharp peaks in each sample were identified or tentatively identified based on matching with some standard compounds and a review of the literature. Ellagic acid, vescalagin/castalagin isomer and di-(hexahydroxy diphenoyl) galloyl hexose isomer were tentatively identified as major components with many hydrolysable types of tannins on the basis of a comparison of its mass patterns with relevant items in the literature. Gallic acid, kaempferol 3-O-β-d-glucopyranoside and quercetin 3-O-β-d-glucopyranoside were identified on the basis of matching retention time (t R ) and mass spectra with the standards. Polymethoxylated flavonoid isomers were also tentatively identified. The antioxidant properties of all samples were found to be associated with the total content of phenolic compounds. This may be considered as the first detailed phytochemical report in identifying the phytochemicals in C. erectus fruits. Due to the high antioxidant activity exhibited by both the crude MeOH extract and its fractions, it could be used as an effective natural antioxidant after further in vitro and in vivo studies.
Similar content being viewed by others
References
Abad-García, B., Berrueta, L. A., Garmón-Lobato, S., Gallo, B., & Vicente, F. (2009). A general analytical strategy for the characterization of phenolic compounds in fruit juices by high-performance liquid chromatography with diode array detection coupled to electrospray ionization and triple quadrupole mass spectrometry. Journal of Chromatography A, 1216, 5398–5415. DOI: 10.1016/j.chroma.2009.05.039.
Abdel-Hameed, E. S. S., Bazaid, S. A., Shohayeb, M. M., El-Sayed, M. M., & El-Wakil, E. A. (2012). Phytochemical studies and evaluation of antioxidant, anticancer and antimicrobial properties of Conocarpus erectus L. growing in Taif, Saudi Arabia. European Journal of Medicinal Plants, 2, 93–112.
Admiczeski, M., Ni, J. X., Jaber, H., Huang, J. S., Kang, R., & Nakatsu, T. (1992). A novel hydrolyzable tannin and related compounds isolated from the leaf surface of Chrysolepis sempervirens. Journal of Natural Products, 55, 521–524. DOI: 10.1021/np50082a024.
Adonizio, A. L. (2008). Anti-quorum sensing agents from south florida medicinal plants and their attenuation of Pseudomonas aeruginosa pathogenicity. Ph.D. thesis, Florida International University, Miami, FL, USA.
Barbosa, W. L. R., Peres, A., Gallori, S., & Vincieri, F. F. (2006). Determination of myricetin derivatives in Chrysobalanus icaco L. (Chrysobalanaceae). Revista Brasileira de Farmacognosia, 16, 333–337. DOI: 10.1590/s0102-695x2006000300009.
Barry, K. M., Davies, N. W., & Mohammed, C. L. (2001). Identification of hydrolysable tannins in the reaction zone of Eucalyptus nitens wood by high performance liquid chromatography-electrospray ionisation mass spectrometry. Phytochemical Analysis, 12, 120–127. DOI: 10.1002/pca.548.
Del Bubba, M., Checchini, L., Chiuminatto, U., Doumett, S., Fibbi, D., & Giordani, E. (2012). Liquid chromatographic/electrospray ionization tandem mass spectrometric study of polyphenolic composition of four cultivars of Fragaria vesca L. berries and their comparative evaluation. Journal of Mass Spectrometry, 47, 1207–1220. DOI: 10.1002/jms.3030.
Fernandes, A., Sousa, A., Mateus, N., Cabral, M., & de Freitas, V. (2011). Analysis of phenolic compounds in cork from Quercus suber L. by HPLC-DAD/ESI-MS. Food Chemistry, 125, 1398–1405. DOI: 10.1016/j.foodchem.2010.10.016.
Fischer, U. A., Carle, R., & Kammerer, D. R. (2011). Identification and quantification of phenolic compounds from pomegranate (Punica granatum L.) peel, mesocarp, aril and differently produced juices by HPLC-DAD-ESI/MSn. Food Chemistry, 127, 807–821. DOI 10.1016/j.foodchem.2010.12.156.
Hanhineva, K., Rogachev, I., Kokko, H., Mintz-Oron, S., Venger, I., Kärenlampi, S., & Aharoni, A. (2008). Nontargeted analysis of spatial metabolite composition in strawberry (Fragaria × ananassa) flowers. Phytochemistry, 69, 2463–2481. DOI: 10.1016/j.phytochem.2008.07.009.
Kassima, M., Achoui, M., Mustafa, M. R., Mohd, M. A., & Yusoff, K. M. (2010). Ellagic acid, phenolic acids and flavonoids in Malaysian honey extracts demonstrate in vitro anti-inflammatory activity. Nutrition Research, 30, 650–659. DOI: 10.1016/j.nutres.2010.08.008.
Liu, G. Z., Ma, J. Y., Chen, Y. Z., Tian, Q. Q., Shen, Y., Wang, X. S., Chen, B., & Yao, S. Z. (2009). Investigation of flavonoid profile of Scutellaria bacalensis Georgi by high performance liquid chromatography with diode array detection and electrospray ion trap mass spectrometry. Journal of Chromatography A, 1216, 4809–4814. DOI: 10.1016/j.chroma.2009.04.021.
Mämmel Vartianen, T. (2000). Analysis of oak tannins by liquid chromatography-electrospray ionisation mass spectrometry. Journal of Chromatography A, 891, 75–83. DOI: 10.1016/s0021-9673(00)00624-5.
Nahla, A. A. (2010). A trimethoxyellagic acid glucuronide from Conocarpus erectus leaves: Isolation, characterization and assay of antioxidant capacity. Pharmaceutical Biology, 48, 328–332. DOI: 10.3109/13880200903131567.
Ncube, N. S., Afolayan, A. J., & Okoh, A. I. (2008). Assessment techniques of antimicrobial properties of natural compounds of plant origin: Current methods and future trends. African Journal of Biotechnology, 7, 1797–1806. DOI: 10.5897/ajb07.613.
Newman, D. J., Cragg, G. M., & Snader, K. M. (2003). Natural products as sources of new drugs over the period 1981–2002. Journal of Natural Products, 66, 1022–1037. DOI: 10.1021/np030096l.
Pfundstein, B., El Desouky, S. K., Hull, W. E., Haubner, R., Erben, G., & Owen, R. W. (2010). Polyphenolic compounds in the fruits of Egyptian medicinal plants (Terminalia bellerica, Terminalia chebula and Terminalia horrida): Characterization, quantitation and determination of antioxidant capacities. Phytochemistry, 71, 1132–1148. DOI: 10.1016/j.phytochem.2010.03.018.
Puech, J. L., Mertz, C., Michon, V., Le Guernevé, C., Doco, T., & du Penhoat, C. H. (1999). Evolution of castalagin and vescalagin in ethanol solutions. Identification of new derivatives. Journal of Agricultural and Food Chemistry, 47, 2060–2066. DOI: 10.1021/jf9813586.
Regazzoni, L., Arlandini, E., Garzon, D., Santagati, N. A., Beretta, G., & Facino, R. M. (2013). A rapid profiling of gallotannins and flavonoids of the aqueous extract of Rhus coriaria L. by flow injection analysis with high-resolution mass spectrometry assisted with database searching. Journal of Pharmaceutical and Biomedical Analysis, 72, 202–207. DOI: 10.1016/j.jpba.2012.08.017.
Romani, A., Campo, M., & Pinelli, P. (2012). HPLC/DAD/ESIMS analyses and anti-radical activity of hydrolyzable tannins from different vegetal species. Food Chemistry, 130, 214–221. DOI: 10.1016/j.foodchem.2011.07.009.
Scordino, M., Sabatino, L., Traulo, P., Gargano, M., Pantó, V., & Gagliano, G. (2011). HPLC-PDA/ESI-MS/MS detection of polymethoxylated flavones in highly degraded citrus juice: A quality control case study. European Food Research and Technology, 232, 275–280. DOI: 10.1007/s00217-010-1386-4.
Seeram, N. P., Lee, R., Scheuller, H. S., & Heber, D. (2006). Identification of phenolic compounds in strawberries by liquid chromatography electrospray ionization mass spectroscopy. Food Chemistry, 97, 1–11. DOI: 10.1016/j.foodchem.2005.02.047.
Sudjaroen, Y., Hull, W. E., Erben, G., Würtele, G., Changbumrung, S., Ulrich, C. M., & Owen, R. W. (2012). Isolation and characterization of ellagitannins as the major polyphenolic components of Longan (Dimocarpus longan Lour) seeds. Phytochemistry, 77, 226–237. DOI: 10.1016/j.phytochem.2011.12.008.
Tsuda, T., Watanabe, M., Ohshima, K., Yamamoto, A., Kawakishi, S., & Osawa, T. (1994). Antioxidative components isolated from the seed of Tamarind (Tamarindus indica L.). Journal of Agricultural and Food Chemistry, 42, 2671–2674. DOI: 10.1021/jf00048a004.
Vivas, N., Glories, Y., Bourgeois, G., & Vitry, C. (1996). The heartwood ellagitannins of different oak (Quercus sp.) and chestnut species (Castanea sativa Mill.). Quantity analysis of red wines aging in barrels. Journal des Sciences et Techniques de la Tonnellerie, 2, 51–75.
Zhang, J. Y., Li, N., Che, Y. Y., Zhang, Y., Liang, S. X., Zhao, M. B., Jiang, Y., & Tu, P. F. (2011). Characterization of seventy polymethoxylated flavonoids (PMFs) in the leaves of Murraya paniculata by on-line high-performance liquid chromatography coupled to photodiode array detection and electrospray tandem mass spectrometry. Journal of Pharmaceutical and Biomedical Analysis, 56, 950–961. DOI: 10.1016/j.jpba.2011.08.019.
Zhu, Z. Y., Zhang, H., Zhao, L., Dong, X., Li, X., Chai, Y. F., & Zhang, G. Q. (2007). Rapid separation and identification of phenolic and diterpenoid constituents from radix Salvia miltiorrhizae by high-performance liquid chromatography diodearray detection, electrospray ionization time-of-flight mass spectrometry and electrospray ionization quadrupole ion trap mass spectrometry. Rapid Communication of Mass Spectrometry, 21, 1855–1865. DOI: 10.1002/rcm.3023.
Zywicki, B., Reemtsma, T., & Jekel, M. (2002). Analysis of commercial vegetable tanning agents by reversed-phase liquid chromatography-electrospray ionization-tandem mass spectrometry and its application to wastewater. Journal of Chromatography A, 970, 191–200. DOI: 10.1016/s0021-9673(02)00883-x.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Abdel-Hameed, ES.S., Bazaid, S.A. & Shohayeb, M.M. RP-HPLC-UV-ESI-MS phytochemical analysis of fruits of Conocarpus erectus L.. Chem. Pap. 68, 1358–1367 (2014). https://doi.org/10.2478/s11696-014-0570-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-014-0570-6