Abstract
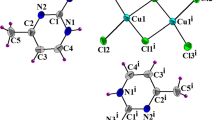
Gaussian analysis of bands between 500 cm−1 and 600 cm−1 attributed to the \( \tilde \nu \) as (Cu4O) stretching vibration of the tetrahedral Cu4O core in Cu4OBr n Cl(6−n)(4-Mepy)4 (n = 0–6) complexes showed two bands, symmetry reduction of the T 2 mode of vibration towards the A 1 and E modes and vibrational coupling with an R-sensitive in-plane pyridine ring bending. The Cu-O bond is considered as vibrationally coupled with the Cu-N bond and the pyridine ring through the donor-acceptor vibrational coupling and the corresponding equilibrium charge distribution. The linear correlation between \( \tilde \nu \) as(Cu4O) and the number of bromides in Cu4OBr n Cl(6−n)(4-Mepy)4 complexes was used for the estimation of partial charges on the 4-Mepy ligands which were positive for the prevailing donors and negative for the prevailing acceptors thus evoking a π-back bonding between the Cu(II) atoms and the 4-Mepy ligands. Correlations involving selected bond lengths and bond angles in the molecular structure of the Cu4OCl6(4-Mepy)4 complex with four symmetrically independent molecules present in the unit cell indicate a symmetry reduction of the T 2 mode of vibration and the π-back bonding between the Cu(II) atoms and the 4-Mepy ligands.
Similar content being viewed by others
References
Anson, C. E., arapKoske, S. K., Jayasooriya, U. A., & Cannon, R. D. (1992). FT-IR spectroscopy: a sensitive probe of concealed symmetry-lowering in tetranuclear copper(II) complexes. Spectrochimica Acta Part A: Molecular Spectroscopy, 48A, 151–154. DOI: 10.1016/0584-8539(92)80017-Q.
Berkesi, O., Andor J. A., Jayasooriya, U. A., & Cannon, R. D. (1992). Vibrations of central oxygen in a high-symmetry tetranuclear metal cluster: a definitive assignment using isotopic substitution in a basic zinc carboxylate complex. Spectrochimica Acta Part A: Molecular Spectroscopy, 48A, 147–149. DOI: 10.1016/0584-8539(92)80016-P.
Bratsch, S. G. (1988). Revised Mulliken electronegativities. II. Applications and limitations. Journal of Chemical Education, 65, 223–227.
Fermi, E. (1931). Über den Ramaneffekt des Kohlendioxyds. Zeitschrift für Physik, 71, 250–259. DOI: 10.1007/BF01341712.
Frank, C. W., & Rogers L. B. (1966). Infrared spectral study of metal-pyridine, substituted pyridine, and quinoline complexes in the 667–150 cm−1 region. Inorganic Chemistry, 5, 615–622. DOI: 10.1021/ic50038a026.
Gill, N. S., & Sterns, M. (1970). The preparation and properties of µ4-oxo-hexa-µ-chloro-tetrakis[(2-methylpyridine)copper(II)] hydrate, Cu4OCl6(2-mepy)4. xH2O, and di-µ-methoxobis[chloro(2-methylpyridine)copper(II)], [CuCl(OCH3)(2-mepy)]2, and X-ray structure analysis of Cu4OCl6(2-mepy)4. xH2O. Inorganic Chemistry, 9, 1619–1625. DOI: 10.1021/ic50089a004.
Ito, T., Hamaguchi, T., Nagino, H., Yamaguchi, T., Washington, J., & Kubiak, C. P. (1997). Effects of rapid intramolecular electron transfer on vibrational spectra. Science, 277, 660–663. DOI: 10.1126/science.277.5326.660.
Jorík, V., Koman, M., Makáňová, D., Mikloš, D., Broškovičová, A., & Ondrejovič, G. (1996). Fine stereochemical effects in tetranuclear mixed-halide copper(II) complexes: The structure of Cu4OBr3Cl3(OPPh3)4. Polyhedron, 15, 3129–3137. DOI: 10.1016/0277-5387(96)00013-7.
Kilbourn, B. T., & Dunitz, J. D. (1967). The crystal and molecular structure of µ4-oxohexa-µ-chlorotetrakis(pyridine copper(II)), Cu4Cl6O.4C5H5N, a polynuclear copper complex. Inorganica Chimica Acta, 1, 209–216. DOI: 10.1016/S0020-1693(00)93172-4.
Lazrek, M., Bicout, D. J., Jaziri, S., & Kats, E. (2005). Vibrational enhancement of the effective donor-acceptor coupling. JETP Letters, 82, 366–370. DOI: 10.1134/1.2137374.
Norman, R. E., Rose, N. J., & Stenkamp, R. E. (1989). Simple, direct synthesis and structure of hexa-µ-chloro-tetrakis(1-methylimidazole)-µ4-oxo-tetracopper(II). Acta Crystallographica, C45, 1707–1713. DOI: 0108-2701/89/111707.
Ondrejovič, G., Koman, M., & Kotočová, A. (2008). Structural and electronic effects involving pyridine rings in 4-methylpyridine Cu4OX6L4 complexes. II. Correlations based on molecular structure of Cu4OCl6(4-Mepy)4 complex. Chemical Papers, in press.
Ondrejovič, G., & Kotočová, A. (2001). Donor-acceptor behavior of ligands in Cu4OX6L4 complexes as indicated by infrared spectra. Chemical Papers, 55, 221–228.
Ondrejovič, G., & Kotočová, A. (2005). Spectral and electrochemical study of coordination molecules Cu4OX6L4: Cu4OBrnCl(6−n)(Pyridine)4 complexes. Chemical Papers, 59, 166–173.
Ondrejovič, G., & Kotočová, A. (2006). Spectral and electrochemical study of coordination molecules Cu4OX6L4: 3-Methylpyridine and 4-methylpyridine Cu4OBrnCl(6−n)L4 complexes. Chemical Papers, 60, 10–21. DOI: 10.2478/s11696-006-0003-2.
Person, W. B. (1973). Theoretical aspects of the study of molecular complexes. In J. Yarwood (Ed.), Spectroscopy and structure of molecular complexes (pp. 50–70). London: Plenum Press.
Sanderson, R. T. (1983). Polar covalence. New York: Academic Press.
Swank, D. D., Nielson, D. O., & Wilett, R. D. (1973). The crystal structure of µ4-oxo-hexa-µ-bromotetrakis[pyridine copper(II)], Cu4Br6O(C5H5N)4. Inorganica Chimica Acta, 7, 91–96. DOI: 10.1016/S0020-1693(00)94786-8.
tom Dieck, H. (1973). Tetranuclear complexes of trigonalbipyramidal copper(II). III. Electronic and infrared spectra. Inorganica Chimica Acta, 7, 397–403. DOI: 10.1016/S0020-1693(00)94852-7.
Wong, P. T. T., & Brewer, D. G. (1968). Nature of the coordination bond in metal complexes of substituted pyridine derivatives. II. The far infrared spectra and metal-ligand force constants of copper complexes of 4-substituted pyridines. Canadian Journal of Chemistry, 46, 139–148. DOI: 10.1139/v68-025.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ondrejovič, G., Koman, M. & Kotočová, A. Structural and electronic effects involving pyridine rings in 4-methylpyridine Cu4OX6L4 complexes. I. Vibrational spectra of Cu4OBr n Cl(6−n)(4-Mepy)4 complexes. Chem. Pap. 62, 480–486 (2008). https://doi.org/10.2478/s11696-008-0055-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-008-0055-6