Abstract
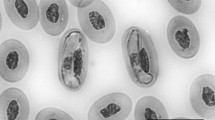
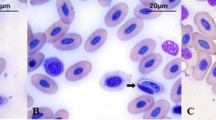
Hepatozoon garnhami n. comb. was redescribed from Schokari sand snakes (Psammophis schokari) collected from Riyadh city in Saudi Arabia. Gametocytes were found in the peripheral blood of 2 of 15 snakes examined. Based on the similar morphological and morphometric characteristics, the same host and a similar host habitat environment, it can be concluded for the first time that the present species is conspecific with Haemogregarina garnhami previously reported from Psammophis shokari aegyptius. To further characterize this parasite, the partial 18S rRNA gene was amplified and sequenced. The sequence analysis also showed that Haemogregarina garnhami should be reassigned into the genus Hepatozoon as Hepatozoon garnhami which has 99.5% (859/863 bp) sequence similarity to Hepatozoon ayorgbor, infecting the erythrocytes of Python regius in Ghana. Phylogenetic analysis showed that H. garnhami formed a mixed clade with Hepatozoon spp. from geckos, snakes and rodents and ophidian Hepatozoon spp. did not form a separated phylogenetic unit. Also, Psammophis schokari-infecting Hepatozoon contained several different genetic lineages. To our knowledge, the present work extends the geographic distribution of H. garnhami and is the first report of Hepatozoon infection in snakes from Saudi Arabia.
Similar content being viewed by others
References
Abdel-Baki A.S., Al-Quraishy S. 2012. Morphological characteristics of a new species of Haemogregarina Danilewsky, 1885 (Apicomplexa: Adeleorina) in naturally infected Acanthodactylus boskianus (Daudin) (Sauria: Lacertidae) in Egypt. Systematic Parasitology, 82, 65–69. DOI: 10.1007/s11230-012-9347-x.
Abdel-Haleem H.M., Al-Quraishy S., Abdel-Baki A.S. 2013. A redescription of Haemogregarina damiettae Ramadan et al. 1996 naturally infecting the Acanthodactylus boskianus from Egypt, with new merognic data. Parasitolology Research, 112, 2045–2048. DOI: 10.1007/s00436-013-3364-9.
Al-Farraj S. 2008. Light and electron microscopic study on a haemogregarine species infecting the viper Cerastes cerastes gasperitti from Saudi Arabia. Pakistan Journal of Biological Sciences, 11, 1414–1421. DOI: 10.3923/pjbs.2008.1414.1421.
Barta J.R., Ogedengbe J.D., Martin D.S., Smith T.G. 2012. Phylogenetic position of the Adeleorinid Coccidia (Myzozoa, Apicomplexa, Coccidia, Eucoccidiorida, Adeleorina) inferred using 18S rDNA sequence. Journal of Eukaryotic Microbiology, 59, 171–180. DOI: 10.1111/j.1550-7408.2011.00607.x.
Boulianne B., Evans R.C., Smith T.G. 2007. Phylogenetic analysis of Hepatozoon species (Apicomplexa: Adeleorina) infecting frogs of Nova Scotia, Canada, determined by ITS-1 sequences. Journal of Parasitology, 93, 1435–1441. DOI: 10.1645/GE-1041.1.
Guindon S., Dufayard J.F., Lefort V., Anisimova M., Hordijk W., Gascuel O. 2010. New Algorithms and Methods to Estimate Maximum-Likelihood Phylogenies: Assessing the Performance of PhyML 3.0. Systematic Biology, 59, 307–321. DOI: 10.1093/sysbio/syq010.
Harris D.J., Maia J.P.M.C., Perera A. 2011. Molecular characterization of Hepatozoon species in reptiles from the Seychelles. Journal of Parasitology, 97, 106–110. DOI: 10.1645/GE-2470.1.
Herbert J.D.K., Godfrey S.S., Bull C.M., Menz R.I. 2010. Developmental stages and molecular phylogeny of Hepatozoon tuatarae, a parasite infecting the NewZealand tuatara, Sphenodon punctatus and the tick, Amblyomma sphenodonti. International Journal of Parasitology, 40, 1311–1315. DOI: 10.1016/j.ijpara.2010.03.018.
Jakes K.A., O’Donoghue P.J., Cameron S.L. 2003. Phylogenetic relationships of Hepatozoon (Haemogregarina) boigae, Hepatozoon sp., Haemogregarina clelandi and Haemoproteus chelodina from Australian reptiles to other Apicomplexa based on cladistic analyses of ultrastructural and life-cycle characters. Parasitology, 126, 555–559. DOI: 10.1017/S0031182003003111.
Kimura M. 1980. A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. Journal of Molecular Evolution, 16, 111–120. DOI: 10.1007/BF01731581.
Levine N.D. 1988. The Protozoan Phylum Apicomplexa. Vol. II. CRC Press, Boca Raton, F. L. 224 pp.
Mackerras M.J. 1961. The haematozoa of Australian reptiles. Australian Journal of Zoology, 9, 61–122. DOI: 10.1071/ZO9610061.
Maia J.P.M.C., Harris D.J., Perera A. 2011. Molecular survey of Hepatozoon species in lizards from North Africa. Journal of Parasitology, 97, 513–517. DOI: 10.1645/GE-2666.1.
Maia J.P.M.C., Perera A., Harris D.J. 2012. Molecular survey and microscopic examination of Hepatozoon Miller, 1908 (Apicomplexa: Adeleorina) in lacertid lizards from the western Mediterranean. Folia Parasitologica, 59, 241–248. DOI: 10.2478/s11686-012-0048-Z.
Merino S., Vásquez R.A., Martínez J., Celis-Diez J.L., Gutieŕrezjimeńez L., Ippi S., Sánchez-Monsalvez I., Lapuente J.M. 2009. Molecular characterization of an ancient Hepatozoon species parasitizing the ‘living fossil’ marsupial ‘Monito delMonte’ Dromiciops gliroides from Chile. Biological Journal of the Linnean Society, 98, 568–576. DOI: 10.1111/j.1095-8312.2009.01302.x.
Morsy K., Bashtar A.R., Ghaffar F.A., Al Quraishy S., Al Hashimi S., Al Ghamdi A., Shazly M. 2013. Developmental stages of Hepatozoon seurati (Laveran and Pettit 1911) comb. nov., a parasite of the corned viper Cerastes cerastes and the mosquito Culex pipiens from Egypt. Parasitology Research, 112, 2533–2542. DOI: 10.1007/s00436-013-3420-5.
Ortuño A., Castella J., Criado-Fornelio A., Buling A., Barba-Carretero J.C. 2008. Molecular detection of a Hepatozoon species in stray cats from a feline colony in North-eastern Spain. Veterinary Journal, 177, 134–135. DOI: 10.1016/j.tvjl.2007.04.009.
Page R.D.M. 1996. TREEVIEW: an application to display phylogenetic trees on personal computers. Computer Applied Biosciences, 12, 357–358. DOI: 10.1093/bioinformatics/12.4.357.
Perkins S.L., Keller A.K. 2001. Phylogeny of nuclear small subunit rRNA genes of haemogregarines amplified with specific primers. Journal of Parasitology, 87, 870–876. DOI: 10.1645/0022-3395(2001)087[0870:PONSSR]2.0.CO;2.
Ramadan N.F. 1974. Morphological, experimental and taxonomic studies on protozoan blood parasite of Egyptian reptiles. PhD Thesis, Ain Shams University, Egypt, 220 pp.
Rubini D.S., Paduan K.S., Perez R.R., Ribolla P.E.M., O’Dwyer L.H. 2006. Molecular characterization of feline Hepatozoon species from Brazil. Veterinary Parasitology, 137, 168–171.
Saoud M., Ramadan N., Mohammed S., Fawzi S. 1996. On two new haemogregarines (Protozoa: Apicomplexa) from Colubrid and Elapidae snakes in Egypt. Qatar University Science Journal, 16, 127–139.
Shazly M.A., Ahmed A.K., Bashtar A., Fayed H.M. 1994. Life Cycle of Hepatozoon matruhensis Comb. Nov. 2. Gamogony and Sporogony inside the vector Culex pipiens molestus. Journal of Egyptian German Society of Zoology, 14, 323–340.
Sloboda M., Kamler M., Bulantová J., Votypka J., Modry D. 2007. A new species of Hepatozoon (Apicomplexa: Adeleorina) from Python regius (Serpentes:Pythonidae) and its experimental transmission by a mosquito vector. Journal of Parasitology, 93, 1189–1198. DOI: 10.1645/GE-1200R.1.
Smith T.G. 1996. The genus Hepatozoon (Apicomplexa: Adeleina). Journal of Parasitology, 82, 565–585. DOI: 10.1645/12-18.1.
Tamura K., Peterson D., Peterson N., Stecher G. Nei M., Kumar S. 2011. MEGA5: Molecular Evolutionary Genetics Analysis using Maximum Likelihood, Evolutionary Distance, and Maximum Parsimony Methods. Molecular Biology and Evolution, 28, 2731–2739. DOI: 10.1093/molbev/msr121.
Telford S.R.Jr., Ernst J.A., Clark A.M., Butler J.F. 2004. Hepatozoon sauritus: a polytopic hemogregarine of three genera and four species of snakes in North Florida, with specific identity verified from genome analysis. Journal of Parasitology, 90, 352–358. DOI: 10.1645/GE-3258.
Telford S.R.Jr., Telford S.R., Butler J.F. 2002. The status of Haemogregarina mansoni Sambon and Seligmann from Zamenis flagelliformis Laurenti. Journal of Parasitology, 88, 783–785. DOI:10.1645/00223395(2002)088[0783:TSOHMS]2.0.CO;2).
Telford S.R.Jr., Wozniak E.J., Butler J.F. 2001. Haemogregarine specificity in two communities of Florida snakes, with descriptions of six new species of Hepatozoon (Apicomplexa: Hepatozoidae) and possible species of Haemogregarina (Apicomplexa: Haemogregarinidae). Journal of Parasitology, 87, 890–905. DOI: 10.1645/0022-3395(2001)087[0890:HSITCO]2.0.CO;2
Thompson J.D., Gibson T.J., Plewniak F., Jeanmougin F., Higgins D.G. 1997. The CLUSTALX Windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Research, 25, 4876–4882.
Tomé B., Maia J.P.M.C., Harris D.J. 2012. Hepatozoon infection prevalence in four snake genera: influence of diet, prey parasitemia levels or parasite type? Journal of Parasitology, 98, 913–917. DOI: 10.1645/GE-3111.1.
Tomé B., Maia J.P.M.C., Harris D.J. 2013. Molecular assessment of apicomplexan parasites in the snake Psammophis from north Africa: do multiple parasite lineages reflect the final vertebrate host diet? Journal of Parasitology, 99, 883–887. DOI: 10.1645/12-95.1.
Vilcins I.E., Ujvari B., Old J.M., Deane E. 2009. Molecular and morphological description of a Hepatozoon species in reptiles and their ticks in the Northern Territory, Australia. Journal of Parasitology, 95, 434–442. DOI: 10.1645/GE-1725.1.
Wicks R.M., Spencer P.B., Moolhuijzen P., Clark P. 2006. Morphological and molecular characteristics of a species of Hepatozoon Miller, 1908 (Apicomplexa: Adeleorina) from the blood of Isoodon obesulus (Marsupialia: Peramelidae) in Western Australia. Systematic Parasitology, 65, 19–25. DOI: 10.1007/s11230-006-9036-8.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Abdel-Baki, AA.S., Al-Quraishy, S. & Zhang, J.Y. Redescription of Haemogregarina garnhami (Apicomplexa: Adeleorina) from the blood of Psammophis schokari (Serpentes: Colubridae) as Hepatozoon garnhami n. comb. based on molecular, morphometric and morphologic characters. Acta Parasit. 59, 294–300 (2014). https://doi.org/10.2478/s11686-014-0241-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11686-014-0241-3